Venlafaxine, a serotonin-norepinephrine reuptake inhibitor (SNRI) antidepressant, has been associated with severe cardiotoxicity in massive overdoses. Treatment is usually supportive. Over the past decade, intravenous lipid emulsion has been gaining interest in the treatment of intoxications with lipophilic substances. We report a case of refractory pulseless electrical activity cardiac arrest following a massive overdose of venlafaxine successfully treated with intravenous lipid emulsion infusion.
Intravenous lipid emulsion, Venlafaxine toxicity, Serotonin-norepinephrine reuptake inhibitor (SNRI), Toxicology, Overdose
Although newer antidepressants carry less toxicity than older tricyclic antidepressants, the SNRI venlafaxine has been reported to be significantly more toxic than selective serotonin reuptake inhibitors (SSRIs) [1]. Serotonin toxicity and seizures are the most important clinical effects in overdose [2] but there is also a concern regarding cardiotoxicity with reports of acute heart failure [3], Takotsubo cardiomyopathy [4], and arrhythmias [2] especially with large ingestions.
Treatment of venlafaxine overdoses, apart from decontamination procedures such as activated charcoal, is usually supportive. Intravenous lipid emulsion (ILE) therapy is an accepted therapy for local anesthetic systemic toxicity (LAST) and has lately been associated with successful outcomes in life-threatening overdoses involving various lipophilic intoxicants [5].
We report a case of refractory cardiovascular collapse secondary to a massive venlafaxine overdose treated successfully with ILE therapy.
A 58-year-old African American man with a history of major depressive and anxiety disorders presented to the emergency department (ED) immediately after witnessed ingestion of approximately 100 pills of extended-release venlafaxine (150 mg 24 hr SA Capsules - total of 15 grams) after being discharged from mental health clinic in an attempt to remain in the hospital. In the ED, he underwent gastric lavage and received activated charcoal about 2 hours after ingestion under the direction of local poison control and was admitted to the hospital for observation. Approximately 7 hours after admission to the hospital, he developed tachycardia with telemetry strip as noted in Figure 1. Shortly after this, he developed intractable vomiting followed by respiratory distress with eventual seizure activity. A rapid response was called for status epilepticus and respiratory failure. He was given intravenous benzodiazepines and loaded with anti-epileptic for seizures as well as intubated and sedated for respiratory failure. Upon arrival to the intensive care unit (ICU), he developed a wide complex pulseless electrical activity (PEA) cardiac arrest. Eventual return of spontaneous circulation (ROSC) was obtained after CPR w/ACLS protocol; however, PEA cardiac arrest continued to recur multiple times despite ongoing supportive efforts and correction for known abnormalities such as acidosis with bicarb, shock with vasopressors, and electrolyte abnormalities. EKG during a period of ROSC demonstrated tachycardia with widened QRS and prolonged QT interval (Figure 2). Bedside transthoracic echo (TTE) demonstrated new onset of global hypo-kinesis with significantly depressed ejection fraction. The refractory and recurrent PEA episodes were considered possibly secondary to serotonin-norepinephrine reuptake inhibitor (SNRI) toxicity and IV Lipids (20% fat emulsion: Bolus of 1.5 ml/kg in 60 seconds followed by an infusion at a rate of 0.25 ml/kg/min over 60 minutes) were administered at the next observed PEA arrest. No further PEA or cardiac arrest episodes were noted. The patient demonstrated improvement in his heart rate as well as QRS, QT intervals (Figure 3). With ongoing continued supportive care including tracheostomy, temporary renal replacement therapy, nutrition via PEG tube and sub-acute rehab, the patient eventually recovered and was discharged from the hospital approximately 6 weeks after admission. Ultimately, he was decannulated, feeding tube was removed and he began living independently again 4 months after his overdose. His cardiac function also demonstrated complete normalization on follow-up TTE and a left heart catheterization was free of any coronary artery disease. Currently he is employed and living with his family.
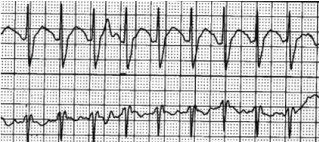 Figure 1: Tele strip.
View Figure 1
Figure 1: Tele strip.
View Figure 1
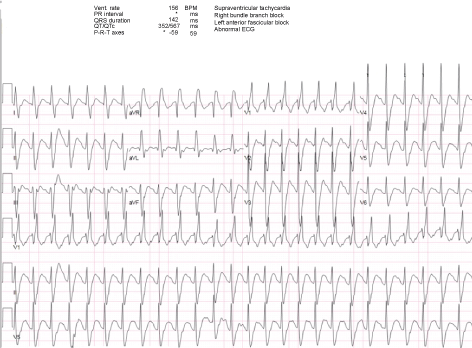 Figure 2: EKG.
View Figure 2
Figure 2: EKG.
View Figure 2
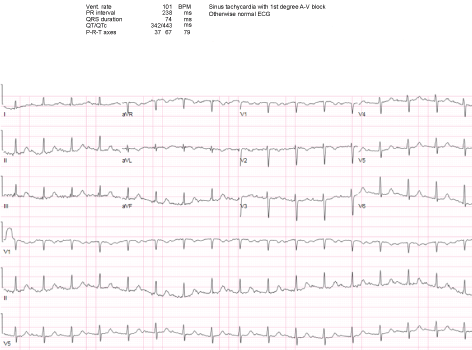 Figure 3: EKG.
View Figure 3
Figure 3: EKG.
View Figure 3
Antidepressants are common in intentional drug overdose and have been reported to be involved in around 20% of fatal cases. The newer antidepressants are less toxic than TCAs, but the relative toxicity of different agents vary. Venlafaxine, an SNRI, is considerably more toxic than SSRIs but still much less than the TCAs [1].
Venlafaxine exerts its effect by inhibiting the neuronal re-uptake of serotonin and norepinephrine [6] and to a lesser extent also inhibits the re-uptake of dopamine [3]. Toxic manifestations in venlafaxine overdose include tachycardia, hypertension, seizures and serotonin toxicity. Severe cardiotoxicity including arrhythmias and/or hypotension is not a major feature except for in cases of massive ingestions (> 8 g) [2]. When cardiotoxicity does occur, it can be severe with reports of venlafaxine associated Takotsubo cardiomyopathy, acute coronary syndromes with normal coronary arteries, and malignant arrhythmias [4,7]. There are also cases of acute heart failure with cardiogenic shock including a case series from Belgium describing 4 patients admitted to the ICU with severe acute left heart failure and cardiogenic shock where 3 out of the 4 patients passed away despite aggressive critical care and vasoactive support.
The treatment for SSRI/SNRI poisoning is generally supportive and fatal outcomes are relatively uncommon [8,9]. However, some patients can become critically ill with shock refractory to standard therapies including vasoactive medications. When faced with a clinical scenario where a patient is rapidly decompensating despite maximal aggressive standard therapy the clinician may have to resort to less rigorously studied or proven interventions. In this case utilization of intravenous lipid emulsion therapy led to a successful outcome.
Intravenous lipid emulsion has long been used in parenteral nutrition but has more recently gained interest as an antidote for toxicity caused by lipophilic agents [5]. It is an accepted treatment for toxicity from local anesthetics [5,10] but has also lately been described in several case reports as a successful intervention for severe toxicity from other lipophilic agents including betablockers, calcium channel blockers, anticonvulsants, antipsychotics, benzodiazepines, cocaine, antidepressants, HCTZ and zopiclone [11].
The mechanism of action of intravenous lipid emulsion as an antidote is not entirely elucidated and several mechanisms have been postulated [5,11,12]. "The lipid sink theory" postulates that the infusion of lipids intravenously creates an expanded plasma lipid phase that absorbs lipophilic molecules and lowers the active free plasma concentration resulting in a re-established equilibrium within the plasma. Another theory suggests that ILE decreases inhibition and other toxic metabolic effects of the "poisons". A third theory suggests that ILE may act via a direct inotrope action as fatty acids are known to increase cardiac myocyte calcium levels resulting in an increased intramyocyte calcium concentration.
There is no randomized controlled human trial investigating intravenous lipid emulsion therapy in overdose. The evidence of efficacy comes mainly from animal studies and case data [5]. Data about side effects come mainly from the use of intravenous lipids as parenteral nutrition and includes allergic reactions, pancreatitis, bacteremia, fat embolism, heart failure, lipoid pneumonia and ARDS [5,11]. Dosing of the intravenous lipid emulsion varies between reports but commonly a bolus of 100 ml of 20% lipid emulsion is given (or 1.5 ml/kg) followed by a continuous infusion of 0.25-0.5 ml/kg/minute [5,10,11]. A recommendation not to exceed 12 ml/kg of intravenous lipid emulsion has been proposed [5].
Given the scarcity of good quality evidence, the use of intravenous lipid emulsion therapy in non-local anesthetic poisoning is controversial. However, it has been deemed reasonable by the American College of Medical Toxicology, the American Heart Association and the European Resuscitation Council to use for patients with intractable hemodynamic instability, or overt cardiac arrest, secondary to lipophilic drug toxicity when proven refractory to standard care [5].
Our case adds to the growing body of literature supporting the use of intravenous lipid emulsion in the treatment of lipophilic drug intoxications outside of the usual indication of local anesthetic systemic toxicity. It is our belief that the intravenous lipid emulsion therapy was a life-saving intervention in our reported case. Unfortunately, the data remains limited in this area and routine use of intravenous lipid emulsion therapy outside the context of refractory life-threatening conditions failing standard therapies cannot be recommended at this stage.
Intravenous lipid emulsion therapy has become standard therapy for local anesthetic systemic toxicity. In recent years, case reports have accumulated describing the successful use of intravenous lipid emulsion therapy in life-threatening overdoses of other lipophilic toxins. Presented is a case of refractory cardiovascular collapse and cardiac arrest secondary to a massive venlafaxine overdose successfully treated with intravenous lipid emulsion. High-quality data on the use of intravenous lipid emulsion therapy for non-local anesthetic toxicity remains limited. Utilization of such practices outside the context of severe hemodynamic instability due to overdoses refractory to standard treatment cannot be recommended but may warrant further exploration. However, our case report adds to a growing body of literature supporting the use of intravenous lipid emulsion in the treatment of intoxications outside of the usual indication of local anesthetic systemic toxicity.