We designed this Observational prospective cohort study to assess the safety, effectiveness and efficiency of the introduction of Regional Citrate Anticoagulation (RCA) as election strategy for Continuous Renal Replacement Therapies (CRRT). All patients in need of CRRT without contraindications to the use of RCA were included. It was necessary to adapt and implement the original RCA protocol, for the first time in a Spanish ICU. A total of 90 patients between October 2016 and October 2017 were included. 324 systems were used, 294 (90.74%) were electively removed without any functioning problem. 30 sets (9.25%) had to be removed due to catheter related problems (21, 6.48%) or protocol violation (9, 2.77%). Median filter lifespan was 60.45 hours. System stability and ionic calcium levels were easily achieved in 93.51% of patients. No significant side effects or complications associated with the use of RCA were observed. This method, compared to the use of heparin in the past in our unit (alone or in combination with epoprostenol) turned out to be more efficient. We concluded that, in our experience, the use of RCA is safe, predictable, effective and efficient in critical patients undergoing CRRT.
Acute Kidney Injury (AKI), Continuous Renal Replacement Therapies (CRRT), Continuous VenoVenous HemoDiaFiltration (CVVHDF), Regional Citrate Anticoagulation (RCA), Citric acid
Patients with Acute Kidney Injury (AKI) needing Continuous Renal Replacement Therapy (CRRT) represent about 4-5% of all patients admitted to Intensive Care Units (ICU); while more than 65% of patients admitted to intensive care develop criteria for AKI according to the RIFLE classification [1]. CRRT is the most used renal support in critically ill patients with AKI in the context of Multiple Organ Dysfunction Syndrome (MODS) due to its better haemodynamic tolerance, slow and continuous liquid removal and its ability to depurate high blood volumes during 24 hours a day [2].
In order to achieve maximum effectiveness of CRRT it is essential to ensure patency of the extracorporeal circuit. Clotting of the filter is the most common cause of CRRT interruption with the consequent loss of blood, dose, time and money.
Unfractionated Heparin (UFH) has been, historically, the first-choice strategy in CRRT. The biggest advantage of its use was its low cost, ease of administration and monitoring, and the availability of an antidote (protamine). However, the problems arising from systemic action (increased risk of bleeding, development of heparin-induced thrombocytopenia and immune disorders) and high variability of effectiveness in critically ill patients explain how circuit patency and side effects are unpredictable using this method, making it the Achille's heel of CRRT nowadays.
Therefore, a new therapeutic strategy in keeping extracorporeal circuits permeable, avoiding premature coagulation and systemic effects in the patient was necessary. In this context emerges the Regional Citrate Anticoagulation (RCA) [2]. RCA has doubtless advantages over heparin anticoagulation in CRRT and has been used since early 90's, [2,3] but has not become the first-choice strategy because it was, also historically, a very complex and dangerous technique.
In the last 20 years we have witnessed the gradual emergence of technological advances that automate CRRT to unbelievable limits. Particularly in the last five years these hardware and software developments have been focused on RCA, which has led to its current development [4].
The aim of this study was to evaluate safety, effectiveness and efficiency of the introduction of a RCA protocol in CRRT.
After developing a RCA protocol based on the one described by Borgström (Kalmar Hospital, Kalmar, Sweden) and providing theoretical and practical courses to ICU staff, a prospective single-center observational cohort study was developed: Its objectives were to assess the safety (occurrence of bleeding complications, electrolyte disorders and acid-base balance), effectiveness (filter lifespan), efficiency (economic analysis) and the practical applicability of the protocol. The study was conducted between October 2016 and October 2017. This study fits Helsinki Declaration recommendations. Authorization from local Clinical Research Ethics Committee was requested. In all cases the authorization of the patient or their legal representatives for participation was obtained, after receiving comprehensive information on the characteristics of the study.
All adult patients admitted to ICU needing CRRT were considered eligible. The contraindications for RCA initiation we considered were:
Mandatory systemic anticoagulation due to previous patient conditions (heart valve prosthesis, deep vein thrombosis, pulmonary embolism); no anticoagulation indication (thrombocytopenia < 50,000; International Normalized Ratio > 2 or Activated Partial Thromboplastin Time > 60 seconds); presence of fulminant hepatic failure (bilirubin > 6 mg/dl); serious calcium metabolism disorders (total serum calcium > 12 mg/dl). CRRT start and end were left to the discretion of the attending physician.
To apply CRRT, Prismaflex monitors (Gambro Lundia AB, Lund, Sweden) were used with software version 6.2 and acrylonitrile-sodium-metanil sulfone membranes (AN69ST) (Prismaflex ST150, 1.5 m2, Gambro, Meyzieu, France), electively replaced every 72 hours of use, following manufacturer's recommendations. The therapy mode was continuous veno-venous hemodiafiltration (CVVHDF). Both CVVHDF and CVVHF are available modes with RCA. The target dose of treatment was set between 25 and 40 ml/Kg/h, dynamic dose adjustment was performed, adapted to the needs of the patient. To do so, we applied the flow settings of a higher weight than the patient's real weight in order to offer a higher dose and the opposite way around, when the needed dose was getting lower. As prefilter liquid infusion we used a diluted citrate solution (trisodium citrate 18, citric acid 0, 140 Na+, Cl- 86 mmol/L, PrismoCitrate 18/0 ®, Gambro, Sondalo, Italy), as dialysis solution a commercial calcium free liquid was used (HCO3 22 Lactate 3, Na+ 140, Cl- 106, Ca++ 0, Mg++ 0.5 mmol/L, Prism0cal B22 ®, Gambro Lundia AB, Lund, Sweden) a bicarbonate based solution was used as postfilter reposition liquid (HCO3- 32, K+ 4, Na+ 140, Cl- 113.5, Ca++ 1.75, Mg++ 0.5 mmol/L, Prismasol 4, Gambro Lundia AB, Lund, Suecia). Calcium chloride 10% (Ca++ 9.13, Cl- 9.13 mEq/10 ml, Braun Calcium Chloride ® 100 mg/ml, B. Braun Medical SA Barcelona, Spain) was administered through the monitor's syringe pump. For vascular access triple lumen catheters with a double shotgun barrel tip design were implanted in the right internal jugular vein with ultrasound guidance as first choice or double lumen catheter in the right femoral vein as an alternative (Power Trialysis ®, 13 F, 15 cm and Niagara ®, 13.5 F, 24 cm, respectively. Bard, New Jersey, USA). Citrate is not yet FDA approved as a regional anticoagulant in CRRT. It should be noted that none of those companies influenced in any way the start, design, analysis and publication of the results of this study.
For system anticoagulation, the citrate solution is added to patient blood when entering the extracorporeal circuit, the monitor adjusts the citrate flow to reach an initial concentration of 3 mmol of citrate per blood liter. Citrate chelates ionized calcium (iCa) causing a concentration drop from physiological values, 1 to 1.3 mmol/L to the target of 0.25 to 0.35 mmol/L. This blood, mixed with the citrate solution, and lacking iCa, i.e, unable to clot, is propelled towards the hemofilter where convection and diffusion processes take place. Subsequently, after the replacing solution is added, the circulating blood is returned to the patient. The syringe pump system, infusing the precise amount of calcium chloride to the plasma to return iCa levels to its physiological values to not alter systemic coagulation of the patient, either through the third jugular catheter lumen or through another central venous catheter dedicated lumen, as it needs a steady flow of replacement and the ease with which the compound may precipitate if mixed with other drugs or fluids (Figure 1).
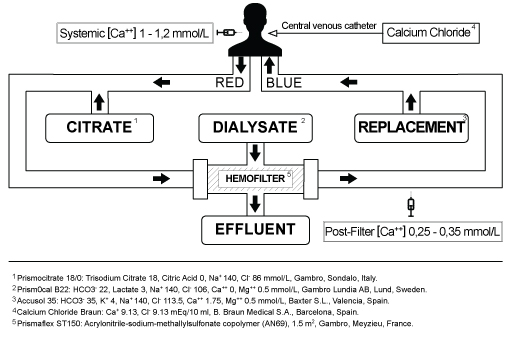 Figure 1: Diagram of Regional Citrate Anticoagulation - CVVHDF circuit and the main materials used to perform it. View Figure 1
Figure 1: Diagram of Regional Citrate Anticoagulation - CVVHDF circuit and the main materials used to perform it. View Figure 1
As RCA control method, paired blood gas samples (patient and post filter) were analyzed at 0, 1, 3, 6 and 12 hours of CRRT, checking iCa levels. If the baseline patient's ionic calcium level was less than 1 mmol/L we supplement it with calcium gluconate until its normalization before starting the CRRT. Corrections following our protocol were made when needed. After reaching the system stability, controls were performed, at least, every 12 hours, or by medical criteria at any time.
According to our protocol, the citrate infusion rate was adjusted to the blood pump flow to reach an initial filter citrate concentration of 3 mmol/L. This concentration was subsequently adjusted in increments or decrements of 0.5 mmol/L, according to control results to maintain an iCa target concentration in the circuit between 0.25 and 0.35 mmol/L. The post filter replacement flow was set by protocol to reach the prescribed CRRT dose, always dynamic, depending on the patient characteristics. The calcium infusion rate to the patient was completely automatic calculated by the monitor software by estimating calcium loss to the effluent, starting at 100% and being adjustable, with variations of 10% depending on the controls, to ensure patient blood iCa concentration of 1 to 1.2 mmol/L. Extraction was set and changed by medical order adjusting it to the clinical characteristics of each patient (Figure 2).
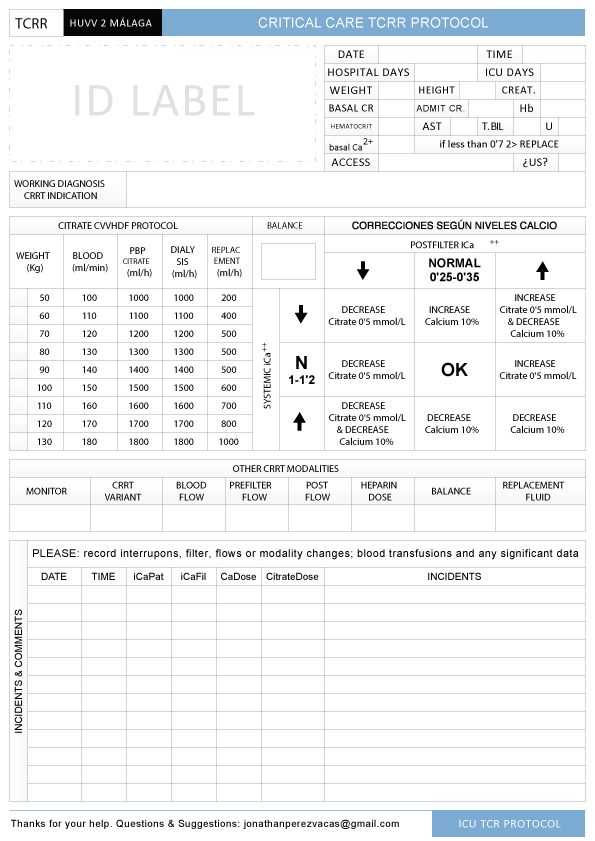 Figure 2: RCA-CRRT protocol designed by Dr. Daga and Dr. Pérez-Vacas. View Figure 2
Figure 2: RCA-CRRT protocol designed by Dr. Daga and Dr. Pérez-Vacas. View Figure 2
The baseline characteristics of the subjects included in the study and the variables related to therapy administered were collected. The qualitative variables are presented with their frequency distribution and quantitative variables as mean and standard deviation (SD) or median and interquartile range (IQR) in case they were not normal distribution. For statistical analysis SPSS ® v15 was used (Chicago, IL. Version 15.0 for Windows).
Between October 1 2016 and October 31 2017, 106 patients were eligible for the study of which 16 had to be excluded: 8 due to coagulation disorders in which no anticoagulant was used in the circuit; 4 due to severe liver failure, 1 secondary to acetaminophen poisoning and the other 3 in the context of MODS, in these cases UFH was used at low doses to keep circuit patency; 3 patients were not considered eligible for RCA because systemic anticoagulation was indicated for other reasons, 2 patients with mechanical heart valves and 1 patient with pulmonary embolism, systemic perfusion of UFH was used in this patients; One patient was excluded from the study due to severe hypercalcaemia (total Calcium > 14 mg/dl) in the context of neoplastic disease (Figure 3).
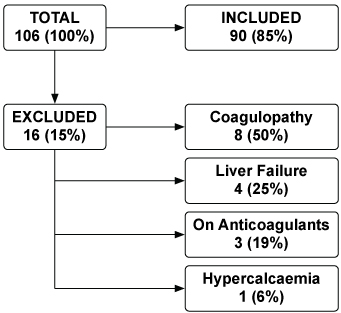 Figure 3: Diagram of included and excluded patients. View Figure 3
Figure 3: Diagram of included and excluded patients. View Figure 3
Finally, 90 patients were included, whose average age was 59.93 ± 14.73 years, male predominance with 70.00%. The mean APACHE II score on admission was 17.77 ± SD 8.67. The length of stay of patients in the ICU was 17 days, with IQR between 7 and 28 days. CRRT was maintained for 6.5 days, IQR 4 to 17.25 (Table 1).
Table 1: Baseline Characteristics of the 90 study patients. View Table 1
324 sets of CRRT, with a median duration of 62.45 hours (IQR 51.25 to 65) were used. 294 systems (90.74%) were removed electively. 285 of them (87.96%) were replaced around 72 hours running, on recommendation by the manufacturer, it was usually possible to coordinate filter change with patient transfers outside the ICU for further tests or surgical treatments. The remaining 9 filters (2.77%) were removed prior to 72 hours of operation for medical reasons, while they were working properly. 30 sets (9.25%) were substituted early due to coagulation, in 21 cases (6.48%) due to catheter related problems and the remaining 9 (2.77%) due to protocol violations. Catheter problems cause high pressure peaks and constantly blood pump stopping, iCa concentration become unstable and the system finally coagulates. Non-ultrasound guided insertions, suboptimal selection of insertion place and agitated/uncooperative patient were the most common reasons for catheter related problems. Regarding protocol violations, they occurred mainly in the first RCA treatments, due to operator's inexperience, for example by hanging reposition bags in their respective scales wrong or by varying flows established by protocol. However, the learning curve of the technique resulted to be very fast in our centre (Figures 4 and Figure 5).
 Figure 4: Diagram of filters used and the reasons for its withdrawal. View Figure 4
Figure 4: Diagram of filters used and the reasons for its withdrawal. View Figure 4
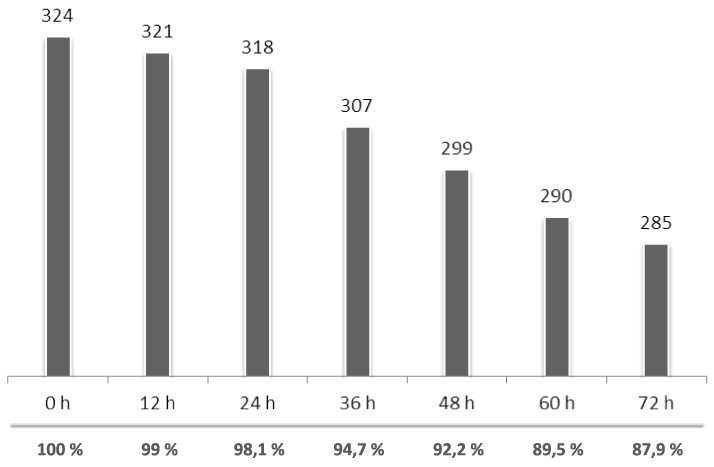 Figure 5: Chart of normally functioning filters per hour. View Figure 5
Figure 5: Chart of normally functioning filters per hour. View Figure 5
iCa levels in the circuit reached the target of our protocol (0.25-0.35 mmol/L) in the vast majority of patients, 303 (93.51%), with the initial dose of 3 mmol/L citrate. Patients diagnosed with acute pancreatitis and those suffering multiple transfusions prior to the beginning required more controls and RCA adjustments to achieve target iCa levels, because they normally had iCa concentration below 1.0 mmol/L at the beginning of RCA-CRRT. After initially experiencing problems we decided to correct iCa levels in every patient to reach iCa higher than 1 mmol/L before start. Patients with moderate hepatic dysfunction were subjected to stricter metabolic monitoring, without observing side effects of RCA on them. The iCa levels in both the system and the patient are represented in Figure 6.
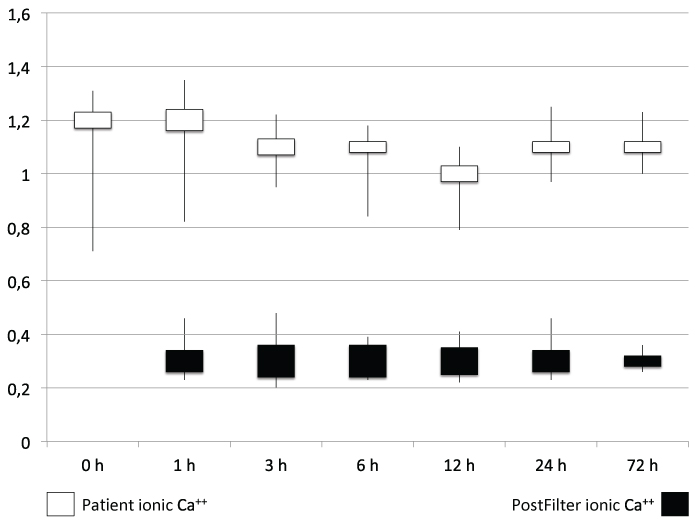 Figure 6: Chart of ionized calcium levels during RCA-CRRT. View Figure 6
Figure 6: Chart of ionized calcium levels during RCA-CRRT. View Figure 6
Total Calcium/Ionized Calcium ratio higher than 2.5 was not exceeded in any patient, we didn't find any clinically significant episodes of hypocalcaemia or metabolic alkalosis to suspect citrate accumulation [5]. Patients undergoing RCA, as every CRRT patients in our centre, presented need for control and ion replacement, we particularly saw a trend to hypomagnesaemia, easily controlled in all patients. In no case flows pumps were modified to change the acid-base status being that one of the objectives of the study was to evaluate our protocol and its capability of homeostasis control (Table 2).
Table 2: Analytical parameters evolution. View Table 2
There were no bleeding events related to RCA-CRRT. Given the absence of clinically significant side effects, it was not necessary to discontinue the therapy in any of the patients in our sample. Several clinical sessions were needed during our protocol implementation.
For the analysis of efficiency, as since the introduction of the RCA in our centre, the use of heparin is exceptional and epoprostenol null, an indirect comparison of current real cost involving RCA treatment against which meant in the past the use of heparin and/or epoprostenol was performed. To do this we rely on historical data of our unit. Being aware of the obvious methodological limitations and that it is a purely theoretical exercise, we performed the comparison based on a duration of 6.5 days (156 hours) in a patient of 82.35 Kg at a dose of 37.64 ml/Kg/h on the three compared branches: Citrate, heparin alone and heparin-epoprostenol. Considering only fluids and filters the use of heparin alone resulted a 23.27% more expensive than using citrate and, in the heparin-epoprostenol therapy it resulted a 57.49% more expensive than citrate. If in addition to the price of liquids and filters, we consider also the cost of blood transfusions associated to anticoagulation with heparin, heparin strategy was an 88.05% more expensive; if, furthermore, the use of epoprostenol is necessary the difference increases to 122.27%. In this comparison nursing staff workload involved in each strategy is not taken into account, the three times higher filter lifespan using citrate, would be expected to widen the gap in favour of citrate; Nor has it taken into account the cost of analytical controls of each strategy, considering that neither gases nor the determination of APTT are extremely expensive in general, this term shouldn't greatly modify the result. In any case, we found RCA as the most economical anticoagulation strategy (Table 3).
Table 3: Theoretical economic comparison. View Table 3
Our work presents the limitations of being a single-centre study, not randomized, but we believe it accurately reflects the daily reality of Intensive Care Units in Spain.
The permeability of the extracorporeal circuits, necessary for CRRT, was achieved in the extracorporeal circuit by administering a continuous infusion of UFH, so this strategy was considered the method of choice to maintain circuit patency [6].
In this observational study we share our initial experience with a new anticoagulation strategy: RCA as the first option to keep permeable extracorporeal circuits. We confirmed in this period that its implementation was easy, safe, effective and efficient [7-10].
Our goal has been localized to the applicability of the RCA in a general ICU with extensive experience in CRRT and with widely trained staff in these therapies. In this sense, the study may show some limitations in the field of external validity.
On the other hand, the results of this study would not apply to patients with severe liver failure or severe calcium metabolism disorders, which were specifically excluded by our protocol.
Using the protocol and monitoring described above we started RCA-CRRT, regardless of bleeding risk. Even in post-surgery patients or those with a previous bleeding history, the use of RCA-CRRT produced no hemorrhagic event and consequently no transfusion need [11].
In our experience, using our protocol, we found no mayor metabolic disorders secondary to the use of the RCA [12,13] (alkalosis, acidosis, hypo or hypercalcaemia), we did not find evidence of accumulation of citrate in patients included in the study [14]. The most common, and expected, laboratory findings were mild hypomagnesemia and hypophosphatemia. Monitoring patients undergoing RCA-CRRT is very important to ensure good metabolic control. If the protocol is followed in a strict way, possible metabolic alterations are easily detected and controlled and corrected without causing negative clinical consequences.
Considering the average filter lifespan, which we have defined as effectiveness, citrate CRRT compared with heparin alone shows consistently higher filter durability using citrate in all studies [15,16]; in our experience it was three times higher (62.5 vs. 20.05 hours). This extension in the lifetime of the extracorporeal circuit allows a good correlation between prescribed and administered doses, prevents blood loss by frequent changes of the extracorporeal circuit and reduces the nursing workload.
From the efficiency analysis point of view, understood as the ability to perform a high quality CRRT at the lowest possible cost, RCA-CRRT is more efficient than UFH.
In view of our results RCA-CRRT is safe, effective and efficient. These results have led RCA as the strategy of choice for anticoagulation in all CRRT prescribed in our centre, using UFH only in those patients requiring systemic anticoagulation for other reasons or in the few patients with contraindications to the use of Regional Citrate Anticoagulation.
None declared.