PET imaging, Dopaminergic system, Parkinson's disease
It is well known that the dopaminergic system plays a major role in Parkinson's disease (PD). Current technological developments in molecular imaging have made it possible to evaluate the dopaminergic system in vivo, including alterations of dopamine synthesis, integrity of dopaminergic terminals, and receptor densities, invaluable information for understanding the mechanisms underlying the pathogenesis of PD and to evaluate efficacy of treatments [1]. Positron Emission Tomography (PET) provides these features, provided the availability of suitable tracers to investigate presynaptic and postsynaptic function [2,3]. Here we present two case reports to show the utility of the combined use of PET radiopharmaceuticals to evaluate PD and the efficacy of treatments to restore dopaminergic transmission.
A 56-year-old right handed Hispanic female with a 10-year history of PD under L-dopa treatment, with a Unified Parkinson's Disease Rating Scale motor score (UPDRSms) of 43 and 24 under "OFF" and "ON" medication conditions, respectively. History of diabetes treated with metformin. She denies any family history of PD or any other neurodegenerative problems and does not drink alcohol orsmokes cigarettes. Patient underwent intraputaminal transplantation of human fetal-derived stem cells (hfSC). Figure 1 shows PET images (fused with MRI) in the baseline state (top row) and at one year after stem cell transplantation (middle row), obtained with two radiopharmaceuticals. For comparison purposes a healthy control (HC) subject is included in the bottom row. Radiopharmaceuticals utilized were (+)-alpha-[11C]Dihydrotetrabenazine (DTBZ) and [11C]Raclopride (RAC). DTBZ is a presynaptic radioligand with a high affinity to type-2 vesicular monoamine transporter (VMAT2) and is used as an in vivo marker of nigrostriatal dopaminergic system integrity. PD patients, compared to HCs, show a reduced uptake of DTBZ in the striatum, which is more prominent in the putamen [4,5]. RAC, on the other hand, is a postsynaptic radioligand. It is a dopamine D2 receptor antagonist and previous studies have shown that RAC binding to the postsynaptic D2 receptors is either normal or increased in PD patients, and the increase has been interpreted as a compensatory reaction to the reduction of striatal dopaminergic terminals [6]. Note the mismatch pattern of DTBZ and RAC uptake in the putamen of PD patients. Note also the increased uptake of DTBZ and decreased uptake of RAC at one year follow up (1YFU) compared with baseline studies, suggesting improvement of the disease after stem cell implantation.
A 60-year-old right handed white male, with a 12- or 13-year history of PD under L-dopa treatment, with a UPDRSms of 28 and 11 under "OFF" and "ON" medication conditions, respectively. His past medical history is significant for irritable bowel syndrome. Patient denies any allergiesand alcohol use. He used to smoke one cigarette per day. Patient underwent intraputaminal transplantation of hfSC. Figure 2 shows PET images (fused with MRI) in the basaline state (top row) and at one year after stem cell transplantation (middle row), obtained with two radiopharmaceuticals. For comparison purposes a HC subject is included in the bottom row. Radiopharmaceuticals utilized were DTBZ and 6-[18F]Fluoro-L-DOPA (FDOPA). FDOPA evaluates the first step in dopaminergic transmission, measuring the dopamine synthesis through the expression of aromatic amino-acid decarboxylase (AADC) in presynaptic neurons. It has been demonstrated that PD patients, compared to HC, show a significant reduction of striatal uptake, which is more severe in the putamen that in the caudate nucleus, and more prominent in the posterior putamen [7,8]. Note the match pattern of DTBZ and FDOPA uptake in the putamen of PD patients. In this case the uptake of both tracers before and after stem cell implantation is very similar, suggesting stable disease.
Research supported in part by Celavie Biosciences and UNAM-DGAPA-PAPIIT IT201115.
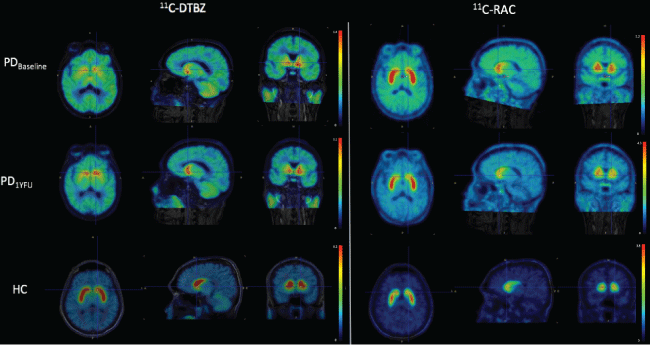
Figure 1: Axial, sagittal and coronal planes of PET/MRI images of a PD patient evaluated with DTBZ and RAC before and after stem cell implantation. A healthy control subject is included at the bottom row for comparison purposes.
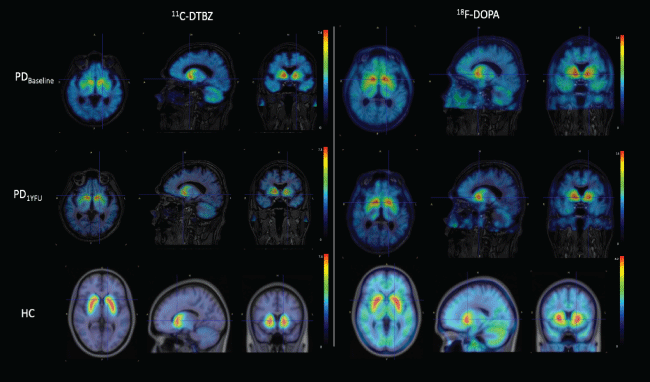
Figure 2: Axial, sagittal and coronal planes of PET/MRI images of a PD patient evaluated with DTBZ and FDOPA before and after stem cell implantation. A healthy control subject is included at the bottom row for comparison purposes.