COVID-19 pandemic has severely disrupted our lives, wrecking havoc in our economies and pushing the healthcare sector to its breaking point. This prompted world governments to come up with different measures and protocols in the hope of containing and preventing the further spread of the COVID-19 virus. Governments and pharmaceutical companies are on the race to develop an effective vaccine with the goal of preventing deaths and halt human transmissions of the virus. One of the COVID-19 vaccines that was approved by World Health Organization and Food and Drug Administration for emergency use in several countries is Sinovac-CoronaVac, which was developed by Sinovac.
Guillain-Barré Syndrome (GBS) is a relatively uncommon acute inflammatory polyneuropathy that typically present as a rapidly progressive, symmetric weakness and are flexia. There have been reports of GBS after vaccination for polio, influenza, hepatitis A and B, and rabies. The earliest reported case of GBS associated with COVID-19 vaccine was in the US in February 2021 following the administration of Pfizer-BioNTech COVID-19 vaccine, an mRNA vaccine [1].
There have been more than 260 million doses of Sinovac-CoronaVac vaccines administered worldwide as of April 2021. Adverse reaction to the vaccines have also been reported and there have been two reported cases of GBS following administration of Sinovac-CoronaVac vaccine as of this writing.
We present a 59 year-old male patient who presented in the emergency room with a 10-day history of bilateral weakness of his lower extremities which gradually progressed to involve both his upper extremities. The patient received his first dose of Sinovac-CoronaVac vaccine two days prior to onset of his symptoms which started as generalized joint pains for two days followed by weakness of both lower extremities. Patient can still stand up and walk slowly with assistance but has to lift his leg with both hands when trying to get into the car. Five days from onset of symptoms, there was total paralysis of both lower extremities and weakness of both arms such that patient can no longer hold a spoon. During this time the patient was already bed-bound. He was initially seen by an Endocrinologist who requested for several diagnostic tests. Laboratory tests came out normal including an unremarkable plain cranial CT-scan. He was eventually referred to a neurologist and was advised admission for further work up and management.
He is a known hypertensive for 20 years with good compliance to his maintenance medications, non diabetic, and non-asthmatic. A non-smoker but an occasional alcoholic beverage drinker with a very good functional status prior to this present problem and he had no history of trauma or fall. There was no recent history of cough or other respiratory symptoms, fever, headache, and loose bowel movement. There was also no weight loss, change in bowel habits, night sweats, and difficulty in breathing and swallowing.
The patient was examined, awake with stable vital signs, afebrile, not in respiratory distress with an oxygen saturation of 99% at room air. Head and neck, chest and lungs, and cardiovascular findings were all unremarkable. Mini Mental State Examination was normal with no visual nor auditory hallucinations and cranial nerves were intact. Motor strength testing based on the Medical Research Council (MRC) showed a decreased motor strength of 2/5 in both lower and upper extremities with no loss of sensation in all limbs. Deep tendon reflex test showed hyporeflexia with negative for Babinski reflex. There was no dysdiadochokinesia, dysmetria, nor nystagmus, as well as signs of meningeal irritation.
His COVID-19 RT-PCR from nasopharyngeal swab result was negative. Complete blood count showed mild leukocytosis with white blood cell count of 10.74 10^9/L with neutrophilic predominance of 64%, hemoglobin and platelet count were within normal range. Sodium was low at 125 meq/L, potassium was normal (4.7 meq/L), with a creatinine of 1.20 mg/dL and EGFR of 65.8 ml/min/1.72m2; SGPT (90.50 u/L) and HBA1C (6.9%) were elevated. Chest X-Ray AP view showed clear lung fields.
Nerve conduction exam showed the following findings: bilateral median, ulnar, radial, sural and superficial peroneal nerve sensory potentials with normal latency values, conduction velocities and potential amplitudes. Motor conduction studies showed prolong distal latencies, severely reduced amplitude with slowed conduction velocities on the right median nerve while absent on the left. The ulnar nerve compound motor unit potentials were also absent on both sides. In the lower limbs, the peroneal and tibial nerves were absent bilaterally. All median, ulnar and tibial F-waves responses were absent. The tibial H-reflexes were also absent. As seen on Figure 1 and Figure 2.
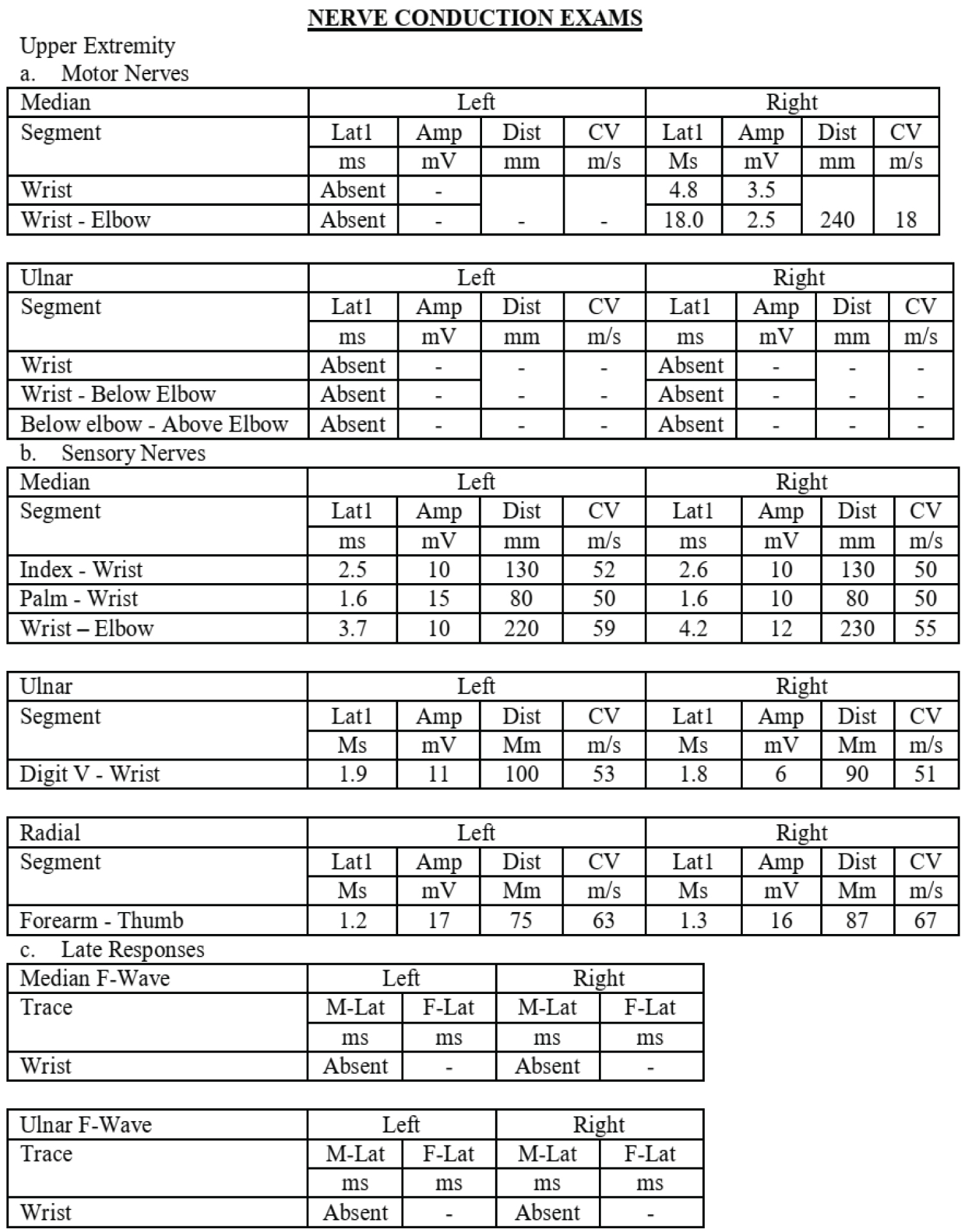 Figure 1: Nerve conduction exams.
View Figure 1
Figure 1: Nerve conduction exams.
View Figure 1
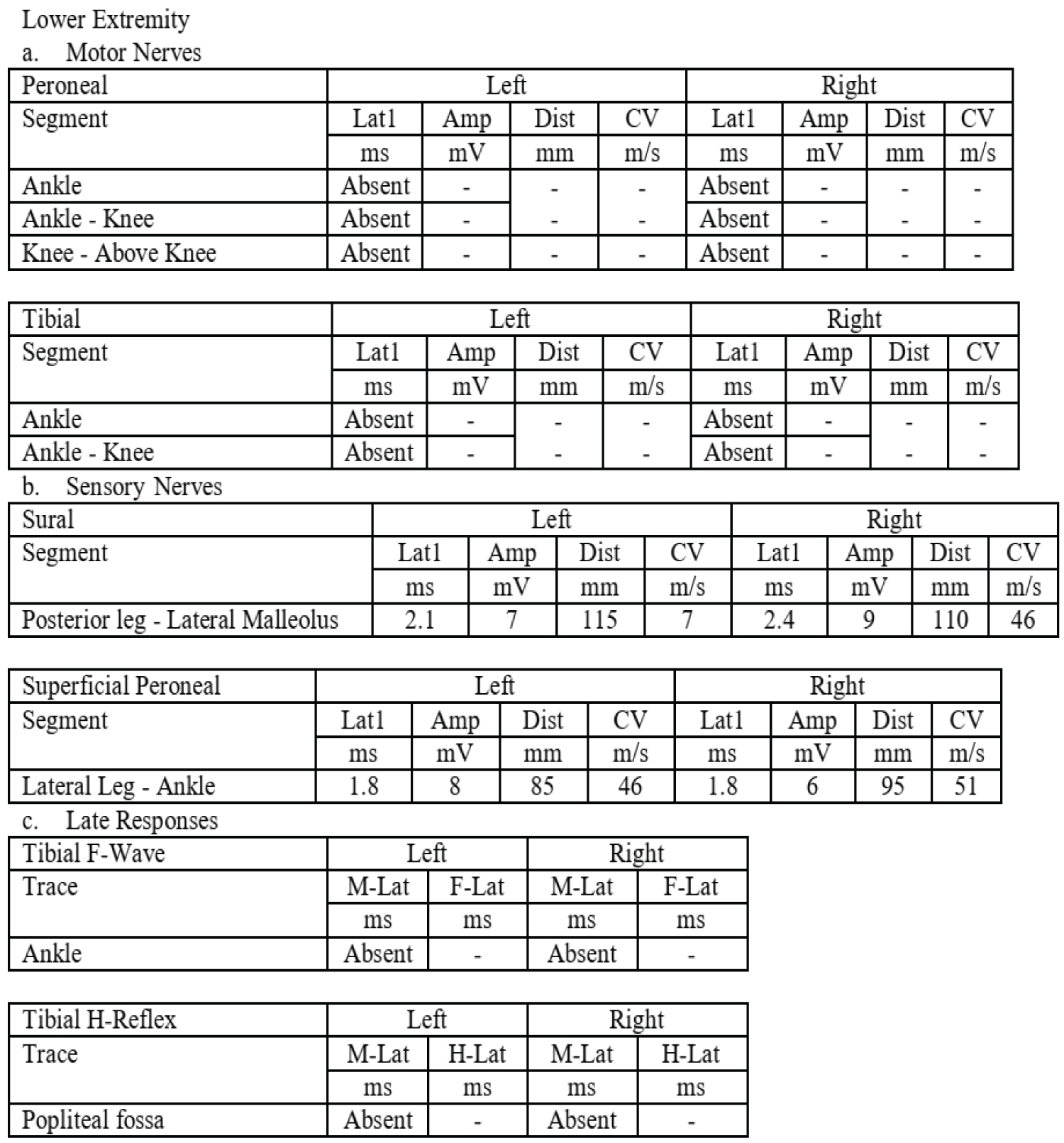 Figure 2: Nerve conduction exams.
View Figure 2
Figure 2: Nerve conduction exams.
View Figure 2
Electromyography findings showed chronic neuropathic changes characterized by large polyphasic motor unit potentials on the right triceps and vastus medialis muscles. The right deltoid, biceps brachii, tibialis anterior and medial gastrocnemius muscles also showed MUPs with prolonged duration and reduced recruitment patterns. No voluntary activity was seen on the right first dorsal interossei muscle on full activation. As seen on Figure 3.
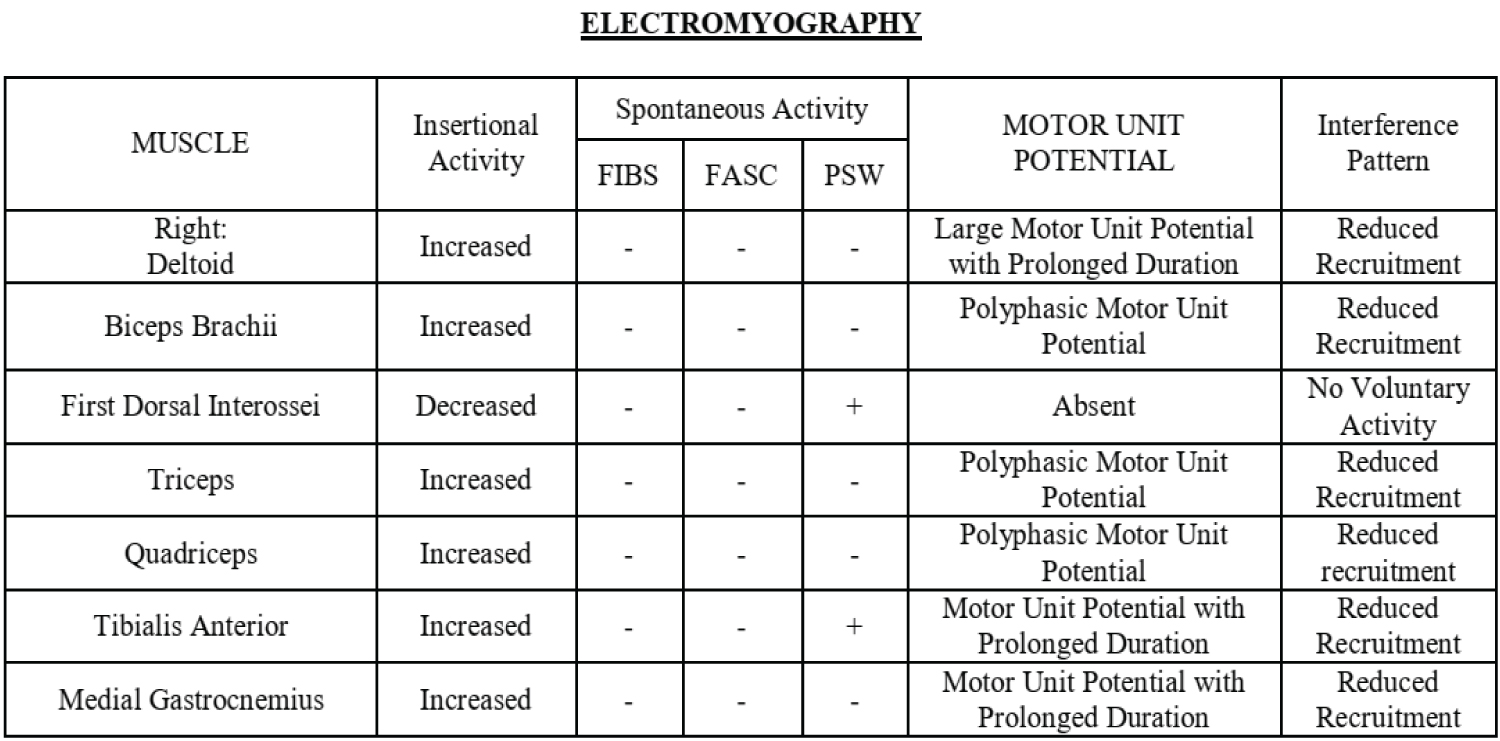 Figure 3: Electromyography.
View Figure 3
Figure 3: Electromyography.
View Figure 3
Electromyogram and Nerve Conduction Velocity (EMG-NCV) studies showed electrophysiologic evidence for a symmetric motor polyneuropathy affecting the lower limbs more than the upper limbs, with predominantly axonal involvement, consistent with Acute Motor Axonal Neuropathy (AMAN) variant. The patient was diagnosed and managed as a case of Guillain-Barré Syndrome.
The patient was given Human Intravenous Immunoglobulin (IVIG) Infusion at a dose of 0.4 gm/Kg/day every 24 hours for five doses. He showed improvement in his motor strength after the third dose of IVIG infusion, with Medical Research Council grading of 4/5 in both upper and lower extremities. Patient also verbalized improvement in his overall sense of well being when he can already move his arms and legs. The patient was discharged after 12 days of hospitalization and with Medical Research Council grading of 4/5 in both upper and lower extremities. Patient was able to stand up and walk with assistance and was able to feed himself. Patient was advised to continue his physical rehabilitation as outpatient.
One month after discharge, the patient is still undergoing regular physical therapy and still with Medical Research Council motor strength grading of 4/5 in both upper and lower extremities without progression of weakness.
According to the product information submitted by Sinovac to the World Health Organization (WHO), the Sinovac-CoronaVac is a Vero cell-based, aluminium hydroxide-adjuvanted, β-propiolactone-inactivated vaccine based on the CZ02 strain. This CZ02 strain of SARS-CoV-2 was isolated from the bronchoalveolar lavage of a hospitalized patient and is closely related to the 2019-nCoV-BetaCoV Wuhan/WIV04/2019 strain [2].
The final vaccine product contained in every 0.5 ml dose is composed of 3 μg of inactivated SARS-CoV-2 virus. The excipients are aluminium hydroxide, disodium hydrogen phosphate, sodium dihydrogen phosphate, sodium chloride, and water for injection. The vaccine does not contain preservatives. None of the excipients are of animal or human origin. The excipients are well established for use in pharmaceutical products [2].
We have been using inactivated viral vaccines for several decades already and they have been successful in preventing several diseases worldwide. Inactivated vaccines doesn't contain any replicating virus thus making it safe to use especially for immunocompromised and pregnant women. Multiple doses are usually required for inactivated vaccines and a booster dose is needed for maintenance of immunity. Inactivated vaccines contain viruses whose genetic material has been destroyed to prevent replication but inactivated vaccines can still elicit an immune response. The aluminum- based compound adjunct mixed in the CoronaVac-Sinovac vaccine can further stimulate the immune system, thus, may trigger an immune reaction by transforming sentinel cells into activated and antigen-presenting cells [3].
The inactivated COVID-19 vaccine Sinovac-CoronaVac was developed by Sinovac. Sinovac-CoronaVac has been authorized as a 2-dose vaccine (3 μ per 0.5 ml dose) for individuals aged 18 years and older. The proposed indication for emergency use listing (EUL) is a 2-dose schedule with a preferred interval of 14-28 days between doses. As of 21 April 2021, more than 260 million doses have been distributed to the public in China and other parts of the world. The vaccine is currently being evaluated in several trials and is licensed under emergency use authorizations in several countries and territories.
The most common reported local adverse reaction to the vaccine was pain at the injection site, experienced by 39.6% of vaccines after the first dose and 42.7% of vaccines after the second dose. While the most common systemic adverse events reported were headache (24.2% after dose 1 in vaccines compared with 24.6% in the placebo group), fatigue and myalgia. Of the serious adverse reactions, there were 6 cases of anaphylactic shock, 5 cases of Henoch-Schönlein purpura, 4 cases of facial paralysis, 3 cases of laryngeal edema, 3 cases of demyelination, 3 cases of cerebral haemorrhage, 2 cases of Guillain-Barré syndrome [4].
Based on the local data from Department of Health - Food and Drug Administration (FDA), the total CoronaVac - Sinovac vaccine administered between March 1 until July 25, 2021 is at 6,731,423 with 665 reports of serious events. These recent data from the FDA includes 3 reported cases of Guillane-Barré Syndrome due to COVID-19 vaccines that is currently under review after receiving Janssen and AstraZeneca COVID vaccines [5]. As of this writing there's no accurate data on the incidence of GBS post Sinovac-CoronaVac Vaccination and there's only 1 published case report of GBS post Sinovac-CoronaVac Vaccination in Istanbul, Turkey [6].
Guillain-Barré syndrome (GBS) incidence ranges from 0.8 to 1.9 per 100,000 persons/year, being more frequent among males, and the incidence increase with age. It is also the most common immune-mediated polyradiculoneuropathy and it is also the most commonly reported severe adverse event following immunization in adults [7]. GBS is an acute or subacute peripheral polyneuropathy, and is accompanied by symmetric flaccid paralysis of the extremities, sensory abnormalities, and cranial nerve palsy. In our case, the patient presented with symmetric flaccid paralysis starting on both lower extremities which later on progressed to involve both arms, but without sensory or cranial nerve involvement. Interest in the risk of GBS after vaccination increased after approximately 500 cases of GBS were reported after the mass administration of the A/New Jersey/76 vaccine during the swine flu epidemic in the United States in 1976 [8].
Although the pathogenesis of the GBS has not been clearly elucidated, recent immunological evidence has supported a mechanism of autoimmune damage following an infection of the gastrointestinal or respiratory tract in 2/3 of cases. Diagnosis for GBS aside from the motor signs and symptoms includes lumbar puncture with cerebrospinal fluid analysis. Patients with GBS will often reveal an increased CSF protein with a normal CSF white blood cell count. The albuminocytologic dissociation finding is present in 50 to 66 percent of patients with GBS in the first week after the onset of symptoms and ≥ 75 percent of patients in the third week [9]. CSF is examined mainly to exclude disorders that are associated with pleocytosis, instead of seeking confirmation of the diagnosis Guillain-Barré syndrome by demonstrating an increased protein concentration [10].
The hallmark for the diagnosis of GBS is still the clinical features of the disease which is the progressive, fairly symmetric muscle weakness accompanied by absent or depressed deep tendon reflexes, and are already present in our patient, thus the need for CSF examination was no longer indicated. Even without the CSF examination our patient already fulfilled the Level 2 diagnostic certainty based on Brighton Criteria for GBS as seen in Table 1.
Table 1: Brighton criteria for GBS [10]. View Table 1
Nerve conduction studies (NCS) and needle electromyography (EMG) are used to confirm the diagnosis of GBS and to provide information about the prognosis of the patient. Electrodiagnostic studies are also used to help determine the main variants of GBS as demyelinating (eg, acute inflammatory demyelinating polyneuropathy) or axonal (eg, acute motor axonal neuropathy) [11]. Nerve conduction abnormalities in GBS usually progress over time, thus, serial electrodiagnostic studies are frequently useful. Normal findings can occur especially early in the course of the disease, and significant abnormal findings are seen approximately two weeks after the onset of weakness. Our patient was already on his 12th day from onset of symptoms when he had his EMG-NCV studies which showed findings consistent with Acute Motor Axonal Neuropathy (AMAN) variant of GBS.
The first line treatment that has been found to be effective in patients with Guillain–Barré syndrome is plasma exchange [12]. Patients who were unable to walk who were started with plasma exchange within the first 2 weeks since the onset of disease showed better response to the treatment. Complement and antibodies were nonspecifically removed in plasma exchange helping to minimize nerve damage and hasten the patient's improvement compared to providing supportive therapy alone.
Our patient was given standard doses of IV immunoglobulin for 5 days and according to studies13it is just as effective when compared to plasma exchange among patients who present with inability to ambulate. IVIG is thought to act by neutralizing the circulating pathogenic antibodies and inhibits the autoantibody mediated complement activation. This would lead to decreased nerve damage and hasten the improvement of the patient. IVIG can be administered within 2-4 weeks from the onset of the disease, but, in a Cochrane 2014 review [13] on IV immunoglobulin therapy for GBS, IVIG showed similar efficacy to plasma exchange if given within the first 2 weeks from the onset of symptoms. In the study by Bril, et al. [14] IVIG has a median time of 14 days to see an improvement of one GBS disability scale compared to plasma exchange which is 16.5 days. Our patient received IVIG on the 13th day from onset of symptoms, with improvement in motor strength initially seen after the 3rd dose of Intravenous Immunoglobulin.
The other advantages of IVIG compared to plasma exchange is that it is more convenient and readily available, making it the treatment of choice in most medical centers. Furthermore, the combination of plasma exchange together with IVIG has no additional benefit when compared to IVIG or plasma exchange alone.
Most patients tend to recover well after the acute phase of GBS with at least 80% of the patients being able to ambulate independently after 6 months. There are however less than 20% of patients who still have a significant disability even after they have received the standard treatment for GBS. Patients who have recovered from GBS may still experience residual fatigue, paresthesias, and/or pain for even up to several years after treatment. Relapse after treatment is not very common at around 2%-5% of cases [15].
Severe complications associated with Guillain-Barré syndrome includes respiratory compromise that may require ventilatory support, bulbar palsies, and. arrhythmia. Severe hypotension or hypertension as a result of fatal autonomic dysfunction may also develop in at least 20% of patients with Guillain-Barré syndrome. A temporary pacemaker may also be needed for patients who develop marked bradycardia [16,17]. Fortunately for our patient, he did not have any of these complications and he responded well to IVIG treatment.
We presented a case of a 59-year-old male who exhibited progressive symmetric ascending paralysis two days after receiving his first dose of Sinovac-CoronaVac vaccine against COVID-19 and was diagnosed with Guillain–Barré Syndrome based on EMG-NCV studies. The patient was admitted in our institution where he received IV Immunoglobulin treatment. The patient showed favorable response to the treatment with dramatic improvement in his motor strength after the third dose of IVIG and was subsequently discharged without any serious sequelae.
Although there have been several reported incidence of GBS following COVID vaccination, there is no clear link nor causal relationship yet that has been fully established between any COVID 19 vaccine and GBS. Thus, the WHO Global Advisory Committee on Vaccine Safety (GACVS) concluded, particularly on the case of Janssen and AstraZeneca COVID-19 vaccine, the vaccines which were reported to have more cases of GBS post vaccination, that the potential benefits of COVID vaccines outweigh any potential risk of developing GBS [18]. This cannot be more emphasized particularly in the threat of the more transmissible Delta (B.1.617.2) variant. Nevertheless, it is still important to have a high index of suspicion and to report any potential side effects or complications that may be attributable to the vaccine. Also prompt recognition of GBS as a possible complication is imperative so that timely and appropriate management be instituted to avoid possible serious sequelae of this potentially life-threatening condition.