Introduction: Bacteria from the vagina are transported into the uterus during labor. We evaluated if the vaginal microbiome in the third trimester of pregnancy was related to the length of the second stage of labor.
Patients and methods: The composition of the vaginal microbiome in 73 women in their third trimester was measured by analysis of the V1-V3 region of the gene coding for bacterial 16S ribosomal RNA. The vaginal concentration of D- and L-lactic acid was determined by ELISA. The time from full dilation to delivery was obtained by chart review.
Results: The length of the second stage of labor was longer when Lactobacillus crispatus was dominant in the third trimester as compared to when L. iners (p = 0.0089) or Gardnerella vaginalis (p = 0.0152) predominated. The vaginal concentration of D-lactic acid, but not L-lactic acid, was also directly related to the time from full cervical dilation to delivery (p = 0.0313).
Conclusion: The composition of the vaginal microbiome in the third trimester is associated with the length of the second stage of labor.
D-lactic acid, Lactobacillus crispatus, Second stage of labor, Vaginal microbiome
The interval from complete cervical dilation to birth is known as the second stage of labor. Oxytocin stimulation of myometrial contractions reaches its maximum intensity [1]. The time of this stage typically is < 3 hours in nulliparous women and < 2 hours in parous women [2,3]. However, little is known about the variables that influence the duration of the second stage of labor.
The vaginal microbiome differs between pregnant and non-pregnant women, with lactobacilli becoming more prominent and bacterial composition remaining stable through all three trimesters [4-7]. One of four different lactobacillus species predominate: Lactobacillus crispatus, L. iners, L. jensenii and L. gasseri. In a minority of women other anaerobic and facultative bacteria, most frequently Gardnerella vaginalis, are predominant [6]. Among the vaginal bacteria only L. crispatus, L. jensenii and L. gasseri produce both the D- and L-isomers of lactic acid. L. iners produces only the L-isomer [8]. Differences in biological properties between the two lactic acid isomers have been noted [8-10].
During labor bacteria from the vagina are sucked into the uterine cavity as a consequence of contraction activity [11,12]. In the present study we evaluated whether differences in vaginal microbiome composition during the third trimester influenced length of the second stage of labor.
Subjects were 73 women in their third trimester of pregnancy seen in the outpatient obstetrics clinic at Weill Cornell Medicine in which detailed information about time from full dilation to delivery was available in their charts. All were White, had uneventful singleton pregnancies and vaginally delivered a healthy baby at term. Exclusion criteria were multiple gestations, premature rupture of membranes, delivery before 37 completed weeks of gestation, signs or symptoms of infection, current smoker, pregnancy complications such as hypertension, preeclampsia, gestational diabetes or any other autoimmune diagnosis or inability to provide informed written consent.
Composition of the vaginal microbiome was determined by amplification of the V1-V3 region of the gene coding for bacterial 16S ribosomal RNA, as described previously [13]. Dominance was defined as a bacterial species present at > 50% of the total number of identified bacteria.
The concentration of the D- and L- isomers of lactic acid in vaginal fluid were determined by ELISA, as previously described [8].
Differences in the length of time of the second stage of labor when different bacteria were dominant were analyzed by the non-parametric Mann-Whitney test. The association between vaginal lactic acid levels and time of the second stage was analyzed by the Spearman rank correlation test. A p value < 0.05 was significant.
The mean (SD) subjects' age was 33.7 (3.6) years, body mass index was 22.0 (2.8) kg/m2, gravidity was 2.2 (1.2), gestational age at delivery was 39.5 (1.4) weeks and baby's birth weight was 3299 (495) grams.
L. crispatus was the dominant bacterium in 35 women, followed by L. iners in 12 women, L. jensenii and L. gasseri in 8 women each, G. vaginalis in 7 women and other anaerobic bacteria in 3 women. The association between dominant vaginal bacteria and time from full dilation to delivery for each individual woman is shown in Figure 1. The median (25%-75%) time was 50.0 (23.0-72.0) minutes for L. crispatus, 34.5 (16.8-100.5) for L. jensenii, 22.5 (10.0-70.8) for L. gasseri, 24.0 (11.0-34.5) for L. iners, 18.0 (16.0-31.0) for G. vaginalis and 13.0 (5.0-47.0) for other anaerobes. Differences between L. crispatus and L. iners (p = 0.0089) and G. vaginalis (p = 0.0152) were significant. Similarly, the time difference between when the three D-lactic acid-producing lactobacilli (42.0 minutes) were dominant and when other bacteria dominated (20.0 minutes) was significant (p = 0.0030). The percentage of women having their first pregnancy in which D-lactic acid-producing bacteria were dominant (38.5%) was similar to the percentage when other bacteria predominated (29.2%).
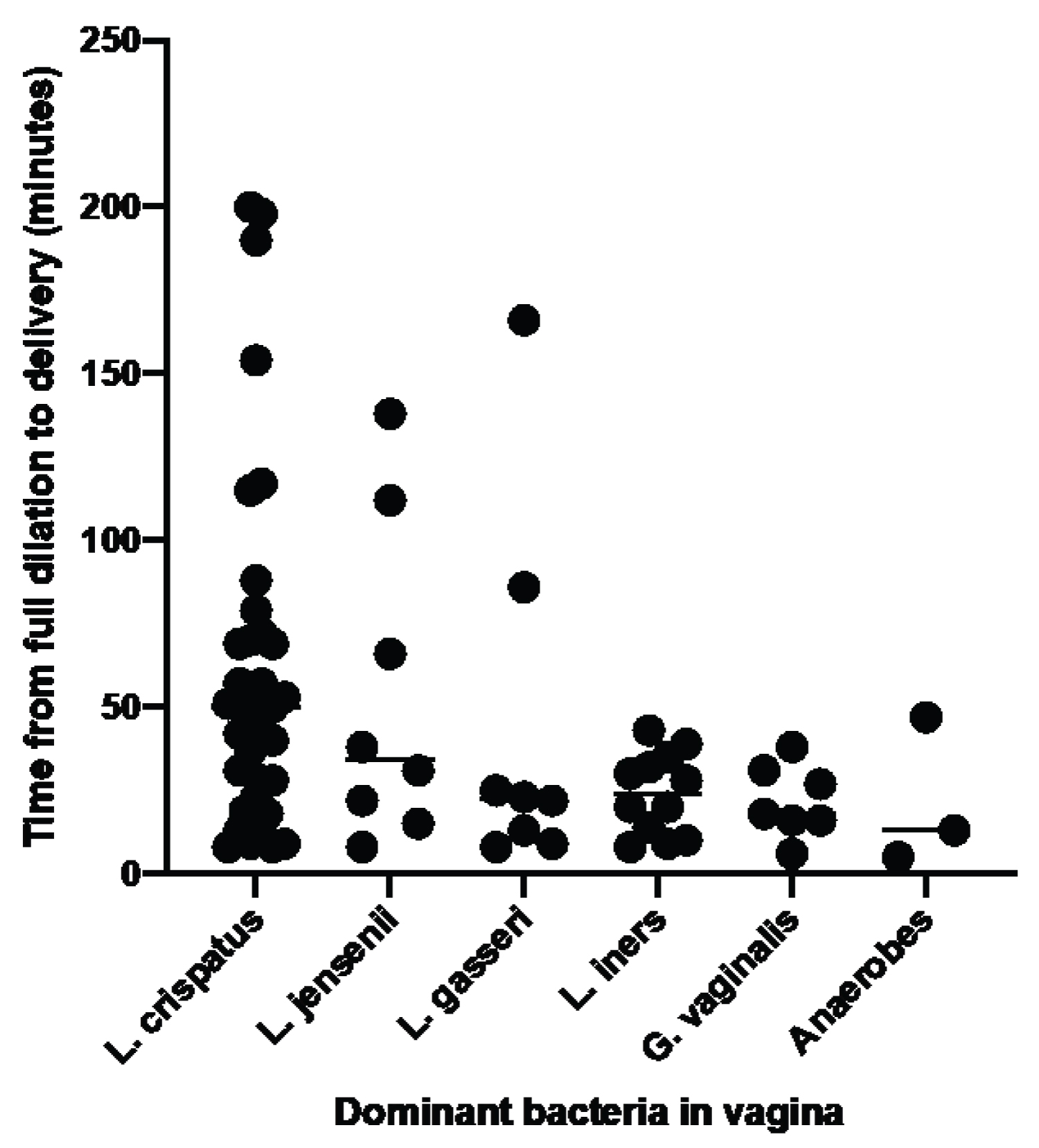 Figure 1: Association between dominant vaginal bacteria in the third trimester and time from cervical dilation to delivery. The dominant bacteria in the vagina was determined by amplification and analysis of the V1-V3 region of the gene coding for bacterial 16S ribosomal RNA. The time from full cervical dilation to delivery was obtained from patients' charts. The three anaerobes were Alloscardovia, Atopobium and Streptococcus. View Figure 1
Figure 1: Association between dominant vaginal bacteria in the third trimester and time from cervical dilation to delivery. The dominant bacteria in the vagina was determined by amplification and analysis of the V1-V3 region of the gene coding for bacterial 16S ribosomal RNA. The time from full cervical dilation to delivery was obtained from patients' charts. The three anaerobes were Alloscardovia, Atopobium and Streptococcus. View Figure 1
As an independent validation of an association between D-lactic acid-producing bacteria and length of the second phase of labor we also measured vaginal levels of D- and L-lactic acid in all subjects. There was a positive association between the D-lactic acid concentration and time to delivery (p = 0.0313), while there was no such association with the L-lactic acid level (p = 0.3354).
In our study population the time distribution from full cervical dilation to delivery was longer when D-lactic acid-producing bacteria, and especially L. crispatus, was numerically dominant in the vagina during the third trimester as compared to when other bacteria predominated.
While the involved mechanism(s) remains to be determined, biologically plausible explanations for this observation are available. As mentioned in Introduction, bacteria present in the vagina are transported to the uterine cavity during labor as a consequence of the creation of a suction-like effect. Therefore, the predominance of different bacteria in the upper genital tract may alter the intensity and/or duration of myometrial contractions. The contractions are stimulated by an increase in uterine pro-inflammatory immune activity plus the release of oxytocin. The extent of uterine inflammation has been shown to be regulated by lactic acid [14,15]. The mechanism may include lactic acid binding to a G protein-coupled receptor (GPR81) [15], as well as by other lactic acid-derived immunoregulatory activities [16]. The rate of oxytocin secretion is also influenced by the local lactic acid concentration [14]. Furthermore, there is evidence that lactobacillus lysates regulated oxytocin release in vitro [17]. The rate of metabolism of L-lactic acid has been shown to greatly exceeds that of D-lactic acid [18]. If this is also the case in the pregnant uterus, then the D-lactate isomer would selectively remain intact and be capable of exerting biological activities for a prolonged period of time.
Our study has several limitations. The small number of women analyzed necessitates that the investigation is designated as exploratory. Further evaluation of larger numbers of women is needed to confirm these initial observations. In addition, we did not determine if vaginal bacteria present in the third trimester corresponded to the bacterial composition in the uterine cavity during labor or to levels of immune mediators or oxytocin. Nevertheless, the findings suggest the existence of a novel mechanism influencing the duration of the second stage of labor that is readily testable and, if validated, could lead to protocols to modulate this time period.