During cardiac surgery, extracorporeal circulation (ECC) causes lung injury. In these inhomogenously affected lungs, the pressure control ventilation (PCV) cannot adequately ventilate differently damaged lung compartments. We invented and used original multilevel lung ventilation method named 3-LV based on alternating 3 or more pressure levels, ventilation frequencies and delivered tidal volumes. The goal of this article is to compare lung mechanics in cardiac surgery patient after ECC using standard PCV when compared to 3LV ventilation.
This study was performed on 88 cardiac surgery patients after disconnection from ECC; the patients were randomly (allocation by weekdays) divided into two groups and ventilated by PCV and 3LV. Group 1 (n = 44) started with 1 hour PCV followed by a second hour with the 3-LV mode. Group 2 (n = 44) was ventilated in the reverse order. Measured parameters were statistically evaluated by the Student's paired t-test.
The static compliance (Cst) and PaO2/FiO2 ratio in 3-LV ventilation mode improved by 25-32% (p < 0.01) and 31% (p < 0.01), in group actually ventilated by 3LV ventilation. The respiratory rate after weaning in Group 1 significantly decreased compared with that in Group 2 (p < 0.05). An improved CO2 washout was observed in each group after switching to 3LV.
3-LV showed a better lung recruitment ability compared with PCV in patients after cardiac surgery, without using high PEEP level.
Lung recruitment, Multilevel lung ventilation, Pressure control ventilation
Anaesthesia and extracorporeal circulation (ECC) during prolonged cardiac surgery often cause lung injury, which is usually represented by grade 1 ARDS (PaO2/FiO2 ratio = 200-300). This phenomenon is already well documented [1] and our own experience shows that the lung mechanics and transport of O2 are significantly impaired during and after anaesthesia [2]. Placing the patient in a supine position during operation increases the dead space over tidal volume ventilation (VD/VT) by approximately 15-20%. The decrease in pulmonary compliance is 10-20% on average and up to 35% in obese persons. The geometry of the alveoli is usually impaired by their cyclical collapsing and reopening, which leads to surfactant damage [2,3]. The typical complications on the level of alveoli are atelectasis, atelectotrauma, biotrauma, impaired ventilation-to-perfusion ratio (Qs/Qt), increased permeability of the alveolar-capillary membrane for liquids and the increase in ventilation work after disconnecting from artificial lung ventilation (ALV). The application of high concentrations of O2, i.e., FiO2 > 0.6-0.9, leads to resorption atelectasis [4,5]. Because of considerable effect on the alveolar geometry, this condition may lead to significant postoperative complications. In many cases, the complications mentioned above can be prevented by a reduced usage of high O2 concentration, retaining the alveolar geometry by maintaining positive end expiratory pressure (PEEP). Applying the principles of "protective ventilation" by performing a lung recruitment manoeuvre during the postoperative period by using necessary PEEP may help. However, a new ventilation technique called programmed multilevel ventilation (PMLV), in this case, "programmed three-level artificial lung ventilation (3-LV)", could be an even better solution. 3-LV is our original method developed especially for the recruitment and ventilation of inhomogeneously affected lungs, which is typical in ARDS of various origins. The pressure control ventilation (PCV) mode in ARDS appears to be less effective because even with the best frequency and pressure adjustment, it cannot achieve an optimal distribution of gases to the lung compartments, which are differently affected. Our objective was to determine the lung recruitment ability by using the 3-LV ventilation technique in comparison with the commonly used conventional PCV mode in postoperative cardiac surgery patients after weaning from ECC. In this paper, we evaluate the parameters of lung mechanics, oxygenation and postoperative adaptation to spontaneous ventilation in 3-LV compared with PCV.
The study was approved by the ethics committee of the East Slovakian Institute of Cardiovascular Diseases: EK no. VZ/7 / KardO/2011. Head of Committee Stanislav Juhas, MD, PhD.
This prospective study was performed starting from February 2014 to January 2017. It comprises 88 cardiac surgery patients, with cardiac arrest lasting 50 min (79 ± 26 min) in ECC. After being disconnected from the ECC, the patients were randomly divided into two groups ventilated by alternating PCV and 3-LV techniques in different orders. Protocol: Group 1 (n = 44) starts with 1 hour of PCV followed by 3-LV mode in the second hour. Group 2 (n = 44) was ventilated in the reverse order, thus starting with 3-LV for 1 hour followed by PCV, as described in the time flow chart in Figure 1. Patients undergoing scheduled cardiac surgery were assigned. Patients undergoing operation on Monday, Wednesday and Friday were assigned to Group 1, while the ones in Group 2 underwent operation on Tuesday and Thursday. The exclusion criteria were a chronic lung disease (bronchial asthma, chronic obstructive pulmonary disease (COPD), ejection fraction of the left ventricle (LVEF) < 30% and low lung compliance at the beginning of anaesthesia with dynamic compliance (Cdyn) < 30 ml*cm H2O-1. The demographic parameters can be found in Table 1.
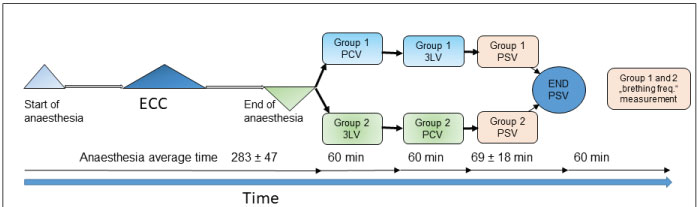 Figure 1: Time flow chart of study from start of anaesthesia to disconnection from ventilation.
Figure 1: Time flow chart of study from start of anaesthesia to disconnection from ventilation.
Average time of anaesthesia: 283 ± 47 min, PCV mode in Group 1: 60 minutes and 3LV mode in Group 2: 60 minutes. Next step 3LV mode in Group 1 and PCV mode in Group 2. Next step PSV mode in both groups: Approx 65 ± 18 min. Next step "T" trial and extubation. Measurement of the frequency of spontaneous breathing during the subsequent two hours.
View Figure 1
Table 1: General demographic parameters as well as numbers of cardiac surgery performed by type: CABG, coronary artery bypass graft, AVR aorta valve replacement, MVR mitral valve replacement or ring, and TVR tricuspid valve replacement or ring. Surgical procedures are mostly combined. View Table 1
Anaesthesia: Identical anaesthesia was applied to both groups in the following combination: Propofol (Fresenius) at the introduction to anaesthesia at a dose of 150 ± 41 mg, a 2.5- to 5-mg bolus of Midazolam (Accord) at the introduction, 15 - 24 µg of Sufentanyl (Janssen) at the introduction to anaesthesia, a continuous dose of Sufentanyl forte (Jansen) of 0.66 ± 0.12 µg* kg-1*h-1, a 50-mg bolus of Atracurium (GSK) repeated as necessary, and a minimal alveolar concentration of Sevofluran (Abbott) of (MAC) = 0.6-1.1 or 0.6 ± 0.2 during ECC. The patients were intubated with an 8.0 mm orotracheal tube. Artificial lung ventilation (ALV) during anaesthesia was performed using the principles of protective ALV listed in Table 2. FiO2 was maintained if possible at not higher than 0.5, and ETCO2 was > 35 but ≤ 41 mmHg (5.6 kPa). Measured parameters: During anaesthesia and during ALV in the postoperative period, the ventilation parameters, parameters of lung mechanics, static compliance (Cst), airway resistance (Raw), oxygenation parameters, concentrations of blood gases and haemodynamic parameters were monitored. After admission to the intensive care unit (ICU), the patients were ventilated according to the study protocol. Ventilator Chirolog Servoventilator AURA-V with a computer assistance module ProfiLungs (Chirana Medical, Slovakia) was used. During ALV the changes in the parameters of lung mechanics, oxygenation index, values of ETCO2, and minute ventilation (MV) were monitored. The pressures applied and the extent of changes of ventilation volumes during 3-LV were also monitored. The measured values of specific tidal volume/ml*kg-1/ (VTspec) fluctuated depending on the individual transition pressure levels of the 3-LV within the ranges of 1.9-2.8 ml*kg-1, 2.9-4.9 ml*kg-1 and 5.0-7.2 ml*kg-1 in 7-8%, 60 - 75% and 15 - 20% of the breathing cycles, respectively. 3-LV caused variable volume, pressure and time ventilation. After weaning from ventilation and extubation, the spontaneous breathing frequency as an indicator of adaptation to spontaneous breathing was observed and afterwards compared between both groups.
Table 2: Average ventilation parameters: MV - Minute Ventilation; VTs - Tidal Volume (specific) in ml per kilogram; f (PCV) - Frequency of Pressure Control Ventilation; f (PEEPh) - Frequency of PEEPh High (PEEP of high level); PEEP - Positive End Expiratory Pressure; f (PC by 3LV) - Frequency of Pressure Control by 3-Level Ventilation; Paw max (PCV) - Maximal Airway Pressure during Pressure Control Ventilation; average PEEPh (3-LV) - Average Positive End Expiratory High Pressure during 3-Level Ventilation; Paw max (3-LV) - Maximal Airway Pressure during 3-Level Ventilation. View Table 2
It is well known that in classical ALV regimes, even the best frequency and pressure optimization procedure of ALV parameters cannot optimally distribute gases to individual but differently damaged compartments [6]. Hence, a single ventilation mode with the ventilation frequency (f), time of inspiration-to-expiration ratio (TI: TE), respiratory volume (VT), positive end-expiratory pressure (PEEP), and ventilation support pressure (Ppc) in pressure controlled mode as the fixed parameters cannot optimally ventilate inhomogeneous lungs [6,7]. Differently damaged compartments are dispersed in whole lungs. It is not possible to divide the flow of gases into each separate lung compartment using fixed ventilation parameters [2,5]. Fixed ventilation parameters allow optimal ventilation for only some compartments of the lungs with well-suited mechanical properties. Other compartments will be ventilated more or less sub-optimally. One possible solution is to use a ventilator with programmed 3-LV to generate variable volume, pressure and time ventilation cycles. It can improve the distribution of gases into the variably damaged lung compartments. The setting of the ventilation parameters of 3-LV is derived from continuously measured lung mechanics by a computer in the Aura-V ventilator with the ProfiLungs system. The principle of pressure changes during 3-LV compared with PCV is schematically depicted in Figure 2.
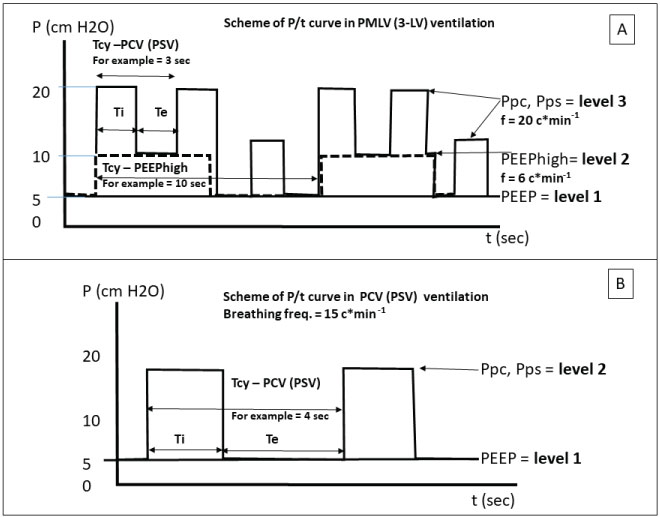 Figure 2: Panel A: Diagram of the pressure curves in 3 level ventilation mode (3-LV). P - Pressure; Tcy PCV(PSV) - Time of Ventilation Cycle of PCV (PSV); Tcy PEEP high- Time of Ventilation Cycle of PEEPhigh; Te - Expiratory Time; Ti - Inspiratory Time; t - Time in Sec. Panel B: Diagram of the pressure curves in pressure control ventilation mode (PCV/PSV). P - Pressure; Ti - Inspiratory Time; Te - Expiratory Time; Tcy PCV(PSV) - Time of Ventilation Cycle of PCV (PSV); t - Time in Sec.
View Figure 2
Figure 2: Panel A: Diagram of the pressure curves in 3 level ventilation mode (3-LV). P - Pressure; Tcy PCV(PSV) - Time of Ventilation Cycle of PCV (PSV); Tcy PEEP high- Time of Ventilation Cycle of PEEPhigh; Te - Expiratory Time; Ti - Inspiratory Time; t - Time in Sec. Panel B: Diagram of the pressure curves in pressure control ventilation mode (PCV/PSV). P - Pressure; Ti - Inspiratory Time; Te - Expiratory Time; Tcy PCV(PSV) - Time of Ventilation Cycle of PCV (PSV); t - Time in Sec.
View Figure 2
The acquired parameters of lung mechanics and oxygenation index in both groups were statistically evaluated by Student's paired t-test (Microsoft Excel).
Based on the data shown below, the 3-LV ventilation method achieved better parameters than PCV. We compared 3LV to PCV using the following parameters: Lung mechanics, oxygenation, carbon dioxide elimination and haemodynamic. For the assessment of recruitment efficiency, the adaptation to breathing after disconnecting from ALV, the oxygenation of arterial blood and Cst were evaluated. The effect of ALV on the circulatory system was also considered. After admission to the ICU, the acute lung injury score (LIS - Murray score) was in the range of 1.5 ± 0.25 points, confirming moderate lung impairment. The change in ALV methods does not cause a significant decrease in the mean arterial pressure (MAP). The average MAP before and after the application of 3LV was 79 ± 8 vs. 75 ± 6 mmHg and 83 ± 8 vs. 79 ± 7 mmHg in the first and second groups, respectively (p = NS. Cardiac output was measured using Monitor Vigileo (Arrow)), the influence of ventilation on haemodynamics was assessed. In all cases, as an indirect indicator of peripheral perfusion and CO, we measured the central venous blood oxygen saturation (ScvO2). After the patient was introduced to ALV and disconnected from extracorporeal circulation (ECC), the arterio-alveolar difference in oxygen (A-aDO2) significantly increased 2 to 4-fold, (average 201 ± 42 mmHg, p < 0.01). After the first hour of ALV, the average SaO2 and SvO2 were within the physiological range 94-100% and 67-77%, respectively. This indicates adequate peripheral perfusion and sufficient CO. The value of the PaO2/FiO2 ratio after weaning from ECC decreased significantly in both groups, to below 300 Torr, which corresponds to grade 1 ARDS (according to the Berlin classification). The PaO2/FiO2 ratio after admission to the ICU was 249 ± 24 mmHg in Group 1 and 266 ± 25 mmHg in Group 2 (p = NS). ALV lasted 3.3 ± 0.4 hr and then the patient was disconnected from ALV. The PaO2 in Group 1 vs. Group 2 was 384 mmHg vs. 321 mmHg (p < 0.01). During 3-LV, we observed in both groups an increased EtCO2 in the first 25 minutes of using 3-LV, which indicated an increasing area of gas exchange as a sign of lung recruitment (Figure 3). The Cst was monitored every 10 minutes and showed major improvement in Group 1 compared with Group 2. (p < 0.01). In all cases when 3-LV was applied, regardless of whether it was applied for the first or second time, the Cst was improved (+ 30%) compared with PCV (p < 0.01). This effect was observed regardless of the order in which the 3-LV technique was applied (Figure 4). At 60 and 120 minutes after extubation, the breathing frequency as an indicator of adaptation to spontaneous breathing was assessed. In both cases, it was higher in Group 2 than in Group 1 (p < 0.05). In Group 1, Cst was higher than that in Group 2. The improved adaptation in Group 1 was probably caused by the lower ventilation work (Table 3).
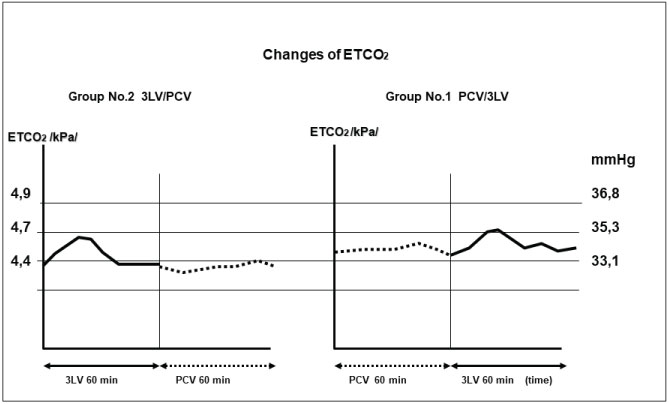 Figure 3: Changes in ETCO2 during 3-LV and PCV in Group 1 and Group 2 during 2 × 60 minutes of ventilation.
View Figure 3
Figure 3: Changes in ETCO2 during 3-LV and PCV in Group 1 and Group 2 during 2 × 60 minutes of ventilation.
View Figure 3
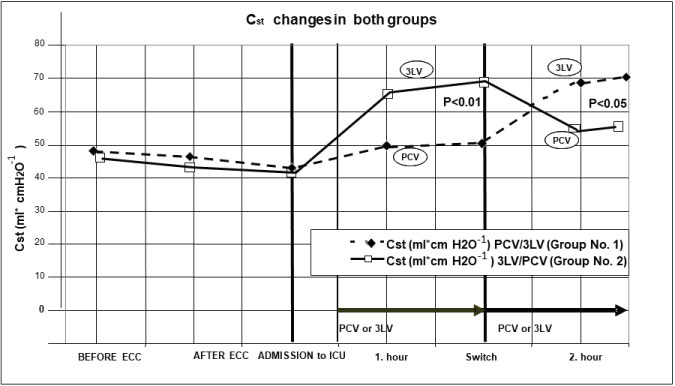 Figure 4: Cst - static compliance changes in patients in Groups 1 and 2. Before ECC, After ECC, admission to intensive care unit (ICU), 1 hour after admission to ICU, Switch to another ventilation mode, and 2 hours after ICU (1 hour after switching of ventilation mode).
View Figure 4
Figure 4: Cst - static compliance changes in patients in Groups 1 and 2. Before ECC, After ECC, admission to intensive care unit (ICU), 1 hour after admission to ICU, Switch to another ventilation mode, and 2 hours after ICU (1 hour after switching of ventilation mode).
View Figure 4
Table 3: Frequency of spontaneous breathing 1 hour (panel A) and 2 hours (panel B) after disconnection from ALV in Group 1 and Group 2. A significant difference was found in the respiratory frequency of spontaneous breathing between groups 1 and 2 (p < 0.05). The frequency of spontaneous breathing was higher in Group 2 in which Cst was lower before disconnecting from a ventilator. The adaptation of patients to spontaneous inspiration within 120 minutes after extubation was faster in Group 1 than in Group 2. A prerequisite for improved adaptation is probably lower ventilation work. View Table 3
It is well known that use of ECC during cardiac surgery is followed by substantial signs of lung impairment. The PaO2/FiO2 ratio decreases to values indicating the presence of grade 1 ARDS [1]. The acute lung injury score (LIS) after the surgical procedure fluctuated on average at 1.5 points, which indicates moderate lung impairment. The mechanism of impairment is multifactorial [1]. During anaesthesia, there is a decline in A-aDO2 associated with moderate hypoxemia and caused by the increased disproportion of ventilation-perfusion due to the decreased ventilation of perfused parts of lungs [2]. A major disproportion of ventilation-perfusion was observed after weaning from ECC. Seventy percent of resorption atelectases arise predominantly in dependent lung zones, which are correlated with the number of hospitalization days [3]. The decrease in Cst in all monitored groups of patients after disconnection from ECC is a sign of development of atelectasis and impairment of alveolar geometry [3]. Mols, et al. [5] define atelectasis as a condition of the absence of gas in an alveolus causing its long-term collapse. Atelectotrauma as a cyclically repeated collapse and filling (opening) of the alveolus during a ventilation cycle. Alveoli open during inspiration and close during expiration. Alveolar recruitment is characterized by the re-opening of a previously collapsed alveolus, whereas derecruitment is the collapse of an alveolus previously opened by recruitment. Ongoing recruitment (lasting recruitment) is defined as a condition of permanent alveolar recruitment, which does not allow derecruitment. From a mechanical-pneumatic point of view, it is the opposite of atelectasis.
All groups of our patients demonstrated a decrease in Cst after being disconnected from ECC as a sign of atelectasis. Our opinion that early recruitment using adequate PEEP or another lung recruitment technique (in our case, 3-LV) has a beneficial effect was already described by Borges, et al. [8]. With increasing the FiO2 value we are able to improve the PaO2, but this may cause absorption atelectasis and hyperoxia; therefore, we did not use it [4,9]. The high FiO2 may have an adverse effect on CO and peripheral vascular resistance (PVR) [10]. We applied this concentration of O2 to maintain PaO2 in a recommended range of 75 - 160 Torr, (10.0 - 21 kPa) [11]. It is well known that increased FiO2 and PaO2 in ICU patients increases mortality compared with patients with adequate PaO2 [9,11]. Peak pressure applied during 3-LV had a minimal impact on circulation if adequate PEEP was used. During 3-LV with the application of adequate PEEP and the minimum possible peak airway pressure (Paw max), a moderate or insignificant decrease in MAP and the heart rate was observed. Similarly, no significant desaturations of venous blood as an indicator of a decrease in cardiac output (CO) was observed during 3-LV. Lung recruitment by 3-LV is probably achieved by irregular changes in respiratory volume, pressure and breathing frequency during each respiratory cycle. Non-constant respiratory volumes, inspiration times and ventilation pressures probably enhanced the opening of collapsed but recruitable compartments [12]. The benefit of 3LV ventilation could be confirmed by the improvement of PaO2/FiO2 and lung compliance as well as by a reduction of A-aDO2 [13]. According to Johnson [2], the increase in Cst is a marker of lung recruitment effectiveness. This finding concurs with our results. The frequency of spontaneous breathing after disconnecting from a ventilator is a good indicator of spontaneous breathing work. Increasing breathing frequency is the most significant predictor of respiratory failure [14]. Therefore, we used this parameter to compare respiratory stability after disconnection from 3-LV or PCV.
1. We compared the effectiveness of PCV with the 3-LV ventilation method in postoperative cardiac surgery patients with ECC. Based on the monitored Cst, PaO2/FiO2, EtCO2 we assessed the lung recruitment, impact on circulation and spontaneous breathing stability.
2. The application of 3-LV compared with PCV during postoperative period improved arterial blood oxygenation PaO2/FiO2, the elimination of CO2, the stability of alveolar geometry (Cst) and the transition from artificial ventilation to spontaneous breathing.
3. The advantage of using 3-LV is effective lung recruitment without using a high PEEP. The mean airway pressure value was comparable with PCV group.
4. In the cases of inhomogeneous distribution of gases in the lungs, for example, ARDS, SARS - (Severe Acute Respiratory Syndrome), H1N1 pneumonia, and lung oedema, 3-LV could be successfully used for lung recruitment and the improvement of gas distribution in such damaged lungs.
5. The effectiveness of 3-LV compared with classic ventilation methods (PCV, PS, SIMV, etc.) will probably be higher.