Respiratory-associated pneumonia refers to pneumonia in which the patient needs help breathing by a machine through a tube inserted in the windpipe. Symptoms of respiratory infections, such as pneumonia, are not limited to infection alone, but also cause respiratory failure in critical care settings. The concept of bronchoalveolar lavage (MinibAl) was developed to diagnose Ventilator-associated pneumonia so that appropriate antibiotics can be prescribed.
Methods: Fifty-one MinibAl samples were collected from cases admitted in the Intensive Care unit at Hospitals, Misrata, Libya. Microbial identification was done by using biochemical following the common standard methods. The susceptibility test was performed by disk diffusion method as stated by Clinical and Laboratory Standards Institute.
The results: The patients were 55% men and 45% women, and their ages ranged from 12-99 years. Microbial growth was detected in 26 samples. Microbial isolates were Klebsiella spp (21%), Candida albicans (14%), Acintobacter spp (8%), Streptococcus spp (6%), Pseudomonas aeruginosa (8%), E. coli (6%), and Staphylococcus aureus (2%). According to research results, almost all bacteria were susceptible to common antibiotics except Klebsiella spp. and Acinetobacter spp. isolates were resistant to most of the antibiotics. Ventilator-associated pneumonia are common cases in Intensive Care units, and the majority of them are associated with multi-drug resistant strains. Suggesting that the critical care department in a hospital performs a proper diagnosis of pneumonia to determine the appropriate antibiotics for the treatment of patients.
Microbial isolates, Ventilator-associated pneumonia, Antibiotics
VAP: Ventilator-Associated Pneumonia; IMP: Imipenem; CRO: Ceftriaxone; CN: Gentamicin; AMC: Amoxicillin/Clavulanic Acid; CXM: Cefuroxime; CTX: Cefotaxime; CIP: Ciprofloxacin; AK: Amikacin; MRP: Meropenem; CST: Colistin
Respiratory-associated pneumonia refers to pneumonia in which the patient needs help breathing by a machine through a tube inserted in the windpipe, a face mask, or other methods within 24 hours of the onset of the infection [1]. Infection in hospitals usually occurs in the intensive care unit and is the main reason for staying in hospitals and increasing the number of deaths [2]. Respiratory-associated pneumonia is also considered the most common type, followed by urinary tract infection and bloodstream infections [3].
Respiratory infections such as pneumonia are not limited to infection only, but can also affect the patient's general health by causing respiratory failure resulting in hospital critical care requirements [4]. Respiratory-associated pneumonia continues to be one of the most common hospital-acquired infections and is associated with a high mortality rate. Healthcare providers should consider the changes or clinical features such as a decrease in oxygen saturation, increased body temperature, change in sputum characteristics, and change in chest radiographic features as markers of ventilator-associated pneumonia (VAP). Initial diagnosis is processed by evaluating patient clinical features and then is confirmed by laboratory investigations including sample collection from the respiratory tract for microbial detection to support of the prescription suitable anti-microbial agents [5].
Bronchoalveolar lavage (Minibal) sample is a technology that has been developed to diagnose VAP to identify appropriate antibiotics for patient treatments. Based on the Centers for Disease Control and prevention recommendations, pneumonia should be identified by performing semi- quantitative or non-quantitative sputum cultures obtained by deep coughing, induction, aspiration or bronchoalveolar washing [6]. The procedure for obtaining appropriate sputum samples from the airways is essential. Commensal cantonments can make it difficult to identify the causative organism, resulting in incorrect antibiotic prescriptions. It was suggested that the usage of fibrous bronchoscopy for sample collection is complex and needs expertise. In this case, a MinibAl sample will be suggested since it is not difficult or time-consuming [7]. Approximately 50-80% of patients with pneumonia die as a result of respiratory failure requiring mechanical ventilation. The mortality rate can be dramatically reduced when antibiotics are prescribed early and accurately [8].
In recent years, miniBAL samples have become increasingly common, especially for diagnosing VAP. MiniBAL was used for the first time successfully in 1989 to diagnose hospital-acquired pneumonia [9,10]. The global prevalence of healthcare-associated infection was estimated between 7-12% according to the World Health Organization. Patients admitted to the intensive care unit are at risk of contracting nosocomial infections by multidrug resistance [11].
Microbial resistance to antibiotics is a global public health threat with a high mortality rate that will continue to rise in the future if there is no solution to this problem [12]. Management of severe acute respiratory syndrome requires a combination of antibiotics, nutritional balance, respirator support [13,14]. Many studies were conducted for bacterial association with respiratory infection. A study in Indonesia 2018 found that most of the isolated microbes were Pseudomonas aeruginosa and fungi [15], while in a study in Egypt (2021) stated that the most of the isolates were Acinetobacter spp and Klebsiella spp . Most these isolates were multi-drug resistant to antibiotics [16]. In a study was done in India (2022) found the most of the isolates were Pseudomonas aeruginosa [17].
As there is no information available about VAP in our region, the current study aimed to identify the most common bacterial species present in miniBAL samples and their antibiotic susceptibility.
The study focused on patients who were admitted in an intensive care unit and had VAP in Misrata, Libya, from 1 of January, 2022 to 31 of August, 2022, which included 51 patients with, different sexes and ages.
MiniBAL samples were inoculated onto blood, MacConkey and chocolate agars culture media. The bacteria were identified by Gram stain and morphology on the agar and biochemical tests [16].
Antimicrobial susceptibility testing was carried out using the following method Bauer-Kirby Disk Diffusion. A bacterial suspension was prepared and adjusted at 0.5 McFarland's. Mueller Hinton agar plate was inoculated and the antibiotics (Table 1) were incubated aerobically at 37 °C for 24 hours [18].
Table 1: Antibiotics used (oxoid). View Table 1
The patients were 45% women and 55% men. The age groups were between 12 to 99-years-old. Most of the patients in the age group 56-75 years (18 cases) and 76-100 years (16 cases) as can be seen in Table 2.
Table 2: Age groups of patients. View Table 2
Microbial growth was detected in 65% (33/51) of collected samples. The majority of microbial isolates (78.9%, 26/33) were bacteria; while the other isolates were belonging to Candida spp. (21.2%). Six bacterial genera were detected and the most common was Klebsiella spp . (42.3%, 11/26), followed by Acinetobacter spp (15.4%, 4/26) and Pseudomonas aeruginosa (15.4%, 4/26), and both Streptococcus spp and E. coli were (11.5%, 3/26), and Staphylococcus aureus (3.9%, 1/26) as shown in Figure 1.
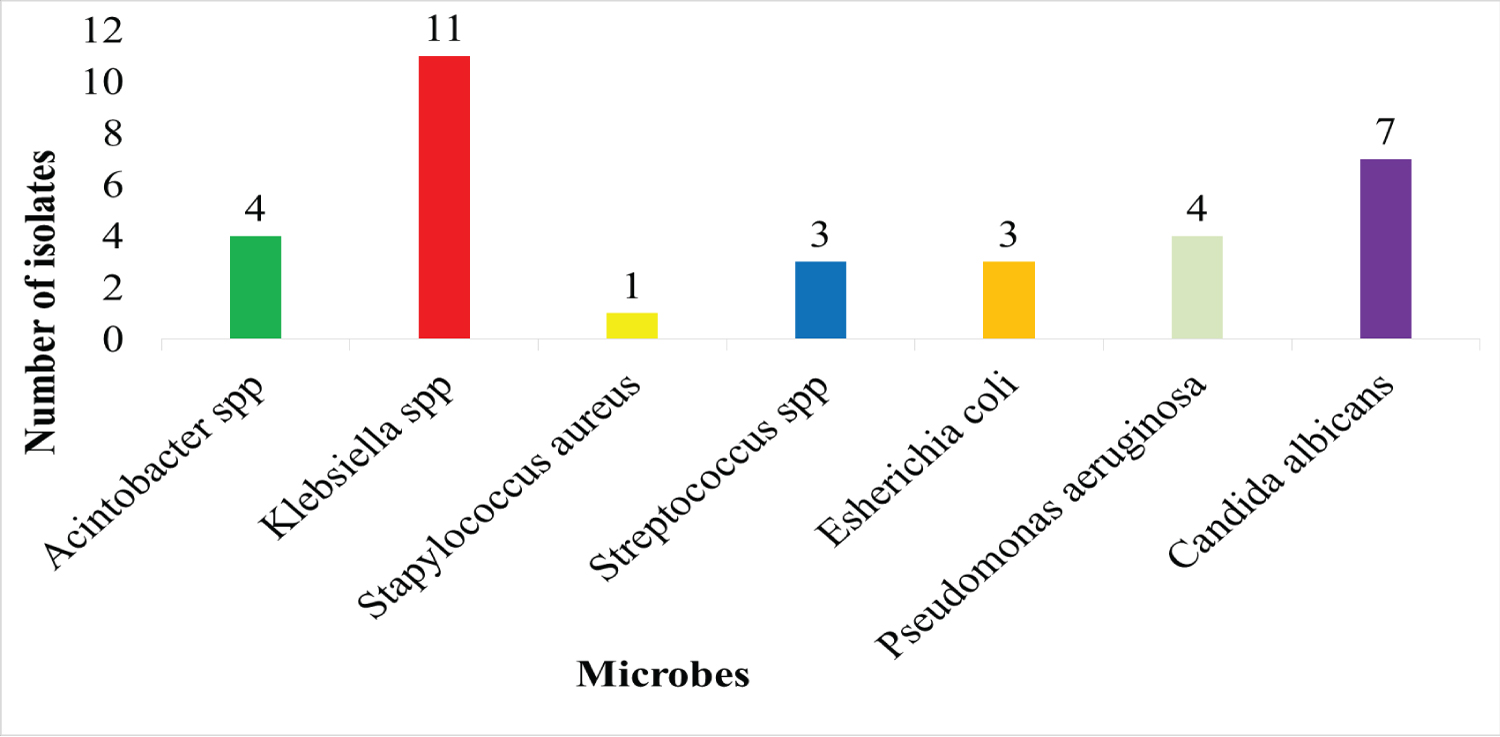 Figure 1: The prevalence of microorganisms isolated from VAP.
View Figure 1
Figure 1: The prevalence of microorganisms isolated from VAP.
View Figure 1
From the obtained results from the sensitivity testing for the isolated bacteria to antibiotics, Staphylococcus aureus was sensitive to CN, CIP, AK (80%), AMC, CXM, IMP, MRP (70%) and CTX, CRO, CST (60%).
While Streptococcus spp (coagulase negative) isolates were resistant to antibiotics AK, CTX, CRO, IMP, while 50% of the isolates were susceptible to CXM CIP, CN, AMC, and 80% of MRP, CST Klebsiella spp E. coli isolates were resistant to most antibiotics, especially AK, AMC, CTX, CRO, CXM and CIP, and 60% of them were sensitive to CST, CN, IMP, MRP. Pseudomonas aeruginosa and Acinetobacter spp were resistant to most of the antibiotics, especially AMC, CTX, CRO, CIP while 55% of the isolates were susceptible to CST, MRP and AK, as Figure 2.
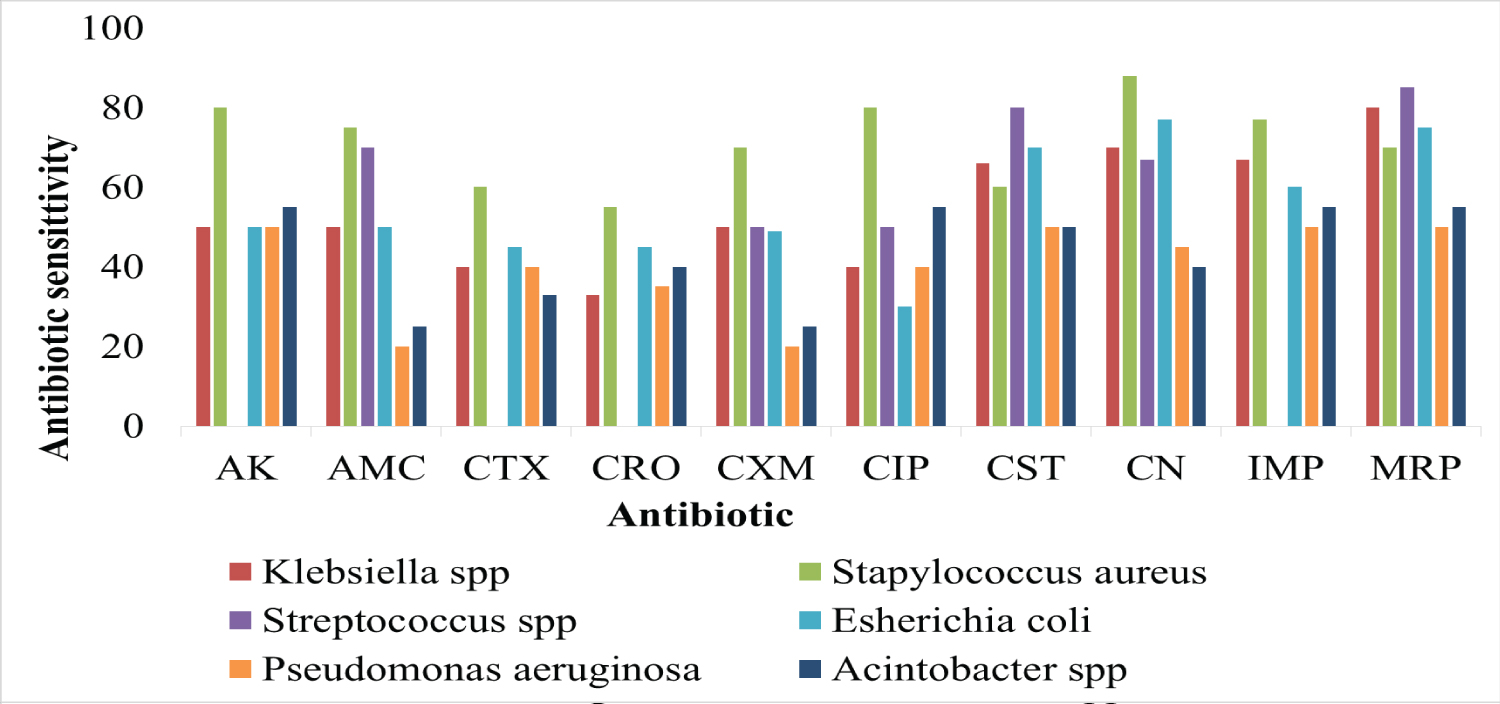 Figure 2: Antibiotic sensitivity.
View Figure 2
Figure 2: Antibiotic sensitivity.
View Figure 2
In this study, the proportion of men to women was similar, whereas in another study, it was significantly higher [19]. A study in Indonesia found similar results, with most cases over 56 years of age [19], which is possibly due to declining immunity with age. Detected microorganisms in this study were similar to those seen in an Indonesian study where most isolates were fungi or Klebsiella species [15], while in an Indian study, most of the isolates were Pseudomonas aeruginosa [17]. According to the present study, most of the Gram-negative bacteria isolates were multi-drug resistant (MDR) and this was in agreement with two studies conducted in France and Indonesia [19,20].
In the current study, it was concluded that Gram-positive bacterial isolates were more susceptible to the most common antibiotics than Gram-negative bacterial isolates, while most Gram-negative isolates were resistant to the antibiotics, especially isolates from Acinetobacter spp and Pseudomonas aeruginosa . To prevent infection in hospitals, It is essential that the critical care department in a hospital performs a proper diagnosis of pneumonia to determine the appropriate antibiotics for the treatment of patients.
The authors would like to thank the Intensive Care Unit team at Hospitals-Misrata for collecting samples and cooperation.