Purpose: Intra-operative hypotension (IOH) is associated with a poor post-operative outcome. Consequently, it seems important to reduce the incidence of hypotensive events during anesthesia. The HemosphereTM (Edwards Lifescience Co., Irwin, CA, USA) platform provides the Hypotension Prediction Index (HPI), a predictive marker for a drop in blood pressure within a few minutes. We report the results of one year of application of a simple HPI-algorithm aiming at reducing the incidence of IOH in open abdominal aortic repair (oAAr), a type of surgery during which hemodynamic stability is notably challenging.
Methods: We report the incidence of IOH in 40 patients submitted to oAAr in our tertiary Humanitas Research Hospital in Milan (Italy). The duration and severity of IOH was calculated by mean the Time Weighted Average (TWA) as follows: TWA = [(65-MAP) × time in hypotension]/Duration of monitoring. We compared TWA in our cohort with our preceding case series monitored by FloTrac/EV1000TM.
Results: TWA, expressed as median and IQR range, was 0.3 (0.00÷0.73) mmHg while the duration of an hypotensive event was 2.0 (1.3÷3.1) minutes. The intra-operative time spent in hypotension was 3.54% and the number of hypotensive episodes for each patient was 1.5 (1÷3). TWA resulted lower than the comparison ones and lower than the value observed in the study performed by Wijnberge (TWA = 0.44 mmHg × minute) conducted in major surgery.
Conclusions: In open abdominal aortic surgery, the application of a simple algorithm based on HPI, seems to be able to reduce the incidence, severity and duration of arterial hypotension.
AKI: Acute Kidney Injury; AoXC: Aortic Clamping; AoUC: Aortic Unclamping; CO: Cardiac Output; CI: Cardiac Index; DB: Dobutamine; DAP: Diastolic Arterial Pressure; dP/dt: Pressure Change in Time Unit; Eadyn = dynamic Arterial Elastance; HPI: Hypotension Prediction Index; ICU: Intensive Care Unit; LV: Left Ventricle; MAP: Mean Arterial Pressure; NE: Norepinephrine; PPV: Pulse Pressure Variation; POP: Post-Operative Pain; RCS: Red-Cell Saver; RVR: Regional Vascular Resistances; SV: Stroke Volume; SVI: Stroke Volume Index; SVV: Stroke Volume Variation; TWA: Time Weighted Average
Each living-being shows a peculiar original response to a given injury [1]. Indeed, if we could measure such injury and such response, we would be allowed to better deal with it and correct the impairment resulted, even before it happens, hence prevent it. Such a principle is also true about hemodynamics and its stability assessment, that is the reason we need to monitor the cardio-vascular function. S.A. Magder considers vascular resistances as the best marker of organ perfusion [2] and specifically the regional vascular resistances (RVR) are considered even more accurate [3]. However, we are not able to measure the resistances properly. Thus far the mean arterial pressure (MAP) has been demonstrated and then considered as indicator of the perfusion pressure of the organs [2].
Several studies showed the association between intra-operative hypotension and poor post-operative outcome, in terms of both morbidity and mortality [4-8]. Consequently, it seems crucial to reduce the incidence of hypotensive events during anesthesia.
The Hemosphere TM (Edwards Lifescience Co., Irwin, CA, USA) platform provides the Hypotension Prediction Index (HPI), a predictive marker of blood pressure dropping within few minutes. HPI can predict a hypotension event (i.e. mean arterial pressure < 65 mmHg for 1 minute, at least) in a timeline of 5-15 min and it is based on 23 proprietary model features, derived from the analysis of the arterial wave [9]. Along with the HPI, other parameters are available: Cardiac output (CO), stroke volume (SV), both also indexed, pulse pressure variation (PPV), stroke volume variation (SVV) and the ratio between the two, named dynamic arterial Elastance (Ea dyn ). Ea dyn gives information about the left ventricle (LV) afterload and the ventriculo-arterial coupling. In addition, the slope of the arterial wave provides the dP/dt. This parameter has been demonstrated to be well correlated with the left ventricle (LV) elastance. As a result, dP/dt provides information about contractility of the heart [8-10]. Integrating all the parameters into a simple protocol may help the operator to prevent and/or correct the cause of hemodynamic instability.
The consistency and benefit of HPI in predicting hypotension have been demonstrated by the decrease of the TWA (time weighted average) which summarizes the total time and severity of patient’s hypotension during surgery (i.e., mean arterial pressure, MAP, < 65 mmHg for at least 1 minutes), [11-14]. We tested the use of the “Predict H-Trial” flow-chart made by Lorente-Olazabal, et al. [15], but modified by us, in hemodynamic management of patients undergoing abdominal aorta open repair. This type of surgery is very challenging for keeping hemodynamic stability. This retrospective analysis of our database reports the results of the first year of application of a simple HPI-protocol aiming to reduce the incidence of IOH in abdominal aortic surgery.
Between March 2021 and April 2022 fifty-five patients were scheduled to elective open abdominal aortic surgery under general anesthesia. Exclusion criteria were: age < 18 years, pregnant patients, emergency surgery, persistent atrial fibrillation and subjects who underwent heart-valve replacement. Endovascular aortic surgery or mixed (endovascular + open procedure) surgery, were excluded because in our hospital a different team of anesthetists perform these procedures. Out of the whole group, 15 patients’ data were not completely collected; hence the final sample resulted in 40 cases. For this observational study we followed the STROBE (The Strengthening the Reporting of Observational Studies in Epidemiology) statement recommendations [2]. This study was approved by our institutional Local Ethical Committee (EC appr. 10/21); the study has not been registered on any clinical trial database. Data will be made available on a reasonable request.
All patients received general anesthesia (fentanyl, midazolam and propofol for the induction; oxygen-air mixture and sevoflurane + remifentanil for the maintenance; rocuronium for the myorelaxation). Mechanical ventilation was set as follows: Tidal Volume 7-8 ml/kg; positive end-expiratory pressure 5 cmH 2 O; minute ventilation to maintain EtCO 2 30-35 mmHg and pCO 2 34-38 mmHg; FiO 2 40-60%. All patients received non-invasive blood pressure monitoring, electrocardiogram, heart rate, peripheral oximetry, end-tidal carbon dioxide and hourly diuresis. An arterial line was placed after induction.
Fluids management and Goal Directed Therapy were performed following the algorithm reported in Figure 1.
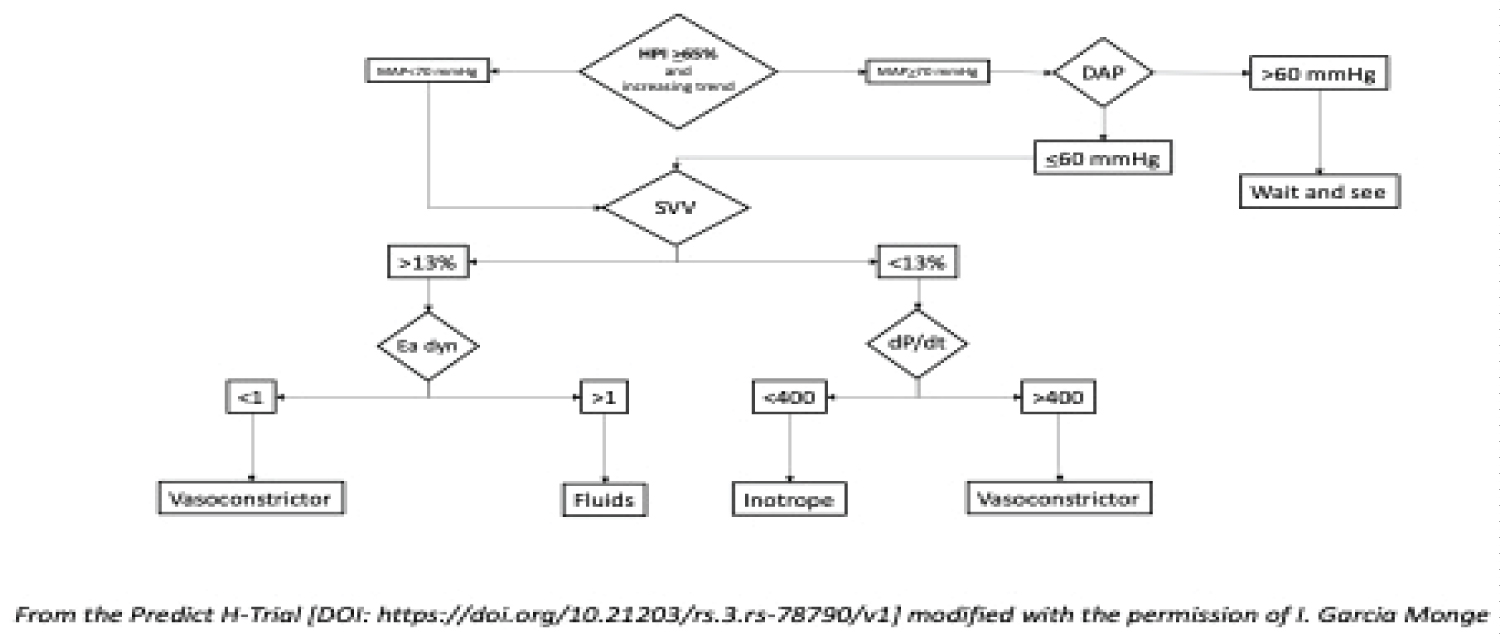 Figure 1: The algorithm.
Figure 1: The algorithm.
From the Predict H-Trial [DOI: https://doi.org/10.21203/rs.3.rs-78790/v1] modified with the permission of I. Garcia Monge.
View Figure 1
Maintenance fluid administration consisted of balanced crystalloids 4-5 ml/kg/h. Intraoperative blood management aimed to keep a level of Hemoglobin > 7 g/dL. We used blood from a Red-Cell Saver (RCS) as first-line choice and Packed-Red Cells (PRC) from the blood bank if RCS was not enough.
Opiates have been used since we do not administer epidural analgesia during and after surgery by more than ten years. For post-operative pain (POP) control, all the subjects received ropivacaine 0.4% 5 ml/h through a supra-fascial catheter inserted by the surgeon at the end of the procedure. The abdominal fascia was anesthetized with Ropivacaine 0.375% + Lidocaine 1% at the beginning of the operation. Multimodality POP control included oral pregabalin (300 mg) given once daily and I.V. paracetamol (1000 mg) given every 6 hours. Rescue analgesia was carried out by non-steroidal anti-inflammatory drug (maximum twice in a day).
Surgical and monitoring time coincide.
All the patients followed our “fast-track” surgery protocol [16]: Surgical ward monitoring (ICU postoperatively admission only if necessary), early oral intake (a cup of tea), mobilization (sitting on an armchair) 4-6h after awakening and a brief walk 2-4h later.
Along with the standard monitoring all patients received the pulse-contour analysis-based hemodynamic monitoring by the Acumen Hemosphere TM device (Edwards Lifescience, Irwin CA, USA) which provides the cardiac output estimation and blood pressure measurements, the hypotension prediction index (HPI), PPV, SVV, the dynamic elastance (Eadyn), cardiac index (CI), stroke volume index (SVI) and arterial dP/dt.
Starting from the “Predict H-Trial” flow-chart by Lorente-Olazabal, et al. [17], we observed that HPI increases when DAP drops < 60 mmHg although MAP is still normal. Therefore, we modified the protocol by lowering the HPI alarm to 65-100 and adding diastolic blood pressure (DAP) control. Our hemodynamic management protocol is shown in Figure 1.
In case of fluid dependence (SVV > 13%), we checked Ea-dyn as it represents fluid responsiveness. If Ea-dyn is < 1, the pressor response to fluids is expected to be weak (so a vasoconstrictor is the best option). Conversely, when Ea-dyn is > 1, blood pressure is supposed to respond to fluid administration [18]. In this case, we administered a fluid challenge of Ringer Acetate of 4 ml/kg within 10-15 min or colloids (albumin 20% 100 ml) monitoring the responsiveness within 3-5 min after the completion of the bolus. If the Ea-dyn was < 1, we administered norepinephrine (NE) 25 mcg i.v. bolus, and if still further needed, after the third bolus we started administering continous NE to decrease the HPI at a value < 65/100.
In case of inotrope drug needing, we tried Ethylephrine (since it has β and α effects) 2 mg i.v. bolus. After the third bolus we started Dobutamine infusion with the aim to maintain dP/dt > 400 mmHg/sec.
Our study is a retrospective analysis about the efficacy of a HPI-protocol in maintaining the hemodynamic stability during the surgery including the two most critical events: The aortic cross-clamping (AoXC) and unclamping (AoUC). For this purpose, we checked TWA in our cohort. TWA was calculated automatically by AcumenTM software, according to the following formula: TWA = [(65-MAP) × time in hypotension]/Monitoring duration. We compared our TWA with the one resulted in a previous cohort of patient monitored with FloTrac/EV1000 TM (Edwards Lifescience, Irwin CA, USA) (mean TWA = 0.45 mmHg; data not published). Intra-operative data were collected at three time points: 20-30 minutes after anesthesia induction (Basal o t0), 5 minutes after AoXC (t1) and 5 minutes after AoUC (t2). Blood-gas-analysis (BGA) was tested at the same timepoints. Postoperative complications recording during the hospital staying were classified as respiratory diseases (Atelectasis or Pneumonia), major cardiac adverse events (myocardial ischemia or arrythmia) and Acute Kidney Injury (AKI) according the KIDGO guidelines, [20]. Other complications included any other minor event spontaneously resolved during the hospital staying.
Data were analyzed as frequencies and percentages for categorical variables and as mean and standard deviation (SD) or as medians and interquartile ranges (IQRs), as appropriate, for continuous variables. We used the ANOVA analysis (with Geisser-Greenhouse correction, as appropriate), Student’s T Test for parametric variables and Wilcoxon rank-sum test and Sign test for non-parametric variables. Normality of distribution was assessed using the Shapiro-Wilkinson normality test. We assumed p < 0.05 for statistical significance. All analyses were performed with STATA (Stata/BE 17.0).
Out of 54 eligible patients, the final sample we considered for the study included 40 patients which data were completely available.
Table 1 shows the characteristic of the population sample. Intraoperative hemodynamic data are reported in Table 2. The time weighted average was 0.3 (0.00-0.73) mmHg, and the median duration of each hypotensive event was 2.0 (1.3-3.1) minutes. The percentage of the time spent in hypotension during the operation was on average 3.54% and the median number of hypotensive episodes for each patient was 1.5 (1-3).
Table 1: Population. View Table 1
Table 2: Hemodynamics management according to the protocol. View Table 2
Stroke volume, cardiac index and dynamic arterial elastance improved significantly along the critical phases of the surgery (p = 0.0015, p = 0.0252 and p < 0.001, respectively). Conversely, even in normal range, mean arterial pressure and dP/dt did not change significantly (p = 0.051 and p = 0.370, respectively). Stroke Volume and Ea-dyn improved significantly from baseline during AoXC (p = 0.0064 and p < 0.001 respectively) and during AoUC (p = 0.0064 and p = 0.0039 respectively). CI increased steadily during the three phases: baseline vs AoXC (p = 0.011) and AoXC vs. AoUC (0.0016).
Twenty-one patients needed vasoactive support, 15 required Norepinephrine and 6 needed norepinephrine and dobutamine. Patients who did not receive vasoactive drugs (n = 19), showed the intraoperative hemodynamic improvement of SV and CI along the three main phases we investigated. Conversely, patients who requested norepinephrine showed improvement only in heart rate (p = 0.0182) and normalization of Ea-dyn (p = 0.0023). Patients who received both NE and DB showed increased CI (p = 0.0035). These results are listed in Table 3.
Table 3: Vasoactive drug administration. View Table 3
Table 4 reports data about the intraoperative fluid management. Crystalloids solutions needing did not differ significantly between patients who received vasoactive drugs and who did not (p = 0.694). Conversely, 90.5% of subjects who received NE + DB needed a higher quote of colloid solutions (p = 0.0015), showed a greater blood loss (p = 0.0029) and received a higher rate of red-cell saver transfusion (p = 0.0014).
Table 4: Intraoperative fluid. View Table 4
The TWA between patients with a safe outcome and patients with complications was not different with a similar duration of the aortic clamping. We observed 6 AKI episodes and 5 pulmonary atelectasis. One case of post-operative atrial fibrillation occurred, and it was pharmacologically treated after cardiologist consultation. Finally, we observed a late recovery of intestinal functions (2 cases) and one case of superior mesenteric artery embolism and serum bilirubin > 1 mg/dL that spontaneously resolved. The needing of vasoactive drugs for hemodynamic support did not result different between patients with a safe outcome and case with postoperative complications, and patients with a complicated outcome showed a longer length-of-stay in comparison with subjects with an uneventful postoperative period (Table 5).
Table 5: Postoperative outcome. View Table 5
The main result of our observational trial was that our protocol of hemodynamic management permitted to reduce the TWA to a median value lower than that Wijnberge, et al. observed in the control group of their HPI validation trial [11]. In those patients they found a TWA value of 0.44 mmHg, whilst in our cases we obtained TWA 0.3 mmHg. Since open aortic surgery is a challenging surgery from a hemodynamic point of view, we consider our result a good outcome.
During the last decade we learned that intra-operative blood pressure control aiming at avoiding hypotensive events, is mandatory to limit the occurrence of post-operative complications [8,19].
Aiming at maintaining a safe mean arterial pressure, we adopted the Acumen IQ HPI software of the Hemosphere platform (Edwards Lifescience, Irwin, CA, USA) which provide the Hypotension Prediction Index (HPI) that alerts and quantify the risk of an incoming hypotensive event by the analysis of the arterial waveform. Since such a monitoring system has not yet been validated in abdominal major vascular surgery, we retrospectively analyzed the results of its application in such a homogenous type of surgical patients.
Abdominal aortic open repair is a challenging surgery for the anesthetist, particularly for the hemodynamic control, both due to the multiple comorbidities the patients and the aortic clamping and unclamping phases, which abruptly provoke changes of the cardiocirculatory setting [20].
Despite the almost unavoidable hemodynamic instability, particularly after the aortic unclamping, the TWA we reached in our cases was higher than the treated group of the validation trial by Wijnberge, et al., but lower than the patients of the control group [11]. The Authors enrolled 60 patients submitted to non-cardiac major surgery and excluded vascular surgery cases. They found a TWA 0.44 (0.23-0.72) mmHg into the control group and a TWA 0.10 (0.01-0.43) mmHg into the treated group in which they adopted a protocol more complex than the one we used. The median TWA we reached was 0.3 mmHg, between those two values. Considering that in vascular surgery avoiding hemodynamic instability is almost impossible it could be a satisfactory result. Moreover, in a case series of 38 abdominal aortic aneurysmectomy in open surgery, monitored by the FloTrac/EV1000 platform (Edwards Lifescience, Irwin, CA, USA), we observed an average value of TWA 0.4 mmHg and a longer duration of hypotension [data not published]. This could be another point in favor of our first experience with the HPI employment in major vascular surgery.
We consider that the administration of vasoactive drugs was coherent with the maintenance or even the improvement of hemodynamics. In fact, patients who received amines support had a similar or improved haemodinamic trend compared to patients that did not receive amines support. It may mean that our algorithm suggested the administration of such drugs properly.
In a trial of HPI application in 214 surgical patients, Maheshwari, et al. found that it was unable to prevent and reduce the intraoperative incidence of hypotension [21]. Although they observed a TWA lower than ours, it is noteworthy that in many cases the clinicians declined to intervene according to their protocol maybe because it was too complex, by their own admission. The Authors reported that “ a simplified treatment algorithm focused on prompt use of vasoactive drugs may reduce hypotension ”.
Our study has several limitations. First, since it is an observational retrospective trial, the results cannot be extended to a general population. Second, since it was a retrospective observational trial, our algorithm of hemodynamic management, even if it showed to be effective, was not compared to a control group. Third, we did not analyze if secondary targets (duration of hypotension < 10.3% of total time) could be associated with postoperative complications because they were not recorded. Finally, the biggest limitation is that we did not investigate if this protocol can reduce complications and improve outcomes. A randomized controlled trial for this should be desirable [22].
In conclusion, intraoperative hypotension often occurs in major surgery procedures, negatively impacting the outcome. Then, Hypotension Prediction Index along with the other parameters provided by the Hemosphere platform (Edwards Lifescience, Irwin, CA, USA), could be helpful to manage hemodynamics also in abdominal major open vascular surgery. Further prospective studies are desirable to confirm or not our results.
Data will be made available on a reasonable request.
E.G. is a proctor for Edwards Lifescience.
No funding received.
E.G.: Study design, manuscript editing; F.N.: Study design, manuscript editing; F.G.: Data collection; R.A.: Statistic analysis; M.I.G.M.: Manuscript revision; M.C.: Manuscript revision.
Thanks to prof. Paul A. Kelly for the English Language revision. https://profesor-ingles-online.com/home-english-teacher-online/