COVID-19 virus does not directly affect the heart. The virus can cause systematic inflammation that can lead to severe cardiac complications like uncontrolled atrial fibrillation (AF), which carries high mortality among patients. We present a case of an 82-year-old female from nursing home with advanced dementia and multiple comorbidities. She initially screened negative for COVID-19 in the nursing home, however her repeat test was positive. Soon after admission she developed progressive, hypoxic respiratory failure and was subsequently diagnosed with ARDS and required endotracheal intubation. The patient was found to have uncontrolled AF on telemetry monitoring. Initially, intravenous calcium channel blockers were unsuccessful in controlling her heart rate, thus this was later changed to intravenous amiodarone. The patient was successfully extubated on day 15 and transferred to the general medical floor on antiarrhythmic medication for AF.
Covid-19, Atrial fibrillation, Arrhythmia
The World Health Organization describes Coronavirus (COVID-19) disease primarily as a respiratory illness [1]. Patient's presentation can range from asymptomatic to severe symptoms such as pneumonia, acute respiratory distress symptoms and organ failure. However, the COVID-19 virus itself is known to cause viral myocarditis in rare instances, although it usually does not directly affect the cardiac tissue itself [2]. Monitoring of patients for cardiac abnormalities is essential due to the systematic illness caused by COVID-19 infection which leads to cytokine storms resulting in cardiac arrest and arrhythmias [3]. Any COVID-19 patients who develop cardiac abnormalities such as atrial fibrillation (AF) may carry an unfavorable prognosis, especially in patients with previous comorbidities or cardiac abnormalities [3]. Thus, it is imperative that AF in patients with COVID-19 infection is managed appropriately. We describe a case in which AF is uncontrolled by rate-controlled medications.
An 82-year-old female with a significant past medical history of advanced dementia, diabetes mellitus type 2 and chronic kidney disease was referred to a nursing home for dysphagia and failure to thrive. On physical exam, bilateral crackles were noted on lung exam, and the remainder of the physical exam was unremarkable. Blood pressure on admission was 110/85 mmHg, heart rate 80 bpm, and oxygen saturation were 95% on 2 liters of oxygen via nasal cannula. Her COVID-19 nasal PCR test was negative at her nursing home, but it was positive on hospital admission. Chest X-ray showed bilateral infiltrates (Figure 1). She was also found to have a urinary tract infection (UTI) and pneumonia on admission and was started on intravenous (IV) fluids and antibiotics.
 Figure 1: CXR showed infiltrate on right side.
View Figure 1
Figure 1: CXR showed infiltrate on right side.
View Figure 1
Her pneumonia was initially treated with vancomycin due to a MRSA positive MRSA screen. Her UTI treated with ceftriaxone. She was intubated due to worsening hypoxic respiratory failure and acute respiratory distress syndrome (ARDS). Brief episodes of paroxysmal AF versus supraventricular tachycardia were found on telemetry monitoring. Initial treatment with IV metoprolol was started. Atrial fibrillation with rapid ventricular response developed that was resistant to beta-blockade and a diltiazem drip was initiated which initially improved her heart rates. She remained in atrial fibrillation with heart rates ranging from the 60s to 140s on diltiazem. Amiodarone infusion was then started for rate and rhythm control. Diltiazem was held, and metoprolol was increased from 5 mg IV every 6 hours to 7.5 mg IV q6hrs. IV Amiodarone was stopped after achieving sinus rhythm, and IV metoprolol was increased to 10 mg every 6 hours. The patient remained in sinus rhythm on metoprolol 10 mg IV every 6 hours. Unfortunately, the patient went to asystole cardiac arrest. Return of spontaneous circulation was achieved after three rounds of cardiopulmonary resuscitation with 1dose of epinephrine. Norepinephrine was started for vasopressor support. The patient then had a resurgence of AF with rapid ventricular response requiring amiodarone infusion to be restarted. Sinus rhythm returned. Amiodarone infusion was then switched to an oral formulation. The patient was extubated on day 15 and transferred out of the intensive care unit and eventually to the general medical floor on telemetry monitoring (Figure 2).
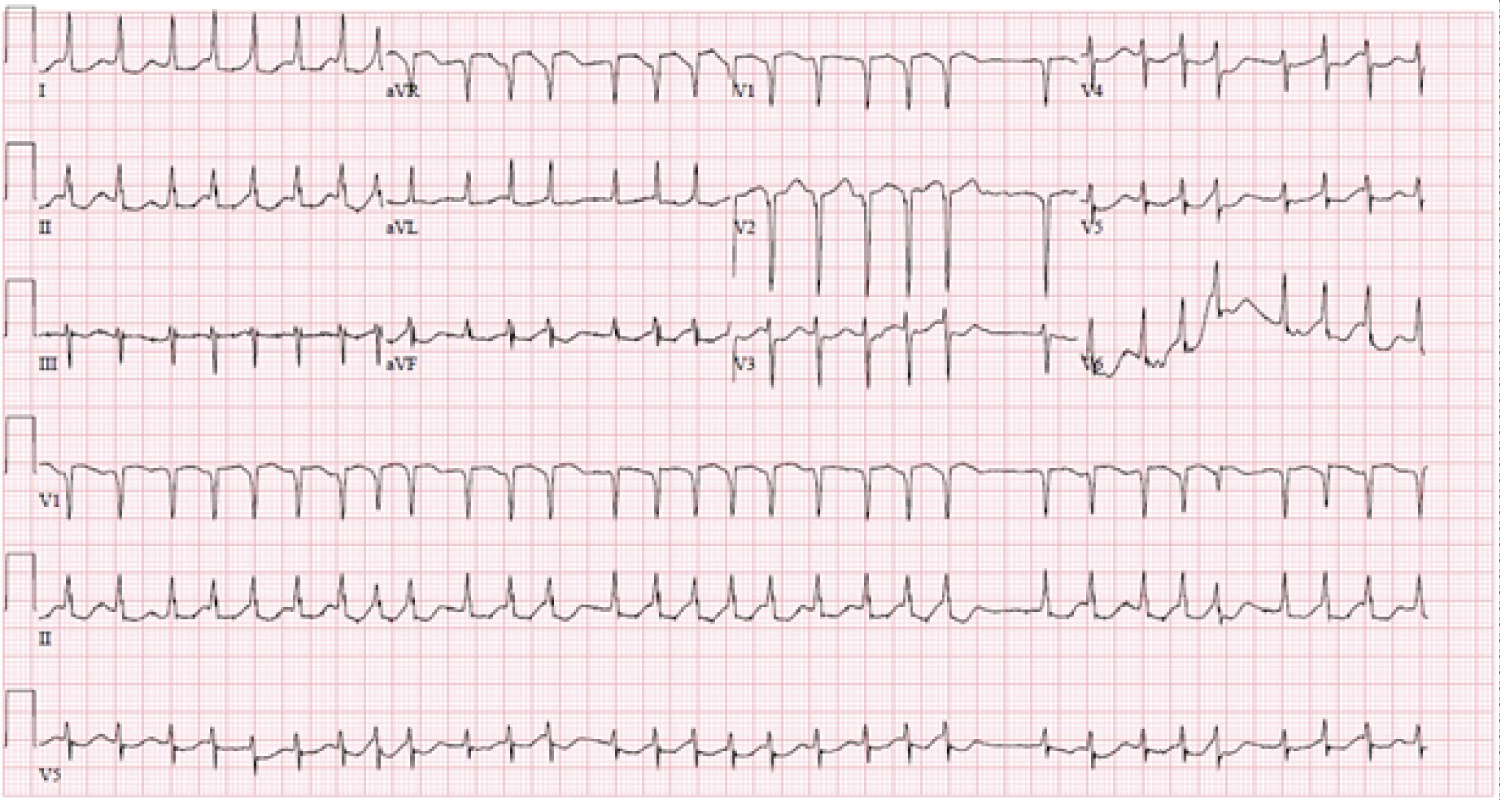 Figure 2: EKG showed atrial fibrillation with RVR and having septal infraction.
View Figure 2
Figure 2: EKG showed atrial fibrillation with RVR and having septal infraction.
View Figure 2
Our patient presented with severe ARDS from COVID-19 which was complicated by AF with rapid ventricular response that was resistant to rate-controlled medications. Antiarrhythmic drug therapy was required for rate control and later for rhythm control. Any COVID-19 patients with multiple comorbidities are at risk for cardiac arrhythmias. Although the COVID-19 virus itself does not affect the cardiac muscle directly, it can lead to an unstable hemodynamic conditions, hypercoagulable states, and cytokine storms, that can significantly increase the risk of developing AF [4]. COVID-19 disease leads to a significant influx of CD4+ T cells into cardiac tissues due to increased inflammatory marker production. This can cause damage to myocardial cells and cause tissue inflammation and fibrosis leading to atrial fibrillation in COVID-19 patients [4]. Any cardiac injury in COVID-19 patients is a significant risk factor in worsening outcomes. Early recognition with rate and rhythm control treatment can significantly improve the patient's overall prognosis.
It is important to minimize potential drug interactions among other medications given to control other disease manifestations caused by a COVID-19 to prevent the worsening of cytokine storms and the subsequent inflammatory response (Table 1) [5]. It would be beneficial to have a patient transferred to the Intensive Care Unit to provide frequent cardiac monitoring and provide necessary rate or rhythm controlling drugs such as beta-blocker or amiodarone when needed.
Table 1: Drug interaction between COVID-19 therapy and rate and rhythm controlling agents. View Table 1
In our case, the patient's heart rate was controlled on amiodarone. A possible hypothesis to this phenomenon is that the COVID-19 patients have high systemic inflammation. This high systemic inflammation or cytokines affect the cardiac muscle. Antiarrhythmic medications work best in systemic inflammation compared to rate-control medications in these patients. One of the sides of amiodarone is pulmonary fibrosis which can worsen the patient's lung inflammation and cause significant pulmonary fibrosis. It would be more beneficial to use dofetilide or other antiarrhythmic medications to control AF with rapid ventricular response in patients with COVID-19 infection.
Our case demonstrates the importance of antiarrhythmic medication in a patient with COVID-19 and uncontrolled AF.
Informed written consent was taken from the patient for case report writing.
There is no conflict of interest between the authors.
Nil.