Psoriasis is a common inflammatory skin disease that is estimated to affect about 2-4% of the population, with rates varying across ethnic groups and geographical regions [1,2]. Biotechnological (more commonly called biologic drugs) drugs have shown great results in treating severe forms of psoriasis. Biologic agents that specifically block the IL-17 pathway have shown promising results with regard to efficacy and safety [3,4]. Specific IL-17 inhibitors are represented by Secukinumab and Ixekizumab, fully human and humanized (respectively) monoclonal antibodies against IL-17A, while Brodalumab is a fully human anti-interleukin-17 receptor A (IL-17RA) monoclonal antibody. We present a single center case series of 6 patients affected by moderate to severe psoriasis treated with brodalumab.
Anti-IL17RA, IL-17, Biologics, Brodalumab, Psoriasis, PASI100, Clinical practice, Real life
Psoriasis is a common inflammatory skin disease that is estimated to affect about 2-4% of the population, with rates varying across ethnic groups and geographical regions [1,2].
Approximately 20% of patients with psoriasis have moderate-to-severe disease and require systemic therapy to control its manifestations [5].
Medical systemic therapy for psoriasis has classically included treatments such as phototherapy, retinoids, methotrexate, and cyclosporine. However, the discovery of the immune mechanisms and alterations at the base of the pathogenesis of this disease has shifted the therapy towards targeting specific inflammatory pathways. Currently, we are experiencing a real revolution in the treatment of psoriasis thank to the introduction of biologic drugs.
Biologic agents that specifically block the IL-17 pathway have shown promising results in terms of efficacy and safety [3,4]. Specific IL-17 inhibitors are represented by Secukinumab and Ixekizumab, fully human and humanized (respectively) monoclonal antibodies against IL-17A, while Brodalumab is a fully human anti-interleukin-17 receptor A (IL-17RA) monoclonal antibody.
We present a single center case series of 6 patients affected by moderate to severe psoriasis treated with brodalumab. The results we have achieved point out brodalumab as a drug of great expectations in terms of efficacy, rapidity, long-term maintenance and safety. Even if our experience is limited by the low number of patients, we have experienced promising results that have confirmed and even exceeded clinical trial evidences.
Brodalumab 210 mg was administered subcutaneously at week 0, 1, 2, following the induction scheme of administration. After week 2, the drug was administered every 2 weeks following the maintenance scheme.
We evaluated the following scores: PASI (Psoriasis Area and Severity Index), BSA (Body Surface Area), PGA (Physician Global Assessment) and DLQI (Dermatology Life Quality Index) results at week 0, 4, 12, 24 and 52 weeks and we documented with clinical photos the development or the resolution of clinical signs of psoriasis.
We decided to treat 6 patients affected by moderate-severe psoriasis (Table 1). Patient 1, patient 3 and patient 6 had palmoplantar involvement; patient 1 and patient 6 had genital involvement. Patient 3 had nail psoriasis; patient 2, 5 and 6 had also scalp involvement. Only patient 6 was previously treated with another biological agent, since he had received a previous treatment with ixekizumab. All our patients were screened for HBV, HCV, HIV and TB before starting the treatment with brodalumab and, for each case, the results were negative or vaccinated for HBV and negative for HCV, HIV and TB.
Table 1: The demographic characteristic of the patients treated with brodalumab. View Table 1
A 28-year-old male patient presented to our attention in June 2018, suffering from severe psoriasis since when the age of 18. In the past, he had been treated with topical agents including topical corticosteroids and systemic therapies, namely oral cyclosporine and phototherapy with only temporary and partial results.
When he first came to our dermatological clinic, he presented with severe psoriasis showing severely indurated plaques extending bilaterally to the elbows, knees and buttocks. He also had genitalia and hands involvement. He reported to be an amatorial boxer and a Koebner phenomenon was evident on the dorsum and knuckles of both hands (Figure 1A).
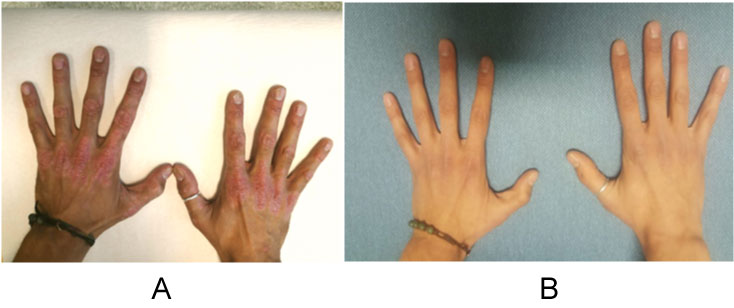 Figure 1: A) Patient 1- Week 0. B) Patient 1- Week 24.
View Figure 1
Figure 1: A) Patient 1- Week 0. B) Patient 1- Week 24.
View Figure 1
At week 0, psoriasis was evaluated as severe with the following scores: PASI 12, BSA 12%, PGA 3, DLQI 26 and it was decided to start a treatment with Brodalumab.
After the induction phase of Brodalumab, the patient was re-evaluated and the scores showed a great improvement of the disease severity: PASI 2, BSA 4%, PGA 1, DLQI 0 (week 4).
After 12 weeks of treatment the patient reached a complete clinical remission of psoriasis, with PASI 0, BSA 0%, PGA 0, DLQI 0 and the clinical condition was maintained through week 24 with a complete remission of the disease, including hands lesions (Figure 1B) (PASI 0, BSA 0%, PGA 0, DLQI 0).
The complete clinical remission was confirmed by the visit at week 52 with a total clearance of skin manifestations (PASI 0, BSA 0%, PGA 0, DLQI 0).
A 61-year-old male patient came to our attention in July 2018, with a clinical history of psoriasis for 30 years.
He was mildly obese with a BMI of 30. The psoriasis involved to the whole trunk, back, upper and lower extremities and the scalp. He had been previously treated with topical agents, phototherapy, cyclosporine, citrating and methotrexate.
During the first evaluation the clinical scores were: PASI 18, BSA 45%, PGA 4, DLQI 20 (week 0) (Figure 2A).
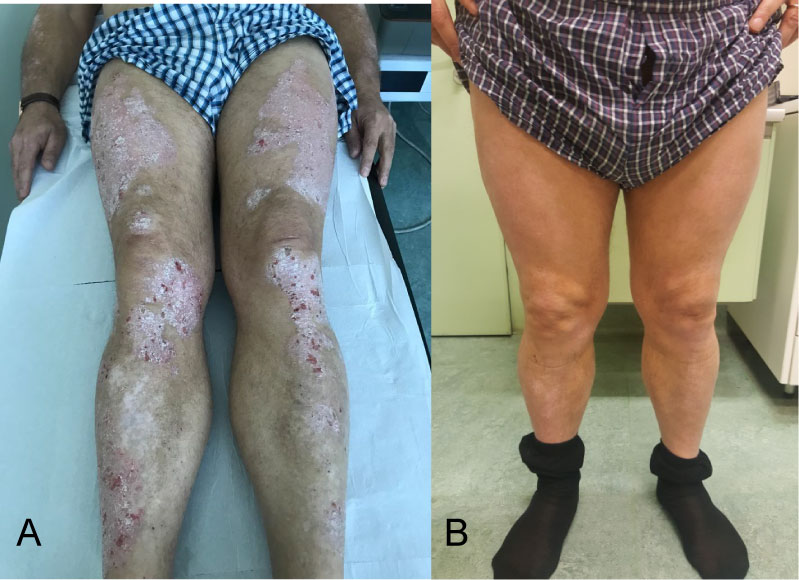 Figure 2: A) Patient 2- Clinical status at Week 0; B) Clinical status at Week 24.
View Figure 2
Figure 2: A) Patient 2- Clinical status at Week 0; B) Clinical status at Week 24.
View Figure 2
Because of the disease' severity and the many systemic therapies attempted and failed in the past, it was decided to start the treatment with brodalumab. After 4 weeks of treatment, psoriasis was significantly improved and the score results were: PASI 4, BSA 5, PGA 1 DLQI 8.
Psoriasis completely remitted after 12 weeks: PASI 2, BSA 3%, PGA 1, DLQI 1.
At week 24 the clinical assessment was PASI 1.5, BSA 2%, PGA 1, DLQI 0 (Figure 2B).
After 6 months of treatment with brodalumab the general practitioner of the patient prescribed some blood tests for other reasons and elevated levels of pancreatic enzymes were noted. In particular, he had Amylase 329 IU/L and Lipase 811 IU/L. In a precautionary way, we decided to discontinue brodalumab administration even if we didn't think that the enzymes 'alterations were related to the drug. After a month of drug suspension, PASI score was still 1.5, BSA 2%, PGA 1 and DLQI 0. The psoriasis was clinically evaluated again after 12 weeks of suspension and we reported the following scores: PASI 4, BSA 4%, PGA 2 and DLQI 1.
The increase in pancreatic enzymes persisted even after the suspension of brodalumab 210 mg. The patient was clinically evaluated by a gastroenterologist and an abdominal US and RMN were requested. The final diagnosis was benign pancreatic hyperenzymemia, a benign condition characterized by elevated pancreatic enzymes in the absence of any pancreatic findings. The patient safely restarted Brodalumab and reached PASI 100 after 8 weeks of retreatment. The complete remission was confirmed at week's 52 visit that showed a total skin clearance (PASI 0, BSA 0%, DLQI 0, PGA 0).
Patient 3 is a 50-year-old male patient affected by psoriasis since 2016. He had palmo-plantar and nails involvement, without any sign or symptoms of psoriatic arthritis. He was previously treated with topical drugs and cyclosporine without significant improvement and therefore became a candidate for the biologic therapy with brodalumab.
At the first dermatological visit, psoriasis was severe: PASI 35, BSA 18%, PGA 4, DLQI 15 (week 0), so the patient was started on brodalumab 210 mg. After 4 weeks the clinical assessment was: PASI 14, BSA 20%, PGA 3, DLQI 5.
At week 12 we recorded: PASI 4, BSA 6%, PGA 1, DLQI 2 and at week 24 the patient achieved a complete clearance with PASI 0, BSA 0%, PGA 0, DLQI 0. At week 52, there was the presence of psoriatic lesions only on the posterior surfaces of both legs and the assessment was PASI 1, BSA 2, PGA 1, DLQI 1.
A 22-years-old male patient, affected by a rapidly progressive form of psoriasis involving the trunk and the upper and lower limbs, came to our attention in August 2018. He was previously treated with topical agents, phototherapy and cyclosporine.
We decided to treat him with brodalumab and at week 0 the clinical assessment was: PASI 12, BSA 10% DLQI 10, PGA 3. After 4 weeks the patient reached the following results: PASI 3, BSA 3%, PGA 1, DLQI 4. At week 12 his psoriasis was in remission: PASI 0, BSA 0%, PGA 0, DLQI 0. The clinical assessment was maintained through week 24: PASI 0, BSA 0%, PGA 0, DLQI 0. At week 52 the assessment was PASI 1, BSA 1%, PGA 1, DLQI 0.
A 49-year-old female patient came to our attention in November 2018 with a new diagnosis of psoriasis that had flared after the death of her husband a few months earlier. The patient was affected by metabolic syndrome, type 2 diabetes mellitus, hypertension, supraventricular arrhythmia, obesity and NASH. She was already on treatment with nebivolol and metformin and, after our clinical evaluation at week 0 (PASI 25, BSA 15%, PGA 3, DLQI 18), we decided to start a therapy with brodalumab 210 mg since she had contraindications to other systemic therapies. After 4 weeks, the clinical indexes of disease severity were: PASI 6, BSA 10%, PGA 2 DLQI 1.
After 12 weeks of treatment psoriasis was particularly improved: PASI 2, BSA 1%, PGA 1, DLQI 0. After 24 weeks psoriasis was completely remitted: PASI 0, BSA 0%, PGA 0, DLQI 0. At week 52 the clinical assessment was: PASI 1,5. BSA 2%, DLQI 1, PGA 1.
A 65-year-old male patient affected by psoriasis for more than 15 years came to our attention in April 2018. He had previously been treated with cyclosporine and topical drugs without significant improvement, so we first decided to treat him with ixekizumab. Before starting ixekizumab the baseline level of disease was evaluated: PASI 14, BSA 12%, PGA 3, DLQI 14. The psoriasis was involving both hands (Figure 3), feet and the inguinal folds bilaterally. After the induction phase (160 mg at week 0, followed by 80 mg at week 2, 4, 6 8, 10, 12), ixekizumab 80 mg was administered every 4 weeks.
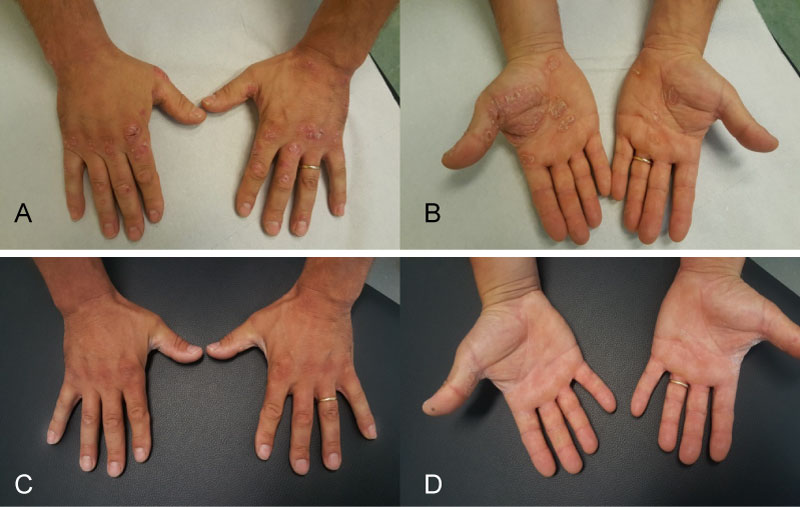 Figure 3: Psoriatic lesions of dorsal (Figure 3A) and palmar (Figure 3B) faces of both hands at first visit (week 0).
Figure 3: Psoriatic lesions of dorsal (Figure 3A) and palmar (Figure 3B) faces of both hands at first visit (week 0).
C,D) Almost complete resolution of hand's psoriasis at week 24, with residual slight desquamation.
View Figure 3
After three months of treatment with ixekizumab, we didn't see any significant improvement of psoriasis and the patient referred a disabling and generalized pruritus that had started in correspondence with the induction phase of the therapy, so we decided to switch on brodalumab 210 mg.
At week 0 the clinical situation was: PASI 12, BSA 10%, PGA 3, DLQI 14. After 4 weeks of treatment the psoriasis improved and the patient was satisfied of the clinical assessment: PASI 2, BSA 5%, PGA1, DLQI 4. After 12 weeks we assessed a further improvement of the disease: PASI 0, BSA 0%, PGA 0 and DLQI 0. At week 24 the clinical assessment was: PASI 1, PGA 1, BSA 2%, DLQI 3. At week 52, PASI was 1, PGA 1, BSA 2%, DLQI 2.
We present the results of a case series of 6 patients with severe psoriasis treated with brodalumab 210 mg s.c. brodalumab 210 mg was administered subcutaneously at week 0, 1, 2, following the induction scheme of administration. After week 2, the drug was administered every 2 weeks.
We considered a treatment period of 52 weeks.
Figure 4 and in Figure 5 report the progress of the scores PASI, BSA, PGA and DLQI of the patients through week 52. At week 0 the mean PASI was 19, with a maximum PASI of 35, and a minimum PASI of 12; the mean BSA score was 18.33%, with a minimum BSA of 10% and a maximum of 45%. The DLQI mean score was 17, 16, with a minimum of 10 and a maximum of 26.
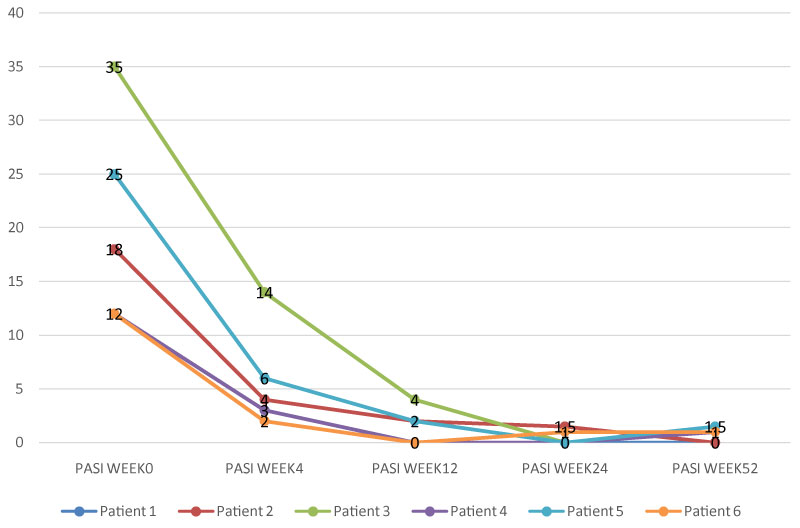 Figure 4: Six patients' PASI scores at week 0, 4, 12, 24, 52.
View Figure 4
Figure 4: Six patients' PASI scores at week 0, 4, 12, 24, 52.
View Figure 4
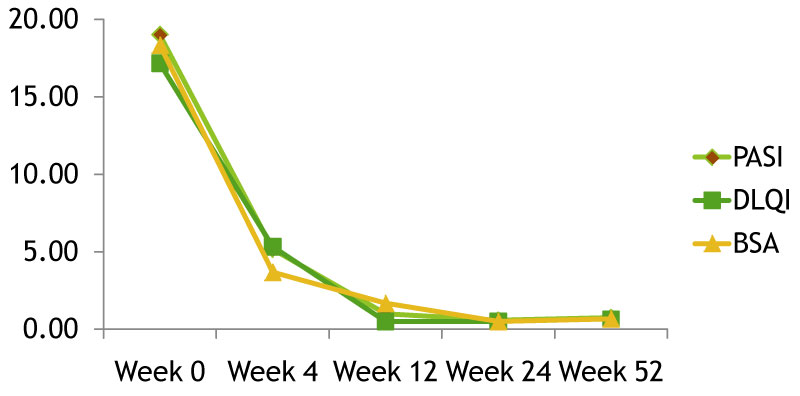 Figure 5: Mean PASI, DLQI, BSA mean score at week 0, 4, 12, 24, 52.
View Figure 5
Figure 5: Mean PASI, DLQI, BSA mean score at week 0, 4, 12, 24, 52.
View Figure 5
After 4 weeks of therapy (week 4) the mean PASI was 5.17, with a maximum value of 14 and a minimum value of 2; the BSA mean score was 7.83% with a minimum score of 3% and a maximum of 20%. DLQI mean score was 5, 33 with a minimum score of 0 and a maximum score of 8.
After 12 weeks of treatment the mean values of the scores were: PASI 0, 83 BSA 1.17% and DLQI 0.5
At week 24 the mean score of PASI was 0.17 the BSA mean score was 0.17% and the DLQI was 0.5
At week 52, the mean PASI score 0.75 the BSA mean score was 0.67 and the DLQI mean score was 0.66.
In conclusion, brodalumab 210 administered subcutaneously at week 0, 1, 2 and with a 210 mg s.c. injection every 2 weeks, showed to be, in our first Italian case series, effective, safe and well tolerated, showing a significant fast reduction of the mean values of PASI, BSA, PGA and DLQI (Figure 4 and Figure 5) without any side effect or adverse event. There were no cases of upper airways respiratory infections or mucocutaneous cadida infections. The drug injections were well tolerated and our patients did not report any side effect at the injection site.
In our case series, only patient 2 stopped brodalumab because of the elevation of amylase and lipase. However, the diagnosis of benign pancreatic hyperenzynemia should be considered an incidental finding rather than a real side effect. There are no cases of pancreatic hyperenzymemia or risk of pancreatic neoplasms associated with biologic therapies or brodalumab in the literature. Indeed the gastroenterologist concluded for a benign pancreatic hyperenzymemia that was probably already present before starting the treatment and we strongly think that brodalumab was not the cause of this incidental finding.
None of our patient reported any symptoms related to depression, suicide thoughts or anxiety even though there has been an alarming concern about the risk of suicide and depression among patients treated with brodalumab in the past. During the AMAGINE 2 clinical trial there were 3 completed suicides and 1 indeterminate case of suicide. It is very important to consider that, in the protocol of AMAGINE 2, there was no exclusion criteria regarding history of anxiety, depression or suicide behaviors. Moreover, all four patients had underlying psychiatric disorders or stressors and none of the suicides occurred during active treatment with brodalumab. While the half-life of brodalumab is approximately 10.9 days, the suicides occurred 58 days, 27 days, and 19 days after each subject's last dose of brodalumab, respectively. The FDA conducted a thorough analysis of the clinical trials and determined that there was no established drug-related risk of suicide or suicidal ideation. No causal or temporal relationship of suicide with brodalumab was found by the FDA, so brodalumab do not increase the risk of depression or suicide more than other drugs used in the treatment of psoriasis [5,6].
The reduction of the psoriasis severity scores was fast: in AMAGINE1, AMAGINE 2/3, more than 50% of patients reached PASI100 at week 12 [7,8], basing on our experience, 67% (4 out of 6 patients) of patients reached PASI 100 at week 12 (Figure 5) and 83% (5 out of 6 patients) reached PASI 90 at week 12.
PASI reduction was also durable over time, even when brodalumab was interrupted, as it happened in patient 2. In the AMAGINE1, 90% of patients treated with placebo 210 mg lost PASI 75 after 24 weeks. In our experience, patient 2 stopped brodalumab injection for more than 12 weeks but he manteined PASI 75 after of suspension. He also reached PASI 100 after 8 weeks of retreatment. In the case of our patient, abrupt brodalumab interruption did not cause a severe and fast relapse of the psoriasis. This observation is in contrast with what previously reported by other authors that observed a fast relapse of the psoriasis after the discontinuation of brodalumab [9,10]. Moreover, brodalumab, has proved to be as effective during retreatment as it is in brodalumab-naïve patients as patient 2 quickly regained PASI 100 after starting retreatment.
Furthermore, in our case series brodalumab showed a strong long-term efficacy at week 52.
Despite the low number of patients, the cases we reported in the series are overall very successful and point out brodalumab as a promising therapeutic option that will enable to reach a fast and strong response in patient with moderate to severe psoriasis.
The authors declare to not have received any funds for this article.
All the authors equally contributed to this article.