Epilepsy is the most common chronic neurological condition in humans and dogs. Unexplained death is up to three times greater in epileptic humans than the general population with less definitive but supportive evidence of this trend in dogs. Status epilepticus is a life threatening medical emergency defined "as an epileptic seizure greater than five minutes; or more than one seizure within a five minute period without returning to normal between them". Convulsive epileptic seizures may be tonic-clonic with muscle contractions, however, in humans up to 25% of cases may be non-convulsive. There is no equivalent veterinary literature on prevalence in dogs and Non-Convulsive Status Epilepticus (NCS) is very rarely reported in humans with less than a dozen peer reviewed cases in the literature over the past 25 years. Non-convulsive seizures manifest as absence, confusion, disorientation, slow mentation, behavioral changes (including psychosis) and unresponsiveness, while motor signs such a stwitching, head deviation, abnormal positioning, flexion or extension of extremities and automatisms mayor may not be present. It is likely that a large proportion of older dogs presenting with clinical disorientation or unresponsiveness in the absence of myoclonic activity may be experiencing NCS. Without Electro-Encephalogram (EEG), NCS cannot be detected in the comatose or anaesthetized patient and is difficult to differentiate from acute confusion in the conscious patient. We have taken the opportunity to consider a rare and recent human case in our hospital where NCS was induced by electroconvulsive therapy and to compare this with a series of canine cases. Animal models provide valuable insights into conditions that are rare in humans and have been used in many diverse fields of medicine as clues for research, development and management.
One health, Non-convulsive status epilepticus, Translational research
Our human case was a 41-year-old, 62 kg (BMI 19.8 kg/m2) childless professional lady with no history of epilepsy being treated by electroconvulsive therapy form major depression. She had an ongoing history of alcohol abuse (12-14 units per night) and apart from an incidentally discovered Factor V Lieden had no other remarkable medical history. Mirtazapine 30 mg nocte had been discontinued 14 days before the seizure and in the hospital setting alcohol intake was restricted. Other medications were zolpidem tartrate 25 mg nocte, chloral hydrate 20 mls nocte and dothiepin hydrochoride 50 mg tid for insomnia. She was also receiving sodium valproate 400 mg BD and lithium carbonate 250 mg TID and PRN paracetamol 1 g approximately every other night for headache. The only reported drug interaction or allergy previous to this was with penicillin. She had no history of seizures.
She had received eight previous ECT treatments over the past four weeks with no major adverse events, experiencing a minor degree of confusion and no memory loss. Small improvements in symptoms were noted from the seventh ECT treatment, amounting to a marginal improvement in her General Assessment of Function (GAF) score of no more than 10 (improving from a rating of 40-50). This amounted to a lessening of serious social impairment and inability to interact socially to symptoms of flat effect and elective avoidance behaviors. She consented to two further ECT treatments.
CT head revealed no abnormalities 1013.8 MC was administered for 8 seconds. After 15 seconds with no EEG or EMG seizure recorded and despite no physical indication of muscle twitches from an arm that had been isolated from partial neuromuscular blockade or other limbs where reduced convulsion activity would be evident. The EEG then registered bilateral seizure activity, she was considered to be tolerant to benzodiazepine and her current dosage of VPA was 800 mg daily. Phenytoin sodium (15 mg/kg) was administered over a 15 second period by intravenous injection at 28 seconds, but EEG seizure activity persisted for a total of 1130 seconds. It was then terminated with intravenous propofol (2 mg/kg). The patient recovered fully with no adverse effects to memory or headache and the period of recovery and confusion post-anesthetic was not significantly different to other general anesthetic recoveries.
Our case-control observational series of ten animal cases drawn over a period of nearly 20-years included Labrador or Labrador-cross breeds where spontaneous seizure progressed to NCS. Eight cases were female and two male. Five were post-surgical during recovery from neuroleptanalgesia and general anesthesia. All were of normal habitus and none had a history of previous seizure. X-ray revealed no gross intracranial pathology. The median age was 10 years 6-months-old (range 8-12). The stimulus for EEG monitoring was unexplained changes in demeanour, heart rate, breathing or co-ordination; half of these were detected during post-anesthetic recovery. Diagnosis was by EEG. Average seizure length was 855 seconds. In all cases patients were responsive to repeated doses of diazepam or midazolam followed by phenobarbitone fusions.
Discussions between 10 to 30% of people who have status epilepticus are dead within 30 days. The majority have an underlying organic brain condition [1]. Without EEG monitoring NCS can be difficult to detect or differentiate from acute confusional states, transient ischemic attacker stroke.
Only twelve human cases met our criteria via critical appraisal as being of adequate quality (rather than anecdote) to be considered as validated NCS over a 33-year period. Of these nine were female and three male. The median age was 41-years-old. There was one case of an 18-year-old male receiving thoridazine (100 mg/day) and trihexyphenidyl [2], the mean age of the other cases was 45-years-old (range 26-87). Of six cases which described the seizure length the median length was 3.5 days (range 350 seconds to 5 days) with three described as bilateral, two as unilateral and the rest uncharacterized. In one case NCS was described after a single ECT treatment [3], however in all other cases NCS occurred after multiple treatments. The mean number of treatments before NCS was recorded was 6-7 treatments (range 1-9). Both in our case and in others where VPA was used [4] refractory seizures were readily controlled. Similar to this case others also reported incomplete seizure control with phenytoin [3-6] and diazepam [5,7]. In a number of cases including our own, neuroleptic drugs had been used [2,3,5,7] (risperidone) and in this case (quetiapine).
In dogs a common factors were neuroleptic use as a preanesthetic agent (acetylpromazine in 5 cases), increasing age (> 10-years-old - 7 cases) and a tendency to obesity (Labrador genetics) and preponderance of females (8 cases) which are trends also reflected in the human literature.
It is likely that NCS is hugely under-diagnosed in humans and dogs, yet is usually easy to treat and responsive to Na valproate and propofol [8].
A number of potential risk factors for NCSE in humans have been suggested. These have included being over 70 years of age and applying right unilateral ECT stimulation [6], medications that lower seizure threshold such as lithium [5,9], paroxetine [3,10], thioridazine [2] and haloperidol [3,4]. Grogan, et al. [5] suggested these to be necessary cofactors in the precipitation of NCS. However the literature is not conclusive and all of the above have been used safely in many patients [11]. No other cases recorded patient's alcohol intake history or withdrawal and we considered alcohol withdrawal to be a relevant factor in our human case. Benzodiazepine reduction was also feature of this case and is noted to have occurred in three other cases of NCSE after ECT [3,7,12]. In six of nine reported cases of NCS, patients had received multiple ECT treatments (mean 6.5) (Figure 1).
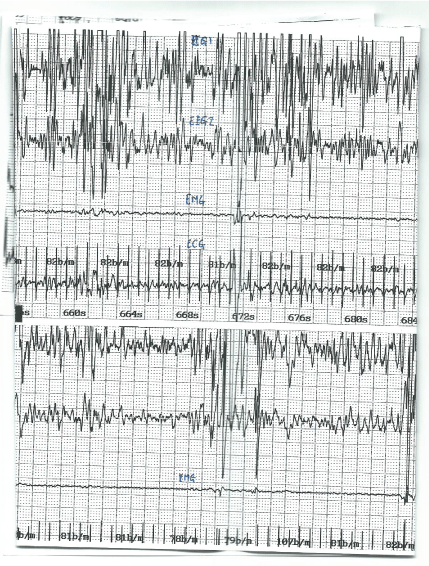 Figure 1: NCSE 0t 660s and 760s, EEG1, 2, EMG, ECG. View Figure 1
Figure 1: NCSE 0t 660s and 760s, EEG1, 2, EMG, ECG. View Figure 1
NCS should be included in the differential diagnosis of patients displaying unusual behavior, particularly after ECT, anesthesia, withdrawal of drugs that Activate the gabba-receptor pathways (including alcohol, benzodiazepines and other anti-epileptic drugs) [13] and introduction of drugs that lower the seizure threshold such as lithium, paroxetine, neuroleptic drugs (including thioridazine, haloperidol, risperidone, quetiapine). Other risk factors for NCS may include: breed of dog, there is insufficient data to suggest predisposition by race in humans, however hereditary factors are clearly involved; perinatal trauma or anoxia; cranial trauma or surgery; metabolic causes (e.g. hypo/hypernatraemia, hypoglycemia, hypocalcaemia, uremia, acutehypoxia); porphyria; toxic causes (e.g. lead, mercury, carbon monoxide), infections (encephalitis, meningitis, neurosyphilis, cerebral abscess, vascular causes (cerebrovascular diseases, cortical venous thrombus, arteritis, vascular malformations), intracranial tumors, degenerative disease (e.g. alzheimers) photosensitivity, sleep deprivation, stress, age (over the equivalent of 70 human years of age or circa 10 years sin dogs with appropriate adjustment for breed).
It is likely that NCS is a greatly under-diagnosed condition in humans and dogs which is life-threatening yet once detected usually easily managed.