A 26-year-old woman (gravida four, para two) delivered an immature infant at 23 weeks of gestation after presenting with abnormal vaginal blood loss and sepsis. Blood cultures and placenta cultures showed bacteremia by Fusobacterium necrophorum. Histopathological examination of the placenta was concurrent with that, showing severe chorioamnionitis with funisitis. A dentist and oral surgeon confirmed the diagnosis of generalised periodontal disease.
Periodontal disease is the most prevalent infectious disease in pregnancy and is associated with other clinical conditions, like cardiovascular disease and diabetes. Pregnancy aggravates periodontal disease due to change in hormones and inflammatory factors. Periodontitis seems to be associated with adverse pregnancy outcome, like low birth weight, prematurity and pre-eclampsia. Treatment of periodontal disease in pregnancy is safe and may reduce adverse pregnancy outcome. With awareness and education of oral hygiene, the incidence of periodontal disease in the most vulnerable patients with low socioeconomic status could decrease.
We present a case of a generalised periodontal disease in pregnancy causing bacteremia with F. necrophorum and immature delivery as a result. Dental screening is not yet a standard procedure in preconceptional health care. However, health care workers should be aware of possible systemic and subsequently obstetric pathology caused by poor oral hygiene. Research on the effects of oral hygiene before and during pregnancy and the benefits of early treatment should be continued.
Periodontitis, Periodontal disease, Pregnancy, Intrauterine infection, Histologic chorioamnionitis, Preterm birth, Immature birth, Septicemia, Preterm premature rupture of membranes (PPROM), Fusobacterium necrophorum
Periodontal disease is a destructive chronic inflammatory disease of bacterial aetiology affecting the periodontium [1]. The most common manifestations are gingivitis and chronic periodontitis. Periodontal disease can be caused by different bacterial species. Since the clinical picture is heterogeneous, each patient needs to be assessed individually in order to appropriately diagnose and manage the periodontal disease [1]. Periodontal disease in pregnancy is associated with increased risk of preterm birth and low birth weight [2]. Preterm birth and low birth weight are associated with increased risk of neurodevelopmental disabilities, behavioural disorder, cognitive impairment and death [2]. There are many risk factors for adverse pregnancy outcomes, but most often the risk factor remains unknown [2]. Therefore, the American Academy of periodontology considers pregnancy a systemic condition of which patients may benefit from co-management by referral to the dentist or the periodontist [3]. This case report illustrates that periodontal disease in pregnancy can result in maternal septicemia and fetal demise. Secondly, we provide an overview of the literature on pregnancy and periodontal disease and prevention.
A 26-year-old Caucasian women (gravida 4, para 2) was referred in first trimester to the obstetric department because of an abnormal psychosocial situation. At 22 weeks and 5 days of gestation, she presented with heavy vaginal blood loss. Hemoglobin level was 5.1 mmol/L (8.2 g/dL), showing moderate to severe anemia. Since the placenta was closely inserted to the internal cervical ostium, a marginal placenta insertion was initially thought to be the cause of the bleeding. Haemoglobin levels of 5.7 mmol/L were reached after two packed cells to prevent clinical complications. After two days in hospital, she developed fever (39.0 ℃) with chills. Blood investigations showed elevated infectious parameters, C-reactive protein of 165 mg/L and leukocytes of 24.0/nL. Vaginal ultrasound showed anhydramnios as a result of immature pre-labour rupture of membranes. There was no sign of the previously mentioned low insertion of the placenta, which had probably been an artefact on ultrasound caused by a hematoma. Amoxicillin/clavulanic acid was started intravenously after blood and urine cultures were taken. Maternal sepsis was diagnosed with intrauterine infection and anhydramnios by spontaneous rupture of membranes resulting in an extremely poor prognosis for the fetus. In order to treat and save the patient, the patient gave her consent for induction of labour after the medical condition was explained to her. After administration of mifepristone and misoprostol, she delivered of a lifeless immature girl of 425 grams and the placenta followed easily. The condition of the mother remained stable over time. Serologic assessment showed no recent infection by Toxoplasma, Rubella, Cytomegalic virus or Parvovirus. Herpes simplex IgG and IgM were both negative. Epstein Barr Virus was not tested. Histopathological examination of the placenta showed severe chorioamnionitis with funisitis in two umbilical vessels (Figure 1 and Figure 2). Blood cultures and placenta cultures were positive for Fusobacterium necrophorum. After consultation with the medical microbiologist, the final diagnosis was septicemia, most likely from severe periodontitis. Assessment of the oral cavity by an oral surgeon and a dentist confirmed severe periodontitis (Figure 3). Measurements of pocket depth were not executed because of the presentation of very severe generalised periodontitis. Patient had not been for dental check-up for many years.
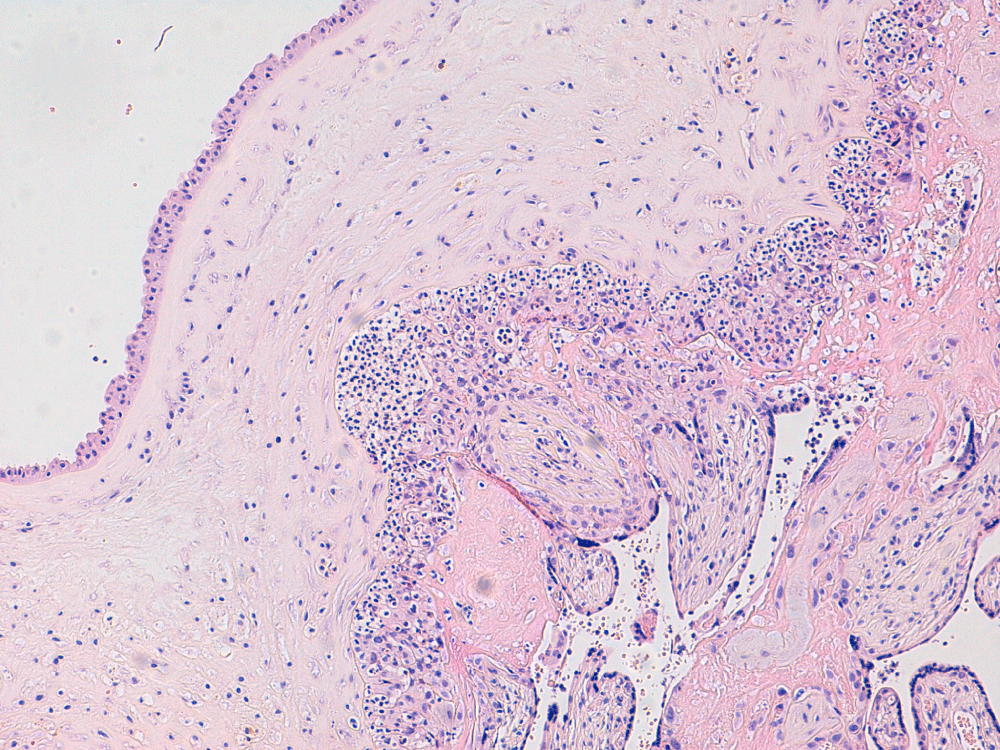 Figure 1: Chorioamnionitis with massive influx of neutrophils in the chorionic plate. View Figure 1
Figure 1: Chorioamnionitis with massive influx of neutrophils in the chorionic plate. View Figure 1
 Figure 2: Infiltration of neutrophils in the wall of an umbilical vessel. View Figure 2
Figure 2: Infiltration of neutrophils in the wall of an umbilical vessel. View Figure 2
 Figure 3: Multiple elements with advanced tooth decay and several lucent apical lesions. View Figure 3
Figure 3: Multiple elements with advanced tooth decay and several lucent apical lesions. View Figure 3
Literature search was executed using PubMed, Cochrane Library, Google Scholar and using references of articles found. Search terms used were 'pregnancy' OR 'pregnant' OR 'pregnan*' combined with 'gingival' OR 'gingivitis' OR 'pregnan*' OR 'periodontitis' OR 'periodontal' OR 'periodontal disease' OR 'periodont*'. Limitations were language in English or Dutch, studies on human, and published in the last ten years. Older articles with adequate references were included as well.
Different bacterial species may be involved in periodontal disease. Concerning F. necrophorum, there are two main reasons for difficulty in reviewing the literature. The first issue is the different names of F. necrophorum used in literature and in time [4]. F. necrophorum (also referred to as Bacillus necrophorus, Bacillus symbiophiles, Actinomyces necrophorus or Bacillus funduliformus) is a much more common and important pathogen in animals than in humans [4]. Secondly, inadequate culture techniques cause that pathology could be easily misattributed to other organisms, like streptococci [4]. Even today, it is hard to differentiate F. necrophorum from normal throat flora . The incidence of Fusobacterium spp was estimated around 0.55:100.000 population per year, of which 25% classified as F. necrophorum [5,6]. In 2012 Bohrer, et al. presented a case of severe chorioamnionitis with term gestation and intact membranes caused by Fusobacterium nucleatum [7]. Just like Fusobacterium nucleatum, Fusobacterium necrophorum (the bacterium presented in this case report) is a strictly anaerobic gram negative non-spore forming rod [4]. Recently, there is still no evidence that F. necrophorum is part of the normal commensal human oral flora, but other species or subspecies of Fusobacterium spp. were found [4]. In the healthy population F. necrophorum is rare, but in the population with tonsillitis and peritonsillar abscess, cultures show disproportionally high proportion of anaerobes, including F. necrophorum [4]. It is most often seen in human septicemic illness after tonsillitis in previously fit young adults (13-40 years) leading to high morbidity and even mortality in incidental cases [4,5,8,9]. The ability of F. necrophorum to invade and cause severe pathology due to metastatic foci, is very remarkable [9]. Only 1% of the bacteremia caused by anaerobic gram-negative bacteria is due to F. necrophorum. Still, it is clinically important because it is related to severe septicemic infections better known as necrobacillosis, postanginal sepsis or Lemierre's syndrome [4]. Already before 1900 the first case of systemic infection with F. necrophorum was described involving a young child where the bacterium was isolated from the knee in a septic arthritis combined with cerebral abscesses and chronic purulent otitis [4]. Clot formation is stimulated by the pathogenic F. necrophorum, resulting in clinical presentations with thrombosis, thrombophlebitis and septic emboli, with possible metastatic abscesses [4]. Infection could arise from the female genital tract, urinary tract or gastrointestinal tract [4]. Recently, attention is growing for the combination of well-known viruses (like Epstein Barr Virus or other members of the herpes family) and host factors (like thrombophilia) possibly increasing pathogenicity of periodontal bacteria in the maternal chronic periodontal disease. More research is needed to draw more specific conclusions [10].
Periodontal disease is very common in the general population (prevalence 35-50 percent). It is the most common infectious disease in pregnancy (prevalence 36-100 percent). Periodontal disease is characterised by erythema, edema, hyperplasia and bleeding [1,10-14]. Incidence of periodontal disease vary due to different definitions used in literature. The numbers are higher in the lower socioeconomic and lower educated population [15,16]. There is a difference in susceptibility for periodontal disease, suggesting individual differences in intrinsic and acquired risk factors. This leads to different risk assessments for current periodontal status and future periodontal disease, for example cross-sectional research on pregnancy gingivitis showed 89% in Ghana, 86.2% in Thailand and 47% in Brazil [1,12]. Since the 1960s, interest has grown in the possible association of periodontal disease and systemic diseases, like arteriosclerosis, cancer, rheumatoid arthritis, aspiration pneumonia and adverse pregnancy outcome, like pre-eclampsia, low birth weight (< 2500 grams) and preterm birth, due to infection and inflammation [12,14,17]. However, although periodontal disease in pregnant women histologically and clinically have the same appearance as in non-pregnant women, pregnant women are more vulnerable for severe periodontal disease [11]. The condition of the gingiva in the first term of the pregnancy and after labour is better than in the second and third term of pregnancy. The gingiva is most frequently affected by edema, hyperplasia, erythema and increased bleeding [11,12,13]. During pregnancy many physiological adaptations occur, like increase of molecules (like TNF-α PGE2), gram negative bacterial overgrowth, a suppressed immune system and the rise of sex hormones. Increased inflammatory mediators could result in increased severity of (already existing) periodontal disease [11,18]. Although the state of severe periodontal disease is self-limiting after labour, increased bleeding in gingivitis and gingival crevicular fluid production may cause bacteremia and elevated serum levels of pro-inflammatory cytokines [19]. Periodontal disease and locally produced inflammatory mediators could stimulate acute-phase response when entering the systemic circulation affecting fetus and placenta by leading to an impaired nutrient transport and pre-eclampsia [17,20]. Periodontal pathogens enter the circulation due to tissue damage (chewing or toothbrushing), causing transient bacteremia and dissemination to the fetal circulation by crossing the placenta [17,20,21]. Han, et al. were able to simulate transient bacteremia from periodontal disease with F. nucleatum by intravenously injecting these bacteria in pregnant CF-1 mice and mimicking chorioamnionitis. This transient bacteremia resulted in stillbirths, premature delivery and non-sustained life births [22].
A large descriptive epidemiologic study in the United States showed no difference between periodontal status in pregnant or non-pregnant women of reproductive age (15-44 years) but did show differences in sociodemographic status [23]. Poor oral health and periodontal disease are more common in women with low education, in racial/ethnic minorities and women with low family income, regardless of their pregnancy status [23]. A meta-analysis by Figuero, et al. confirmed a significant increase of periodontal disease throughout pregnancy compared to postpartum or non-pregnant women [13]. Soucy-Giguère, et al. examined amniotic fluid of pregnant women between 15 and 24 weeks of gestation who had amniocentesis done for prenatal karyotyping [24]. They measured intra-amniotic concentration of pro-inflammatory markers matrix metalloproteinase-8 (MMP-8) and interleukin-6 (IL-6) which were two to three times higher in pregnant women with periodontal disease. This result suggested an association between periodontal disease and intra-amniotic inflammation (not significant). They found a possible association between periodontal disease and pre-eclampsia, but not between periodontal disease and preterm birth. Subsequently, periodontal examination was done and they found a significant association between periodontal status and pre-eclampsia but not with preterm birth [24]. Abati included 750 Italian women who delivered prematurely, small for gestational age, low birth weight, experienced pre-eclampsia/pregnancy induced hypertension or premature prelabour rupture of membranes [25]. Within 5 days postpartum they examined periodontal status and did not see an association between periodontal status and adverse pregnancy outcome. Unfortunately they did not have any information about the periodontal status before or during pregnancy [25]. Due to a high variability in parameters, definitions, recruitment and assessment of study populations, meta-analyses are hard to perform [21,26]. A systematic review of Ide and Papapanou showed that maternal periodontitis is significantly associated with pre-eclampsia and modestly, but significant, with preterm birth and low birth weight [26].
In the general population, the most effective strategy would be to improve mouth cleaning and alter smoking behaviour [16]. Periodontal disease will rarely cause social embarrassment or isolation due to poor oral health related quality of life [16]. The findings of Azofeifa, et al. show that especially pregnant women of 15-24 years old with less than high school education and those from minority race/ethnicity report a lower percentage of visiting a dental health worker in the previous year, compared to non-pregnant women [23]. Fear of dental treatment, the perception of having no oral health issues and socioeconomic barriers (like time or costs) might play a role in not visiting the dental health worker on regular basis [23]. Stelmakh reported that Dutch pregnant women are able to perceive significant differences in periodontal status before and during pregnancy [27]. The Dutch pregnant women perceived differences like increase in bleeding gums or painful swollen gums. Fifty percent of the questioned women who had disclosed (planned) pregnancy to their dental care professional did not receive specified information on periodontal health during pregnancy [27]. There is a lot of confusion caused by the lack of consensus between experts, but there is a high morbidity and mortality caused by periodontal disease when systemic disease leads to harming the fetus in pregnancy. Therefore effective improvement of oral health is important to reduce systemic risks of bacteremia and chronic local inflammation, irrespective of the stage of pregnancy [11].
Geisinger found a significant decrease in gingival disease of pregnant patients undergoing intensive dental prophylaxis and oral hygiene instructions between 16 and 24 weeks of gestation 8 weeks after treatment [19]. They highlighted the need for large scale prospective interventional trials to determine the effect on pregnancy outcome [19]. Polyzos, et al. published a systematic review and meta-analysis of randomised controlled trials before 2011 to examine whether treatment of periodontal disease with scaling and root planning during pregnancy is associated with a reduction in the preterm birth rate [28]. They concluded that treatment of periodontal disease with scaling and root planning during pregnancy was safe but did not reduce the risk of preterm birth and should not be routinely recommended [28]. Penova-Veselinovic concluded that there was an association between small-for-gestational-age-infants and the severity of periodontitis [29]. However, they did not observe differences in the pregnancy-related outcome although post-treatment clinical dental parameters were improved [29]. Randomized trials of low methodological quality tend to overestimate the effect of treatment whereas high quality trials provide strong evidence that no significant effect of treatment exists [28]. The women who received comprehensive periodontal treatment, including multiple sessions of scaling and root planing, oral hygiene instructions and frequent maintenance visits, showed a significant reduction in the percentage of sites with plaque, gingival bleeding, bleeding on probing and probing depths. However, the potential beneficial effect of periodontal treatment on the reduction of preterm birth and/or low birth weight rates was not significant [2,28,30,31]. Schwendicke concluded that periodontal therapy could reduce the risk of preterm birth and low birth weight only for the high-risk population. Yet, they could not provide firm evidence due to the bias of overestimation of benefit [32]. A Cochrane review on periodontal treatment in pregnancy and adverse pregnancy outcomes finally selected 15 relevant studies. They concluded that there was no significant difference in preterm births and only moderate association of fewer low birth weight infants after periodontal treatment (all low-quality evidence) [33]. Recent randomized controlled trials are hard to compare because of inter-trial differences and therefore possible bias caused by ineffective intervention, inappropriate timing of the intervention or irreversible damage caused by the factor before the intervention [21]. Therefore, health care professionals are still unable to adequately translate results into actual practice [21]. There is no clear proof of positive association between the formulated adverse pregnancy events and periodontal treatment, but neither is there clear proof that there is no relationship between periodontal disease and adverse pregnancy events [21].
We report a case of immature preterm prelabour rupture of membranes caused by F. necrophorum septicemia caused by periodontal disease and poor oral health. No distinct studies were found that described the relation between periodontitis caused by F. necrophorum and immature foetal demise. Infection with F. necrophorum can be very severe with septicemia and metastasis of bacteria in previously healthy young adults. The multifactorial pathology of periodontitis and the increase in severity in pregnancy emphasizes the importance of dental health in women of child bearing age. In early literature an association between periodontal disease and preterm birth, preeclampsia or low birth weight (< 2500 grams) was reported. Both periodontal disease and these adverse pregnancy outcome measures share the same multifactorial cause, such as cigarette smoking, infectious diseases, low socioeconomic status and other possible confounding factors.
The effect of periodontal treatment on the reduction of adverse pregnancy outcome is still inconclusive. Treatment of periodontal disease during pregnancy is not contraindicated. After reviewing literature, we can conclude that treatment is beneficial for the periodontal status, but conclusions of the effect on adverse pregnancy outcome still remain pending. There is a high risk of bias in literature due to lack of consensus on the definition of the periodontal disease, the interventions necessary to treat the disease and the timing of the treatment. It is possible that, concerning pregnancy, the timing of the intervention to treat or even prevent periodontal disease comes too late. It might be preferable to formulate risk groups, like women who previously delivered preterm, delivered a child of low birth weight, smoke, have low socioeconomic status, have low educational level and/or do not attend dental screening regularly. Women from high risk groups could be screened and educated early in pregnancy to increase prevention and decrease repetition of periodontal disease. Oral health can be promoted and improved by dental care takers, but there is a role for other health professionals as well. They can create awareness of the dental health status in the most vulnerable patients. In order to improve obstetric care for pregnant women in the future, a detailed description should be made of risk factors for adverse pregnancy outcome in pregnant women, such as low socioeconomic status, cigarette smoking, evidence of periodontitis, poor immunological status and complicated obstetric history. This should include the awareness of the health care worker for the dental status of the patient.
In conclusion, we have reported a rare case of severe maternal periodontal disease causing bacteremia and amniochorionitis with F. necrophorum, an anaerobic gram-negative bacterium, resulting in immature delivery and foetal demise. There might be an association between periodontal disease and adverse pregnancy outcome (preterm delivery, low birth weight and pre-eclampsia). However, there is no evidence that improving the dental status during pregnancy will decrease the risk of adverse pregnancy outcome. Future research should focus on low bias and high-quality data and the efficacy of preconceptional dental care, particularly in high risk women. This requires a health care setting where women are regularly screened for periodontal disease at childbearing age. Until then, health care workers should be aware that poor oral hygiene may be a risk factor for adverse pregnancy outcome in the conceptional period and during pregnancy and be aware of the attributions of their patients towards dental care in general, but also their attributions of dental care during pregnancy.
None.