Breast carcinoma secondarily involving the uterus is very rare. It usually occurs as a manifestation of widespread disease. There are few case reports available in the English language literature on this seldom seen occurrence.
We report 2 cases of breast carcinoma metastatic to the uterus.
Patient A was a 47-year-old African American lady admitted for thrombocytopenia. A mammogram done a few weeks prior to admission was reported as BIRADS 4 for the right breast. She had a biopsy done as an inpatient which revealed invasive lobular carcinoma. Due to persistent vaginal bleeding a gynecological consult was requested; accordingly, endometrial curettage was done with pathology reported as metastatic lobular carcinoma of the breast. Further testing confirmed a diagnosis of stage 4 carcinoma of the breast. The patient died two months later.
Patient B was a 59-year-old Caucasian lady who was referred to gynecologic oncology for evaluation of post menopausal bleeding. She had a history of stage IIIA, grade 1 lobular cancer in the left breast, in remission for 7 years. A recent CT scan showed an enlarged lobular uterus and irregular cervix. She underwent dilatation and curettage with endometrial biopsy and cervical biopsy. Pathology report showed metastatic breast lobular carcinoma of the uterus.
The morphology and immunophenotype of the endometrial samples revealed the neoplastic nature of the endometrial lesions and confirmed their origins from the breast in both cases.
Atypical bleeding in patients with known breast carcinoma should prompt screening for endometrial metastasis by a gynecologist. Metastasis to the uterus carries a grim prognosis.
Extragenital carcinomas secondarily involving the uterus are very rare [1]. There are few case reports available in the English language literature on this seldom seen occurrence. In the majority of the reported cases the site of origin was the breast [2,3].
A 47-year-old African American lady was admitted for thrombocytopenia. A mammogram done a few weeks prior to admission was reported as BIRADS 4 for the right breast. She had a biopsy done as an inpatient which revealed invasive lobular carcinoma. The tumor was estrogen and progesterone receptor positive, HER-2/neu negative. A previous screening mammogram was done two years before this and it was negative for pathology. She was started on hormonal therapy with Arimidex. A Gynecological consult was requested for abnormal vaginal bleeding and anemia. Pelvic ultrasound showed multiple uterine fibroids with endometrial thickness 15.7 mm. Endometrial curettage was done. Pathology assessment of the endometrial tissue specimen revealed metastatic lobular carcinoma of the breast to the endometrium (Figure 1, Figure 2 and Figure 3). As this was a rare phenomenon, a second opinion was obtained which concurred with the initial assessment. In more detail, the histologic sections showed scattered benign endometrial glands. A malignant process had overrun the majority of the stromal component, consisting of discohesive small to medium sized cells which frequently contained intracytoplasmic lumina. These cells were positive for pan-cytokeratin (AE1/AE3) and GATA3. Endometrial curettage was helpful in alleviating her abnormal bleeding; although this effect was temporary. She eventually had bilateral uterine artery embolization for recurrent severe uterine bleeding during a subsequent admission.Further testing including imaging studies and bone marrow biopsy confirmed a diagnosis of stage 4 carcinoma of the breast. The plan was to start chemotherapy; however patient died from disseminated disease two months after the diagnosis.
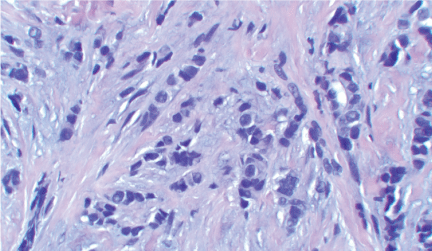 Figure 1: Neoplasm showing an "Indian file" infiltrative pattern composed of discohesive small to medium sized cells, with a background desmoplastic reaction (hematoxylin-eosin, original magnification X400). View Figure 1
Figure 1: Neoplasm showing an "Indian file" infiltrative pattern composed of discohesive small to medium sized cells, with a background desmoplastic reaction (hematoxylin-eosin, original magnification X400). View Figure 1
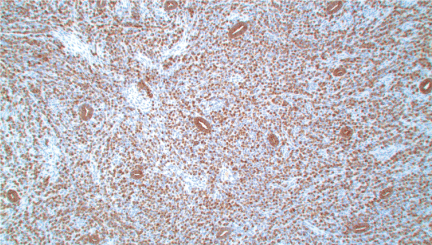 Figure 2: Positive staining of both benign endometrial glands and neoplastic cells infiltrating the endometrial stroma by pan cytokeratin (AE1/AE3), immunohistochemical stain (original magnification X100). View Figure 2
Figure 2: Positive staining of both benign endometrial glands and neoplastic cells infiltrating the endometrial stroma by pan cytokeratin (AE1/AE3), immunohistochemical stain (original magnification X100). View Figure 2
This is a 59-year-old Caucasian lady with a history of sage IIIA, T2N2aM0, grade 1 lobular cancer in the left breast which was in remission for 7 years. She presented with post-menopausal bleeding, with CT findings of an enlarged uterus and irregular cervix. 8 years previously, she had felt a lump in her left breast; fine needle aspiration done at that time showed the presence of atypical cells. She then had a lumpectomy which revealed a 4 cm invasive lobular carcinoma with margin 1 mm. The tumor was Estrogen and progesterone receptor positive and HER-2/neu negative. Due to the high chance of bilateral breast involvement, the patient underwent left modified radical mastectomy and right simple mastectomy with adjuvant chemotherapy. This consisted of adriamycin plus cytoxan followed by paclitaxel. She was then started on tamoxifen for 3 years which was switched to aromasin for the last 4 years. Patient was referred to gynecologic oncology for evaluation because she reported post-menopausal bleeding for a few weeks with abnormal CT findings as mentioned above. She underwent dilatation and curettage with endometrial biopsy and cervical biopsy. Pathology report showed metastatic breast lobular carcinoma to the uterus.
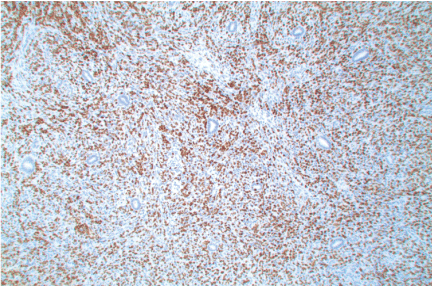 Figure 3: Positive staining of metastatic mammary lobular carcinoma cells within the endometrial stroma by GATA3 immunohistochemical stain. Benign endometrial glands and stroma cells negative (original magnification X100). View Figure 3
Figure 3: Positive staining of metastatic mammary lobular carcinoma cells within the endometrial stroma by GATA3 immunohistochemical stain. Benign endometrial glands and stroma cells negative (original magnification X100). View Figure 3
Uterine metastases of malignant tumors are rare [4]. Usually abnormal uterine bleeding is the first sign of uterine metastases in a woman with a history of malignant disease [2]. A secondary location in the uterus can occur many years after the diagnosis and treatment of primary breast cancer [5].
The extragenital tumors most often metastasizing to the uterine corpus are breast carcinoma (47.3%), stomach carcinoma (29%), and melanoma (5.4%) [1]. Lobular infiltrating carcinoma of the breast is the histological type that most frequently metastasizes to the uterus [2,4,6]. However, infiltrating ductal carcinoma of the breast has also been reported to metastasize to the uterine corpus [7-10].
Breast cancers spread by contiguity, lymphatic channels and blood-borne metastases. The most common disseminated sites are the lungs, bone and liver, whereas uterine involvement by metastatic breast disease is rare [2,3]. Stemmermann suggested that the most uterine metastases are secondary to local retrograde lymphatic spread from preceding ovarian metastases, while spread is probably haematogenous when isolated uterine metastases are found [5].
When the uterus is attacked by breast cancer, infiltration of the endometrium appears as common as involvement of myometrium [7]. Isolated endometrial metastases in the absence of myometrial involvement are uncommon [10]. When the endometrium is involved, the tumor usually tends to infiltrate the stroma, sparing the endometrial glands [2].
The histologic diagnosis of metastatic breast lobular carcinoma could be difficult, because fairly uniform cells might be mistaken for endometrial stromal cells with decidual changes due to tamoxifen therapy. Signet ring carcinoma and endometrial carcinoma should also be considered in the differential diagnoses. A positive immunohistochemical stain for hormone receptors suggests metastatic breast carcinoma, but GCDFP-15 is more specific for breast carcinoma, especially if signet ring cells are present [11]. GATA3, a recent breast immunohistochemical stain, was found to be more sensitive than GCDFP-15 in metastatic breast carcinomas [12].
Distinguishing metastatic carcinoma from primary endometrial carcinoma is important, since the treatment of these carcinomas is different. When uterine metastasis from breast carcinoma is diagnosed, surgical intervention does not seem to be indicated, because the disease is usually wide-spread and involves other extragenital sites as well. Palliation and conservative management were advised for metastatic breast disease, in view of the fact that metastases to the uterus and to other organs of the genital tract can be considered as a pre-terminal event [13]. On the other hand, when primary endometrial carcinoma is diagnosed, surgical intervention is indicated as part of the primary treatment, even in patients with a history of breast carcinoma. In one of our cases, the patient was treated with palliative and conservative management, and died from disseminated disease two months later.
Atypical bleeding in patients with known breast carcinoma should prompt screening for endometrial metastasis by a gynecologist. Metastasis to the uterus carries a grim prognosis.