Several lines of evidence suggests that cytosolic Low Molecular Weight Protein Tyrosine Phosphatase (cLMWPTP), encoded by ACP1 gene, could be a candidate gene for T2D. Previous studies, however, have not found a direct association between enzyme phenotype and susceptibility to the disease but have only found associations with its clinical manifestations. The data reported in present note suggest that in subjects with advancing age a significant direct associations between cLMWPTP and susceptibility to T2D may indeed exist.
Two hundred seventy four subjects with T2D from the population of Penne were studied. All patients gave informed verbal consent to participate in the study that was approved by the Sanitary Direction of Hospital. 380 subjects without T2D were studied in the same population as controls. cLMWPTP phenotype was determined by starch gel electrophoresis. Statistical analyses were carried out by SPSS programs.
In patients aging more than 67 years at onset of T2D (corresponding to 90° centile of age distribution) the proportion of cLMWPTP phenotype is statistically different from that of patients aging 67 years or less at onset of the disease and from that of controls. Most of the difference is due to ACP1 B and CB phenotypes: B phenotype shows a progressive decrease of frequency with advancing age at the onset while CB shows an increase of frequency with advancing age of onset. Environmental, alimentary and life style factors could anticipate the onset of T2D: these factors may overshadow the effect of cLMWPTP on susceptibility to T2D: this effect seems to emerge in subjects experiencing the disease in advancing age.
An important role of cytosolic Low Molecular Weight Protein Tyrosine Phosphatase in glucose metabolism is suggested by several lines of evidence: Insulin receptors and Band 3 Protein (B3P) are hydrolyzed by cLMWPTP [1]; diabetic subjects with low enzymatic activity have low glycemic level [2,3]; lowering enzyme activity by a specific antisense oligonucleotide improves insulin sensitivity and decrease blood glucose level in obese mice [4]. On these basis cLMWPTP could be considered a candidate gene for Type 2 Diabetes. In our previous study, however, no statistically significant direct association has been observed between this enzyme and susceptibility to T2D but only an association with clinical manifestations of the disease [5]. In this report we show that a statistically significant association between cLMWPTP activity and susceptibility to T2D is indeed present but in older subjects only.
cLMWPTP is encoded by ACP1 (Acid Phosphatase locus 1) that shows three alleles *A, *B and *C and correspondingly six genotypes with increasing activity in the order *A/*A < *A/*B < *B/*B ≤ *A/*C < *B/*C < *C/*C. The enzyme is composed by two isoforms F and S that differ each other for enzymatic activity and molecular proportion among phenotypes and probably for physiological functions also (Table 1). The enzyme acts as phosphotyrosine-protein phosphatase and it is able to hydrolyze phosphorylated insulin receptors and B3P influencing glucose metabolism. The enzyme acts also as flavin-monocleotide phosphatase regulating flavo-enzymes activity and energy metabolism [6].
Table 1: F and S isoform concentrations in relation to the ACP1 genotype. View Table 1
Two hundred seventy four subjects with Type 2 Diabetes (T2D) were studied in the population of Penne, a rural town in the east side of Italy. These patients have been considered in previous studies also [3,5]. 380 subjects without T2D were studied in the same population as controls. Informed consent was obtained to participate to the study that was approved by the Direction of the Hospital. These subjects were studied a few years ago before the establishment of an Ethical Committee.
Acid Phosphatase locus 1 (ACP1) phenotype was determined by starch gel electrophoresis according to Hopkinson, et al. [7].
Chi-square test of independence, variance analysis and correlation analysis were carried out by SPSS programs [8]. Three way contingency table analysis was carried out by a log linear model according to Sokal and Rohlf [9].
Table 2 shows the distribution of some clinical parameters in relation to the age at onset of T2D. 67 years corresponds to the 90° centile of the distribution of age at onset. A border line difference is observed for blood glucose level and positive family history for diabetes: Both are lower in subjects with advancing age at the onset pointing to a low importance of positive family history in subjects older at the onset.
Table 2: Distribution of clinical parameters in relation to age at the onset of T2D. View Table 2
Table 3 shows the distribution of ACP1 phenotypes in relation to age at the onset of the disease. A statistically significant difference in the distribution of ACP1 phenotypes is observed in patients aging more than 67 years at the onset as compared both to subjects with an age at the onset equal or less than 67 years and to general population. This suggests that in older subjects there is a significant association between ACP1 and susceptibility to disease. In younger subjects such association is probably overshadowed by other factors that prevail on the effect of ACP1. The statistical analysis shows that most of difference between subjects aging more than 67 years and those aging 67 years or less or controls is due to a lower frequency of B phenotype and a higher frequency of CB phenotype in older subjects.
Table 3: Distribution of ACP1 phenotypes in relation to age at the onset of T2D. View Table 3
Figure 1 and Figure 2 depict the relationship between age at the onset and B and CB phenotypes respectively. Starting from an aging at the onset equal to 53 years (50° centile of distribution of age at the onset) with advancing age at the onset is observed a progressive decrease of the frequency of B phenotype with a minimum after 73 years (Figure 1). Concerning CB phenotype (Figure 2) an increase of CB is observed after 67 years only with a maximum after 73 years (95° centile of the distribution of age at the onset).
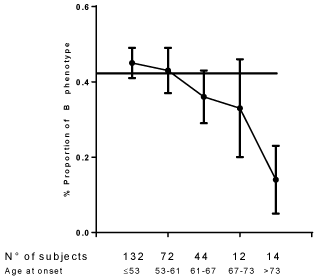 Figure 1: The relationship between the proportion of B phenotype and age of onset of type 2 diabetes. Rank correlation: p < 0.01. The horizontal line indicates the proportion of B phenotype in the general population. View Figure 1
Figure 1: The relationship between the proportion of B phenotype and age of onset of type 2 diabetes. Rank correlation: p < 0.01. The horizontal line indicates the proportion of B phenotype in the general population. View Figure 1
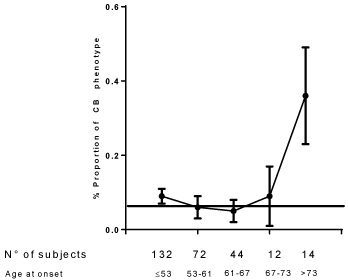 Figure 2: The relationship between the proportion of CB phenotype and age of onset of type 2 diabetes. Linear correlation: p = 0.091. The horizontal line indicates the proportion of CB phenotype in the general population. View Figure 2
Figure 2: The relationship between the proportion of CB phenotype and age of onset of type 2 diabetes. Linear correlation: p = 0.091. The horizontal line indicates the proportion of CB phenotype in the general population. View Figure 2
Table 4 shows the age at the onset of diabetes in relation to ACP1 phenotype. In subjects aging 53 or less at onset no significant relationship is observed between ACP1 phenotype and age at the onset.
Table 4: Age at the onset of diabetes in relation to ACP1 phenotype. View Table 4
In subjects aging more than 53 years a significant relationship is observed due to a highly significant difference between B and CB phenotypes: B phenotype shows the lowest and CB the highest age at the onset of diabetes. We have considered the possible effects of sex and obesity in subjects aging more than 53 years at the onset: A statistically significant association between ACP1 genotype and age at the onset is observed in males (p = 0.05) and in non obese subjects (p = 0.01) but not in females (p = 0.551) and in obese subjects (p = 0.970).
Figure 3, Figure 4 and Figure 5 show respectively the mean concentration of F, S isoforms and F/S ratio in relation to age at the onset of diabetes. F isoform concentration is lower while S isoform concentration is higher in subjects aging more than 67 years as compared to those aging 67 years or less. A lower F/S ratio results in subjects aging more than 67 years (2.83) at the onset as compared to patients aging 67 years or less (3.31, p = 0.32).
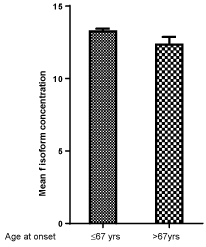 Figure 3: Mean ACP1 F isoform concentration in relation to age at onset of diabetes: p = 0.110. View Figure 3
Figure 3: Mean ACP1 F isoform concentration in relation to age at onset of diabetes: p = 0.110. View Figure 3
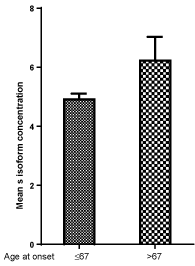 Figure 4: Mean ACP1 S isoform concentration in relation to age at onset of diabetes: p = 0.047. View Figure 4
Figure 4: Mean ACP1 S isoform concentration in relation to age at onset of diabetes: p = 0.047. View Figure 4
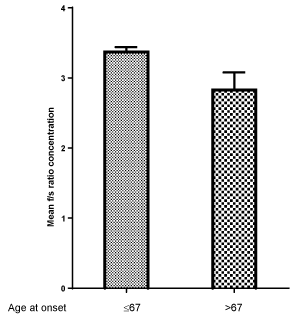 Figure 5: Mean ACP1 F/S ratio concentration in relation to age at onset of diabetes: p = 0.032. View Figure 5
Figure 5: Mean ACP1 F/S ratio concentration in relation to age at onset of diabetes: p = 0.032. View Figure 5
We have performed a three way contingency analysis by a log linear model on the association among age at the onset, ACP1 and positive family history for T2D. The analysis has shown a statistically significant association between age at the onset and ACP1 (p = 0.027) and a border line association between age at the onset and positive family history (p = 0.07). There is an additive contribute of ACP1 and positive family history to age at the onset (p = 0.015). In patients aging at the onset more than 67 years, positive family history is less frequent and CB is more frequent as compared to patients aging 67 years or less, (data not shown).
A statistically significant association between ACP1 and T2D is observed in patients aging 67 years or more at the onset of the disease. Subjects with B phenotype (high F isoform) seem to be predisposed to an early onset while subjects with CB phenotype (high S isoforms) to late onset. In subjects aging more than 53 years at onset there is a significant association of ACP1 phenotype with age at the onset mainly due to B and CB phenotypes.
Environmental, hereditary and life style factors could anticipate the onset of T2D. Positive family history seems to be one of the factors that anticipate onset. These factors may overshadow the effect of ACP1 on susceptibility to T2D: The effect of the enzyme could emerge when other factors have already exerted their action. It is also possible that metabolic modification due to aging favors the emergence of a significant role of ACP1 in the susceptibility to diabetes.
An important role of F/S isoform ratio concentration in relation to age at the onset of disease emerges from our investigation. The two isoforms have different biochemical properties and probably different physiological functions also. Further studies on their relationship with risk and clinical manifestations of diabetes would be rewarding.
The role of genetic factors on the age at the onset of diabetes has been scarcely considered in the literature. Associations with chromosomal areas, with ethnicity, with FTO and interleukins have been reported [10-13]. ACP1 could be a candidate gene for T2D with a significant role in the clinical manifestations of the disease including blood glycemic level, coronary artery disease [14] and age at the onset.