Background: Glioblastoma developed from cranial nerves are extremely rare. We report the case of a 71-year-old woman with glioblastoma arising from both trigeminal root entry zones and Internal Auditory Canal (IAC) nerves.
Observations: The patient presented with facial numbness and paresthesia in both trigeminal sensory territories with initial Magnetic Resonance Imaging (MRI) at 1-month follow up showing contrast enhancement and thickening of both trigeminal nerves root entry zone and moderate thickening of IAC bundle, suggesting inflammatory disease. Cranial nerves deficit and neuroimaging were rapidly evolving within few months, with appearance of multiples necrotic lesions of pons, midbrain and left cerebellar hemisphere.
Histopathological examination on cerebellar biopsy revealed glioblastoma. The patient underwent no further treatment and died one week after biopsy.
Lessons: To the author's knowledge, this is the first case of primary glioblastoma arising simultaneously from multiple cranial nerves.
Glioma, Glioblastoma, Cranial nerve, Trigeminal nerve, Internal auditory canal
GBM: Glioblastoma Multiforme; CNS: Central Nervous System; CN: Cranial Nerve; IAC: Internal Auditory Canal
Glioblastoma multiforme (GBM) is the third most frequently reported central nervous system (CNS) histology and the most common malignant brain tumor in accounting for approximately 48% of all cases [1,2]. Infra-tentorial location of GBM is rare, cerebellar GBM occurs rarely in adults accounting for 0.4-3.4% of all GBM [3-5]. While brain stem location represents less than 2.5% of all GBM [4].
Glioma primitively arising from cranial nerve (CN) and subsequently GBM, are extremely rare [6]. To our knowledge, only 11 cases of glioblastoma with CN involvement have been described (Table 1).
We report the case of a 71-year-old woman who initially presented facial numbness and paresthesia, with radiological lesions of both CN V and IAC bundle, rapidly evolving and found to be glioblastoma on histopathological analysis.
A 71-year-old woman with history of total thyroidectomy, glaucoma, hypertension and nose skin cancer (non-melanoma) presented with facial numbness in all three trigeminal sensory territories, bilaterally, including forehead, nose, lips and the front two-third of the tongue, predominating on the left side. Initial symptoms were fluctuating, with appearance of forehead and circumoral paresthesia. She had no additional symptoms, specifically no motor weakness, fever or headache.
There was no head trauma or infectious syndrome.
Neurological examination was significant for decreased sensation to light touch in all three trigeminal territories, bilaterally, static cerebellar ataxia with negative Romberg test, no oculomotor dysfunction or other sensory-motor deficit.
Gadolinium enhanced MRI at 1-month clinical follow-up demonstrated contrast enhancement and thickening of both trigeminal nerves root entry zone (Figure 1A). Both acoustic-facial bundles in IAC also showed moderate contrast enhancement (Figure 1B).
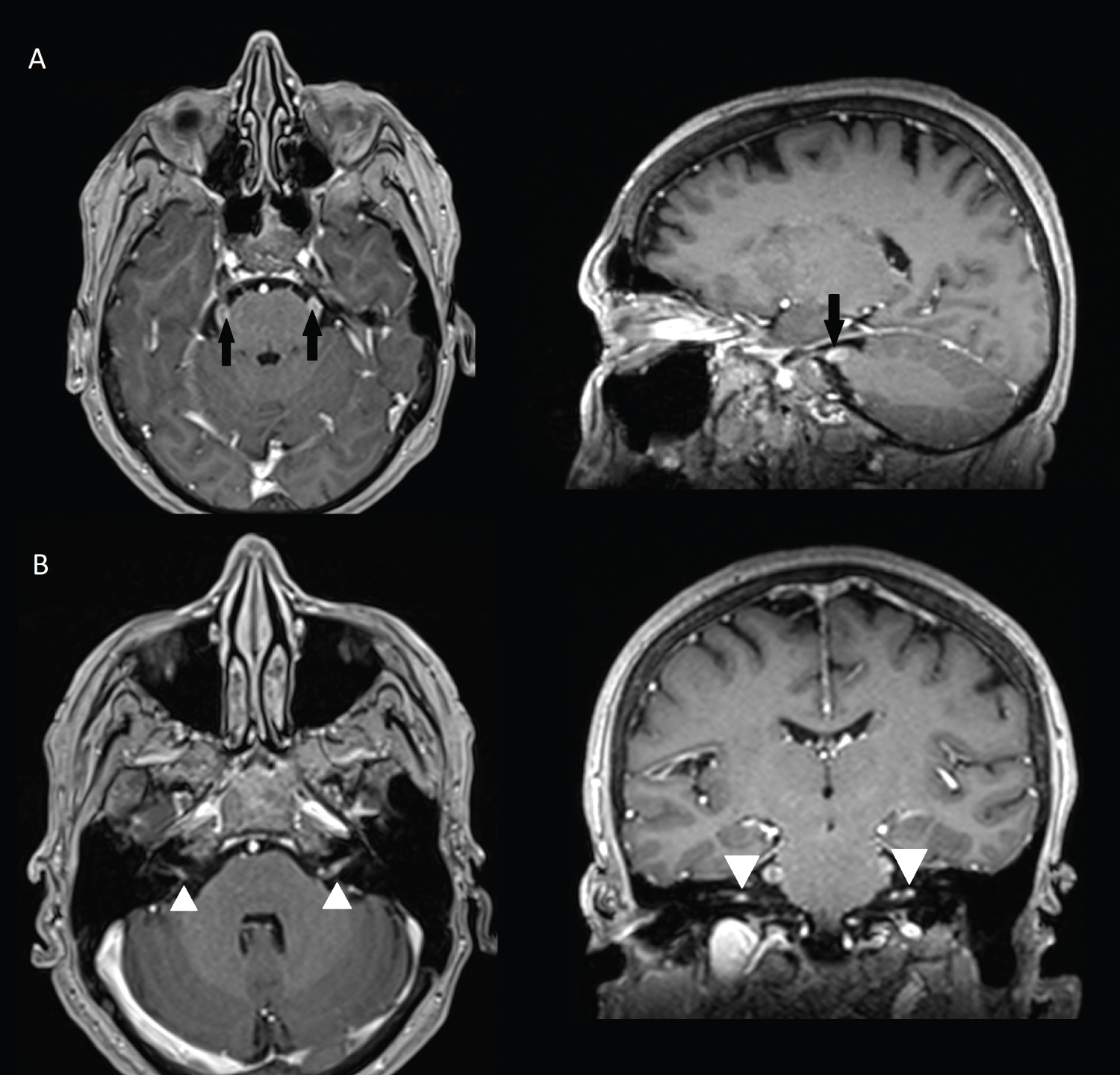 Figure 1: MRI at 1-month follow-up showing contrast enhancement and thickening of both trigeminal nerve root entry zone (1A, black arrows), and IAC bundle (1B, white arrowheads).
View Figure 1
Figure 1: MRI at 1-month follow-up showing contrast enhancement and thickening of both trigeminal nerve root entry zone (1A, black arrows), and IAC bundle (1B, white arrowheads).
View Figure 1
Laboratory examination revealed no hematological abnormality.
Cerebrospinal fluid examination revealed slightly increased protein level (0.64 G/l), normal WBC count, and no malignant cell.
Thorax, abdomen and pelvis CT-scan at two-month follow-up did not show any sign of neoplasic or inflammatory disease.
Within one month, she presented an extended CN palsy with appearance of partial anosmia and taste loss. A previous corticoid empirical treatment was tested and stopped after three days, leading to worsening of sensory symptoms.
Within four month of evolution, previous symptoms were worsening with appearance of imbalance, hearing loss and tinnitus (buzzing) and left facial paralysis.
PET scan revealed high metabolic activity of left cerebellar hemisphere extending to the pons (SUV max 16.2).
Second gadolinium enhanced brain MRI at 5 month clinical follow up (Figure 2) showed the appearance of multiple necrotic lesions of pons, midbrain and left cerebellar hemisphere, with rapid growth and confluence of lesions on MRI one month later (Figure 3).
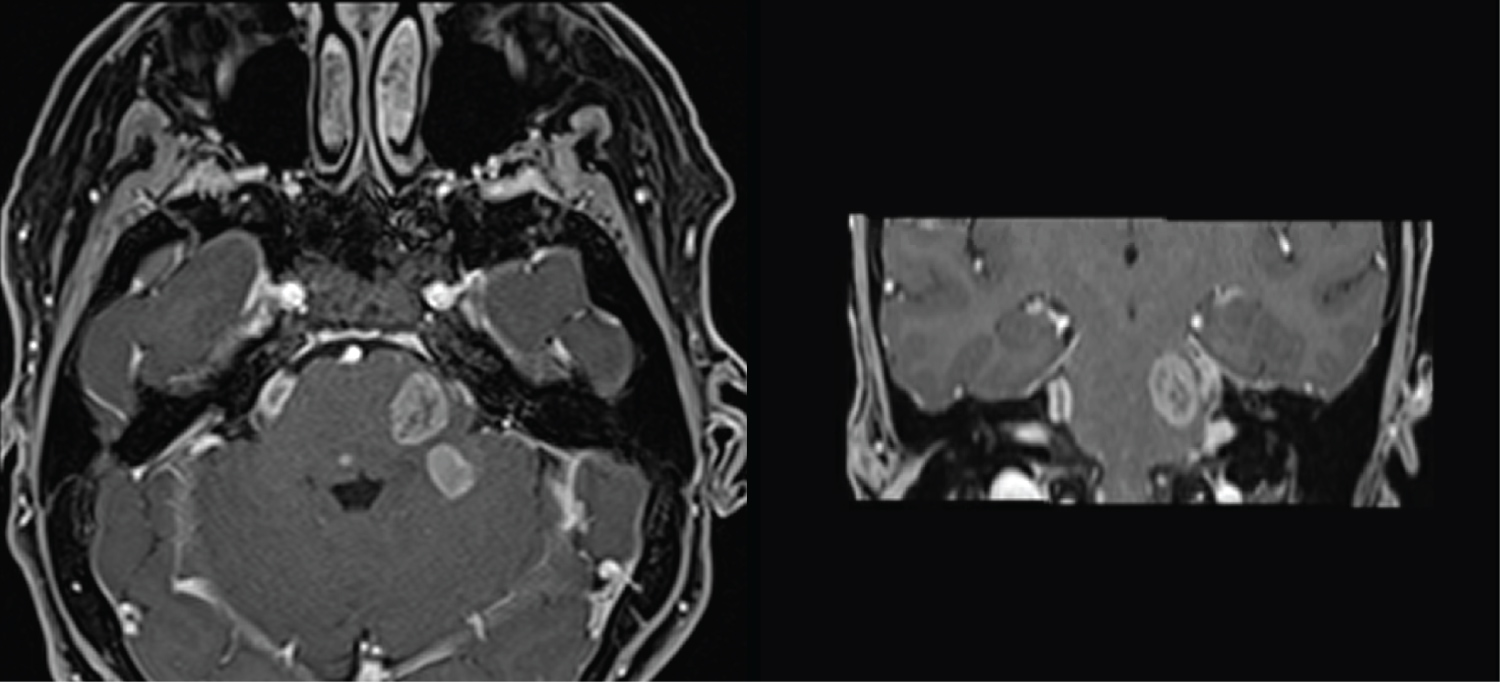 Figure 2: MRI at 5-month follow up, with appearance of multiple necrotic lesions of pons, midbrain and left cerebellar hemisphere.
View Figure 2
Figure 2: MRI at 5-month follow up, with appearance of multiple necrotic lesions of pons, midbrain and left cerebellar hemisphere.
View Figure 2
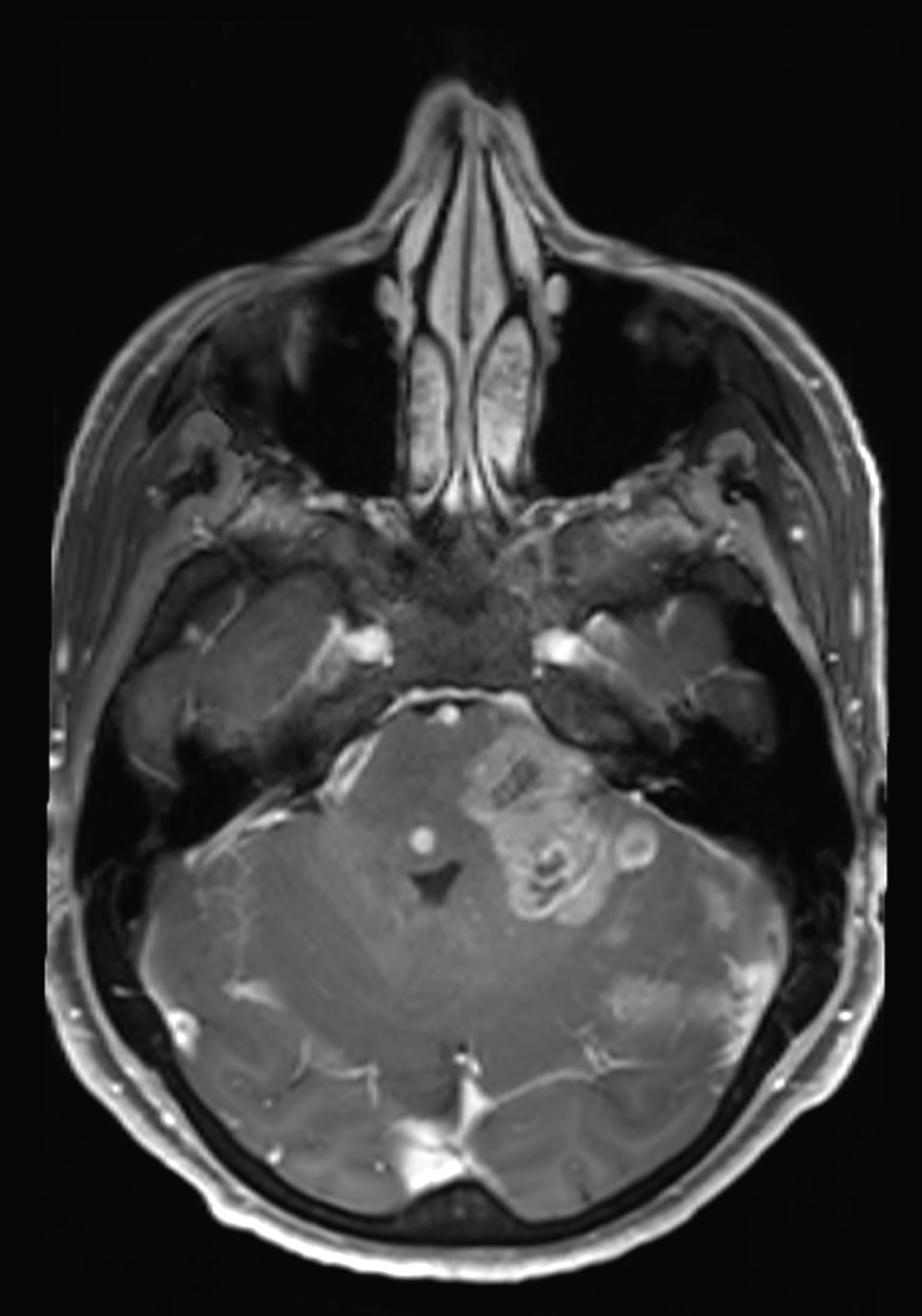 Figure 3: MRI at 6-month follow up, with rapid growth and confluence of lesions.
View Figure 3
Figure 3: MRI at 6-month follow up, with rapid growth and confluence of lesions.
View Figure 3
Proton MR Spectroscopy revealed increased choline and non-decreased N-acetylaspartate within lesions, not really indicative of glial type neoplasm (Figure 4). Perfusion MRI revealed high hyperperfusion of the lesions, with relative cerebral blood volume (rCBV) estimated between 3 and 5.2.
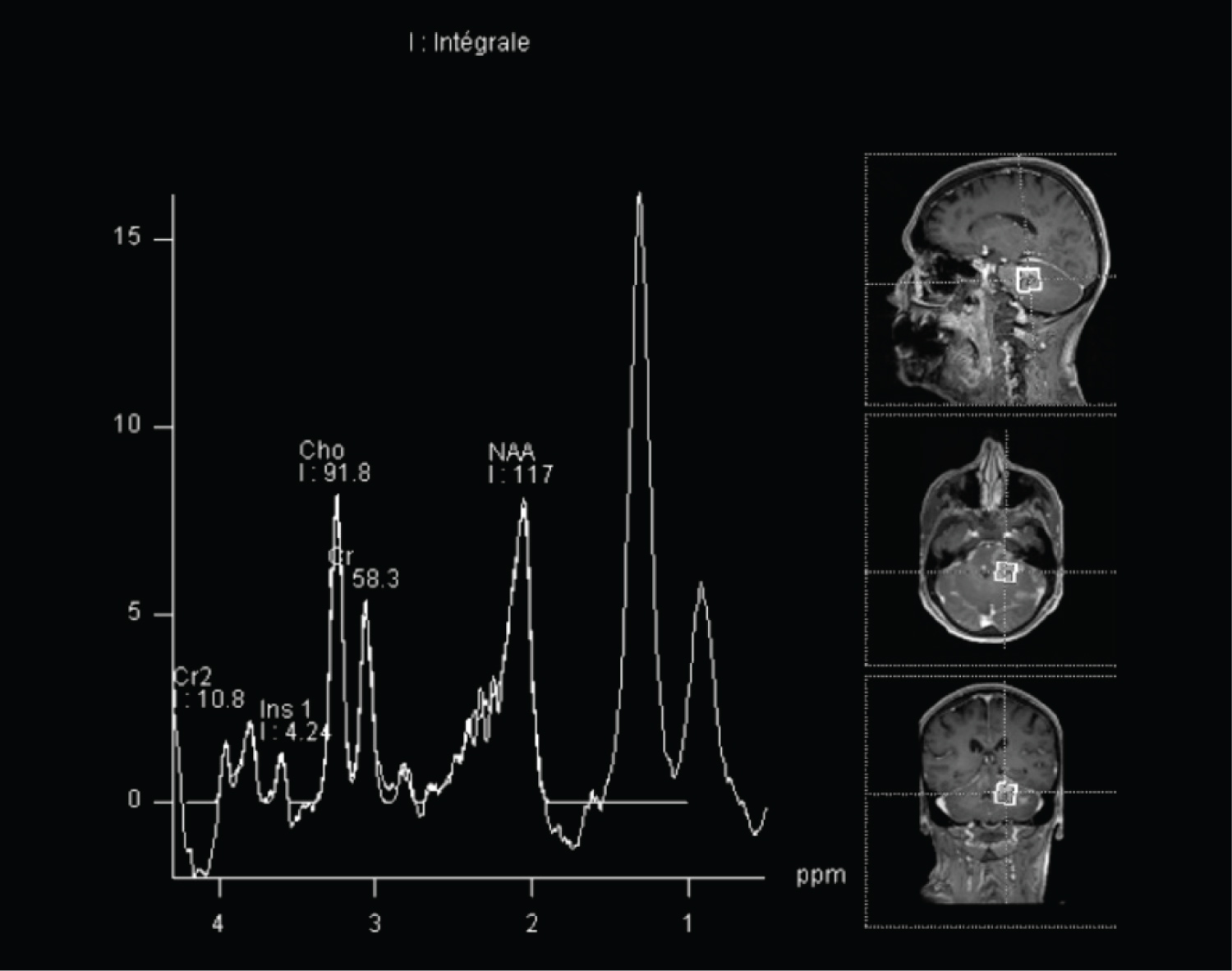 Figure 4: Single-voxel spectroscopy at TE 30 ms, showing increased choline et non decreased NAA.
View Figure 4
Figure 4: Single-voxel spectroscopy at TE 30 ms, showing increased choline et non decreased NAA.
View Figure 4
The main diagnostic suggestion was the metastatic evolution of previous nose skin cancer treated several years ago, thought to be squamous cell carcinoma (histological analysis unavailable). Other differential diagnosis included inflammatory disease such as neurosarcoidosis.
A biopsy of cerebellum lesions was performed via occipital craniotomy. Necrotic and hemorrhagic liquid was encountered and sampled. Multiple cerebellar samples were performed and sent for further histopathologic analyses.
Postoperative course: The patient died one week after cerebellar biopsy, before the beginning of adjuvant treatment.
Histopathological findings: Histopathological analyses of tissues confirmed the diagnosis of high-grade glioma, with immunohistochemical profile suggesting glioblastoma IDH wild type (WHO grade IV).
Tissue sections demonstrated highly cellular lesion with astrocytic phenotype cell populations, high mitosis activity, focal microvascular changes, necrosis and hemorrhagic deposits.
Immunohistochemical analyses in tumor cells were positive for glial fibrillary acidic protein (GFAP) and oligodendrocyte transcription factor (OLIG2), consistent with glial type neoplasm.
Tumor cells maintained ATRX expression and were negative for IDH1 R132H and AE1/AE3, no P53 overexpression. Ki-67 proliferation index was estimated between 25 and 30%.
Molecular analyses revealed PIK3R1 mutation (undetermined signification), probable homozygous deletion of CDKN2A and probable EGFR amplification. No mutation of IDH1, IDH2, H3F3A or HIST1H3B were identified.
Few cases of glial tumors arising from CN remain extremely rare with only 11 cases of GBM reported (Table 1).
Table 1: Summary of reported cases for glioblastoma involving cranial nerves. View Table 1
Reifenberger and more recently Marchesini described GBM arising from oculomotor cranial nerve [7,8].
Takami, Wu and Yang described GBM at the pontocerebellar angle involving CN VIII [9-11]. Breshears reported extra-axial GBM arising from trigeminal nerve root entry zone and several cases were exposed by Mabray, involving CN III, V, VII and VIII [6,12].
According to previous reported cases, the repartition of primary glial lesion developed from CN seemed to be correlated with the length of the root entry zone, thus more frequent in CN VIII [6,13].
Although extra-axial primary GBM in this case could not be proven by surgical exploration and histopathological analysis, the clinical and radiological evolution on 6 month follow-up suggest a primary glial neoplasm arising simultaneously from multiples CN.
This case is the first report of glioblastoma primarily developed from multiples CN (trigeminal and acoustic-facial bundle) with bilateral involvement and secondary intra-axial extension.
Several hypothesis were made regarding the origin of extra-axial CN involving GBM, including malignant transformation of glial cells located to a "transition zone" from central to the peripherical nervous system in proximal CN [6,13], or transformation of heterotopic glial cells located in the pia at nerve's exit point [14]. A third theory suggest a primary brainstem GBM extending to cranial nerves with no clinical or radiological manifestation on initial examination [7,12], which could be more appropriate in this case.
In conclusion, the primary origin of glial lesion in cranial nerve is difficult to prove. However, in case of atypical lesions, an extended evaluation including multiparametric MRI and surgical exploration in CN evolutive lesions could lead to early treatment to impact short and medium-term prognosis.
The authors report no conflict of interest concerning the materials or methods used in this study or findings specified in this paper.