Traumatic brain injury (TBI) is a major cause of mortality and morbidity worldwide. The medical management of TBI involves stabilization of the individual and focusing on preventing further injury. There are no proven pharmacological therapies to enhance recovery after severe TBI.
We present case reports of two patients who had severe head injury due to a motor vehicular accident, taken from the NeuroAiDSafe Treatment (NeST) Registry, which is a prospective, real world data registry initiated to monitor the use and safety of the NeuroAiD in various clinical indications. The computerized tomography (CT) brain scan of both patients showed multiple intracranial haemorrhages, subdural and contusion hematoma. Both cases required resuscitative care and ventilatory support management in the intensive care unit and subsequent medical management including intensive rehabilitation. MLC601 (NeuroAiD) was started after 3 weeks and 11 days for patients 1 and 2, respectively. Improvement in the Glasgow Coma Scale (GCS), motor and sensory functions were noted as early as one month with gradual improvement by 3rd and 6th month. There was a residual slurred speech and partial gaze paresis noted in the first patient, while the other had a full recovery. There were no adverse events reported related to MLC601. NeuroAiD may play a role in enhancing recovery after severe traumatic brain injury.
Traumatic Brain Injury (TBI) remains a global health burden. It is a major cause of mortality and morbidity, with 69 million individuals worldwide estimated to sustain a TBI each year [1]. The global burden of TBI increased from1990 to 2016 [2]. It will remain a growing public health concern and represents the greatest contributor to death and disability among all trauma-related injuries [3,4].
There is limited medical intervention to reverse the initial brain damage, prevent nerve damage or promote nerve healing after TBI [5]. Surgery may be selectively indicated if there is a large hematoma compressing the brain.
NeuroAiD originates from Traditional Chinese Medicine, and comprises natural herbal and animal components: MLC601 has 9 herbal and 5 animal components, while MLC901 only has the 9 herbal components. Both formulations have been shown to have neuro restorative and therapeutic benefits in experimental TBI [6]. The prospective, real world data registry called the NeuroAiD Safe Treatment Registry (NeST) NCT02536079 [7] was initiated to monitor the use and safety of the NeuroAiD in various clinical indications. We present two case reports from the Registry on the use of NeuroAiD in severe TBI.
A 29-year-old male admitted due to severe head injury after a motor vehicular accident (MVA). On clinical assessment, the initial Glasgow Coma Scale (GCS) was 5/15 (E1V1M3). His blood pressure was140/94, pulse rate of 103/min, respiratory rate at 14/min. His pupils were unequal. His right pupil was 3 mm reactive to light and left pupil was 6 mm unreactive to light. There were multiple facial fractures involving left maxillary and left inferior orbital fractures. CT scan showed multiple intracranial haemorrhages with contusions and right sided subdural haematoma with interhemispheric and tentorial subdural haematoma (Figure 1). There were also a C6 left facet joint fracture extending into the left lamina with presence of retropulsed fracture segment in the spinal canal, and bilateral first rib fractures.
 Figure 1: Punctate hyperdensity at both right and left frontal lobe in keeping with contusional bleeding. Extra-axial hyperdensity along the frontoparietal region in keeping with subdural bleed. It measures 0.35 cm in maximum thickness. Subdural bleed along the left tentorium cerebelli. No midline shift.
View Figure 1
Figure 1: Punctate hyperdensity at both right and left frontal lobe in keeping with contusional bleeding. Extra-axial hyperdensity along the frontoparietal region in keeping with subdural bleed. It measures 0.35 cm in maximum thickness. Subdural bleed along the left tentorium cerebelli. No midline shift.
View Figure 1
He was intubated and ventilated for 2 weeks. His head injury was managed conservatively. Tracheostomy was done in the ward. He had total gaze paresis and global aphasia during the first week. Three weeks after the head injury, he commenced on intensive physical rehabilitation, and he was started on NeuroAiD4 caps three times a day TDS via nasogastric feeding for three months. The baseline clinical assessment prior to starting NeuroAiD was GCS score of 8/15 (E2V1M5) and motor score of 2/5. Subsequently, he was discharged well after 40 days of hospital stay. He had gradual improvement as early as one month. The total gaze paresis improved to partial gaze paresis. His GCS improved to GCS score of 11 (E4V1M6). By the third month, there was improvement of motor power from 2/5 to 4/5 motor score and a GCS score of 15/15 (E4V5M6). He was able to ambulate and walk with assistance. He was also able to tolerate NeuroAiD capsule orally.
Tracheostomy was removed on the 11th month. At 18th month, he was ambulating and living independently. He had residual slurred speech and partial divergent squinting of the left eye. No adverse effects were reported throughout the follow-up.
A 17-year-old male presented unconscious after an MVA. On initial assessment, his GCS score was 4/15 (E1V1M2). His blood pressure was 154/90 mmHg, pulse rate was 94/min, respiratory rate was 25/min (tachypnoeic) and oxygen saturation was 100% under room air. Pupils were 4 mm bilaterally and not reactive to light. CT brain scan showed right sided subdural haemorrhage with interhemispheric extension, frontal contusions accompanied with multiple skull fractures. There were also tentorial subdural and traumatic subarachnoid haemorrhages (Figure 2a).
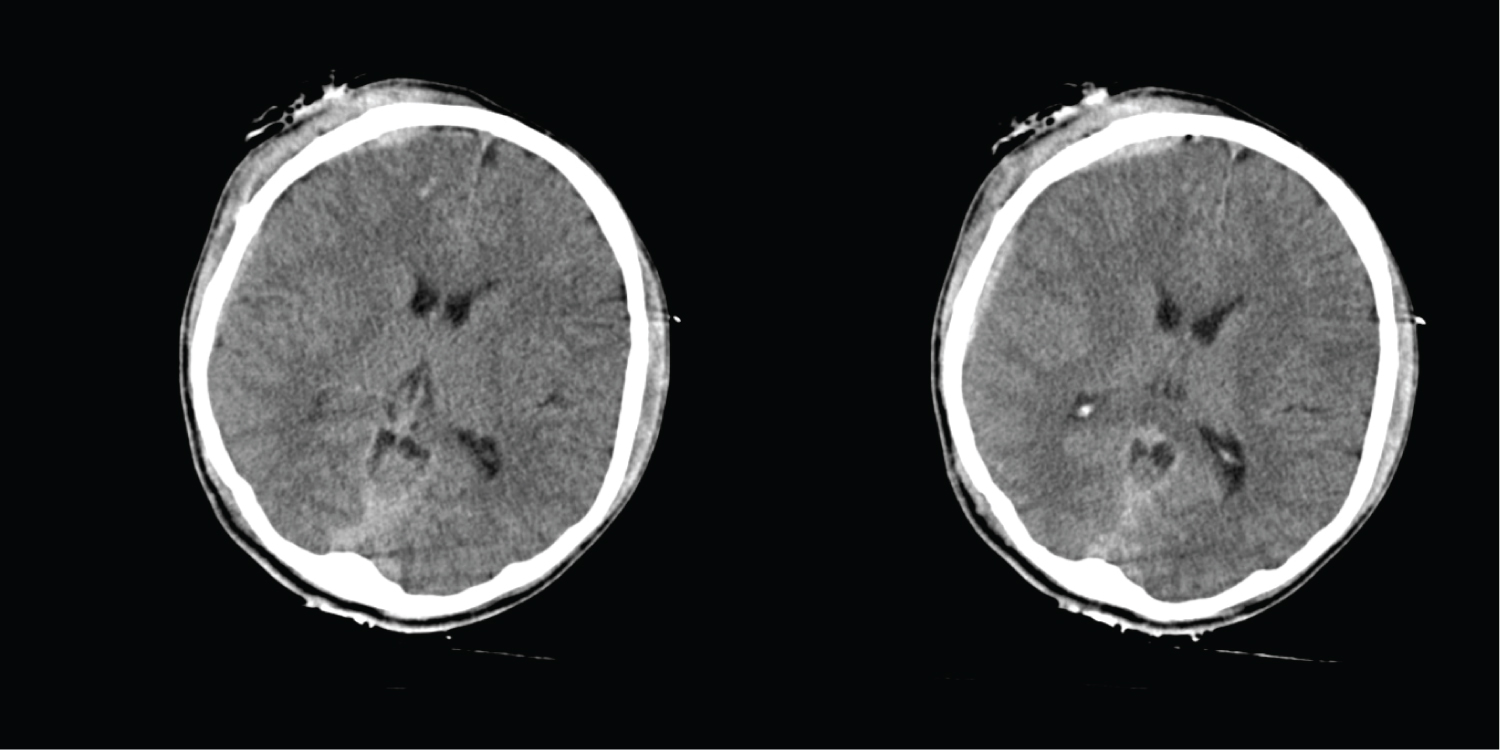 Figure 2a: (Post-trauma Day 1) There is presence of right subdural and subarachnoid bleed at right frontoparietal region. The maximum thickness of the SDH measuring 0.5 cm. There is extension of the bleed into the interhemispheric fissure, cerebellar tentorium and prepontine cistern. Multiple small contusional bleedings in right frontal lobe. There are multiple fractures involving the right frontoparietal skull vault, anterior and posterior wall of right frontal sinus, right lamina papyracea, anterior wall of left sphenoid sinus.
View Figure 2a
Figure 2a: (Post-trauma Day 1) There is presence of right subdural and subarachnoid bleed at right frontoparietal region. The maximum thickness of the SDH measuring 0.5 cm. There is extension of the bleed into the interhemispheric fissure, cerebellar tentorium and prepontine cistern. Multiple small contusional bleedings in right frontal lobe. There are multiple fractures involving the right frontoparietal skull vault, anterior and posterior wall of right frontal sinus, right lamina papyracea, anterior wall of left sphenoid sinus.
View Figure 2a
The patient was intubated and transferred to ICU. His head injury was managed medically. He was successfully weaned off from the ventilator after 4 days. However, he started developing desaturation whereby the SpO2 (oxygen saturation) dropped below 90% under room air and required tracheostomy. On post-trauma day 11, NeuroAiD was started with four capsules TDS via nasogastric feeding for three months. Baseline assessment prior to start of NeuroAiD revealed GCS score of 4/15 (E2V1M1). Intensive rehabilitation and NeuroAiD were started concurrently.
He was transferred to the general ward and continued on physical and occupational rehabilitation. He developed one episode of generalised seizure that was managed with sodium valproate. A CT brain scan was repeated one month after the head injury which showed resolution of the right subdural hematoma (Figure 2b). Patient was subsequently discharged after one-month stay in hospital. One month after starting NeuroAiD, his GCS score improved to 15/15 (E4V5M6), with improvement in his motor and sensory functions. He was able to tolerate NeuroAiD in capsule form. By the sixth month, his motor score improved from 3/5 to 5/5 on all limbs. He was able to walk independently without assistance. There were no reported adverse events.
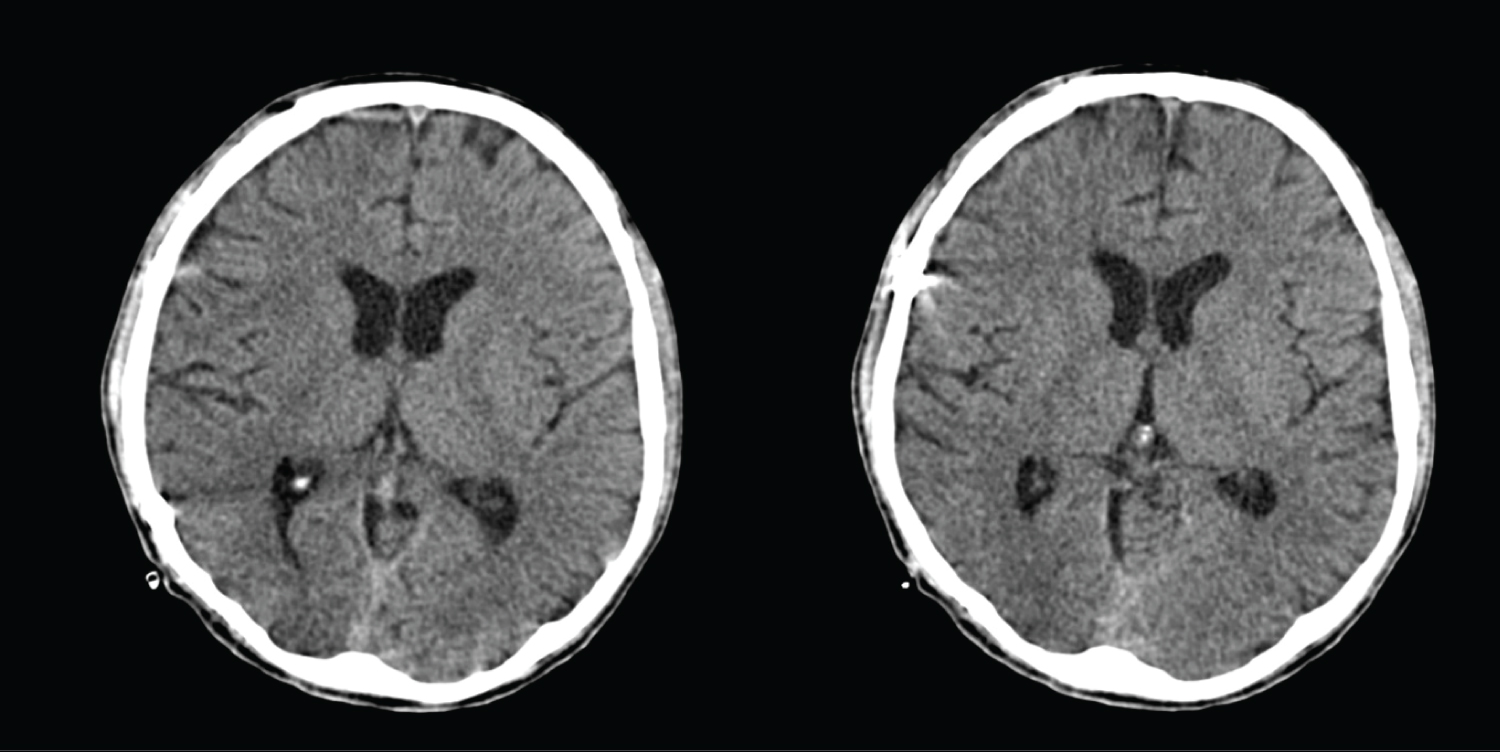 Figure 2b: (Post Trauma Month One) The previously seen right subdural and subarachnoid bleedings at right frontoparietal region are no longer seen.
View Figure 2b
Figure 2b: (Post Trauma Month One) The previously seen right subdural and subarachnoid bleedings at right frontoparietal region are no longer seen.
View Figure 2b
Outcome of patients who had a closed TBI largely depends on the initial severity [8]. Patients with severe TBI have the worst outcome. Only 25 to 33 percent of these patients have positive outcomes. Moderate and severe disability each occurs in about a sixth of patients. About 33 percent of these patients do not survive. The remaining are persistently vegetative. For those who survive, similar effects of a chronic disease can be seen, which a moderate or severe TBI may lead to long-term or life-long health problems that may affect all aspects of a person's life, including family and community. A person with a moderate or severe TBI may need ongoing care to help with their recovery [9].
Data from US TBI National Database for people at 16 years of age or older who received inpatient rehabilitation services for primary diagnosis of TBI showed that the five-year outcomes of persons with TBI includes 22% died, 30% became worse, 22% stayed same, and 26% improved [10].
TBI can induce subtle changes in molecular signalling, alterations in cellular structure and function, and/or primary tissue injury, such as contusion, haemorrhage, and diffuse axonal injury. TBI results in increased neuroinflammation brought about by damage and leakage in blood-brain barrier (BBB), which results to a secondary injury that occurs from hours to days to months after the initial trauma [11].
Standard medical and surgical interventions always play a significant role in the acute management for TBI patients. There is no proven medication to improve recovery after TBI. This calls for major research of TBI to be shifted into the area of neurorestoration and neurorehabilitation [12] to meet the need for a therapeutic agent that will help in the rehabilitative therapy for TBI patients to regain functional independence.
Herbal medicine may be a potentially useful therapeutic intervention. MLC601 shows evidence in pre-clinical studies of neuroprotective and neuroproliferative effects in rodent model of ischemia of post stroke with similar cascades of events to traumatic brain injury [13].
In preclinical studies, MLC601 has neuroproliferative properties by enhancing the expression of BDNF which lead to neuron outgrowth and proliferation. It also reduces the BAX expression reducing A1 hippocampal CA1 cell degeneration enhancing cognitive function [14]. A study of MLC601 involving experimental traumatic brain injury in rats, showed that post-TBI treatment with MLC601 significantly and dose-dependently attenuated TBI-induced cerebral contusion in rats. The beneficial effects of MLC601 were correlated with reduction in neurological and motor deficits, neuronal apoptosis, and microglial activation [15].
Preclinical study of MLC901 administration (NeuroAiD II) can increase number of neuronal cell (neurogenesis) in a rat model with brain injury [16].
Several clinical studies of MLC601 and MLC901 have been performed showing its safety and improvement in functional and cognitive assessments as compared to placebo. There are limited studies investigating NeuroAiD's effects in TBI. One study investigated Clinical Outcomes of MLC601 (NeuroAiDTM) in Traumatic Brain Injury. An Indonesian pilot study aimed to investigate clinical outcomes of MLC601 for moderate TBI [17]. This randomised controlled trial included subjects with non-surgical moderate TBI who were allocated into two groups: with and without MLC601 over three months in addition to standard TBI treatment. Clinical outcomes were measured by the Glasgow Outcome Scale (GOS) and Barthel Index (BI) observed upon discharge and at months (M) 3 and 6. Thirty-two subjects were included in the study. Outcome of the study was significant for BI which showed significant improvement for the MLC601 group compared to the control group at Month 3 and Month 6. This study showed a potential for a positive effect on clinical outcome with no adverse effects. However, the main limitation is the small sample size with an open label study design.
A study by Pakdaman, et al. investigated the MLC901 for moderate to severe traumatic brain injury [18]. In this pilot, randomised, double-masked, placebo-controlled trial, patients with a diagnosis of moderate to severe TBI were enrolled. Subjects were randomly assigned to receive either MLC901 or placebo capsules three times per day over 6 months. Eighty-one patients completed the 6-month follow-up. Evaluation of patients was carried out at baseline, 3rd month and 6th month after injury. Modified Rankin Scale (mRS) and GOS were used to examine patients. Functional outcome scales of GOS, mRS at 3rd and 6th month post-injury were significantly better in MLC901 group compared to placebo (p < 0.05). The limitation of the study is a lower sensitivity of GOS compared to GOSE in detecting functional outcome and the small sample size.
A study by Theadom, et al. was a pilot study looking at the effects of MLC901 (NeuroAiDTMII) on cognition after mild or moderate TBI [19]. Patients who had 1-12 months after mild or moderate TBI were randomized to receive MLC901 (0.8 g capsules 3 times daily) or placebo for 6 months. The primary outcome was cognitive functioning as assessed by the CNS Vital Signs online neuropsychological test. A total of 78 participants were included in the analysis. Linear mixed-effect models controlling for time, group allocation, repeated measurements, adherence and baseline assessment scores revealed significant improvements in complex attention and executive functioning at 6 months in the MLC901 group compared with controls. A limitation of the study is that safety and effects of MLC901 remain unknown for those who have experienced a severe TBI. Only 3% of participants had sustained a moderate TBI and findings need to be interpreted with caution for this population.
Our two patients, following severe traumatic head injury were started with a three-month treatment with NeuroAiD combined with rehabilitation. This is a shorter duration of NeuroAiD therapy compared with previous studies of Pakdaman and Theadom where NeuroAiD was given for 6 months [18,19]. No cognitive and functional tests were done for our cases. Still, our case reports support the safety and efficacy in post-traumatic brain injury recovery.
This study is limited to two case reports, but both cases highlighted the neurologic and functional improvements in a post-traumatic head injury. The extent of the functional and motor recovery is variable which make it hard to quantify the efficacy of MLC601.
TBI is a complex heterogenous disease process involving multiple mechanism hence the failure to show improvement in neurological outcome of a compound acting in a specific pathway. Thus, it is necessary to emphasize the importance of targeting treatments to groups selected according to TBI severity levels [20], and the benefits of compounds with multimodal effect such as NeuroAiD (MLC601/MLC901) with its multimodal effects making it possible to address disease mechanisms as potential targets for intervention.
In conclusion, given the irreversible damage induced by TBI with significant morbidity, these case reports provided support for a clinical trial of MLC901 in more severe TBI.