The regulation checkpoints of CD4CD25 expansion in presence and absence of transcription factor FOXP3 are critical to maintain balance between inflammatory TH17 and anti-inflammatory FOXP3CD4CD25 Treg cells. We investigated impact of potent inflammatory mediator nitric oxide NO(.) on commitment of normal spleen T cells following activation. The single cell suspension of healthy C57BL/6 mice splenic CD4 cells was stimulated by plate-bound antiCD3/antiCD28 antibodies in presence of cytokine cocktail IL2, IL6, TGFβ and anti-IL4, anti-IFN γ antibodies with and without GSNO for 72h. The results showed that incubation with GSNO reduced number of activated CD4CD25 cells. We found decreased level of CD4FOXP3 than CD4RORγt cells in presence of GSNO. The AnnexinV-propidium iodide staining of CD4CD25 cells followed by flow cytometry suggested presence of GSNO increased apoptosis of CD4+CD25+ but not CD4+CD25-cells.
CD4 cells, Nitric oxide, Apoptosis, Immune response
Impact of small bioreactive gaseous nitric oxide (NO.) is critical in induction of DNA damage, point mutation, post-translational modifications and stabilization of protein structures thus regulate cell survival versus death signaling pathways in a manner not clearly understood. Recent findings suggested cell damaging effect of thiol (S)-nitrosylation or glutathionylation in stressed endoplasmic reticulum (ER) in activated cells during inflammation [1-4]. Radical nitric oxide elevates ER stress due to endogenous glutathionylation of Cysteine residue to form misfolded proteins in endoplasmic reticulum. The inflammatory responses induce S-nitrosylation as an end-stage process and modulate NF-κBp65 signal pathways in activated cells by redox regulation [5-7]. The S-nitrosylation process are reported to inactivate sirtuin deacetylase SIRT1 and increased acetylation of p53 and NF-κBp65, thus positively regulate pro-inflammatory gene expression [8,9].
The influence of nitrosylation in T cell repertoires is unclear. In thymus, deficiency of GSNO- producing enzyme GSNOR causes apoptosis of thymocytes [10]. Recently, Niedbala, et al. demonstrated nitric oxide in the suppression of aryl hydrocarbon receptor and attenuation of functional TH17 cells [11].
We found that presence of reactive NO(.) carrier GSNO markedly decreased activated CD4CD25 cells and FOXP3CD4 cells in TH17 promoting defined cell culture of C57BL/6 mice spleen CD4 cells. Further, we found presence of GSNO induces late apoptosis of CD4+CD25+ but not CD4+CD25- cells.
The cell culture medium (RPMI 1640), Glutamax, and antibiotics (Penicillin-Streptomycin sulphate solution) were purchased from Invitrogen. The RBC lysis solution was purchased from Sigma (USA). The decomplemented fetal bovine serum (FBS) was purchased from Gibco (USA). The CD4 isolation kits were purchased from Miltenyei Biotech, USA.
Female C57Bl/6J mice (4-6 weeks old) were purchased from Jackson Laboratory. The mice were kept in the Medical University of South Carolina, Charleston animal facility center with food chow and water ad libitum.
Single cell suspension of mononuclear cells was prepared in RPMI 1640 (2% FBS) from mice spleen tissues (n = 5). The mononuclear cells were depleted from red blood cells by using red blood cell lysis buffer (Sigma). Viability of the cells was determined by trypan blue exclusion using Tc20 automated cell counter (BioRad). The viable (> 95%) spleen cells were used for isolation of CD4 cells following the instruction manual of CD4 isolation kit (Miltenyi Biotech).
The CD4 cells (> 95% viable) were distributed equally in 48 well tissue culture plates, so that each well contained 5 × 105 cells in 500 µl RPMI 1640 medium (Glutamax, Hepes and bicarbonate containing complete medium, pH 7.2) supplemented with 10% fetal (v/v) bovine serum and 100 U/ml Penicillin-100 µg/ml Streptomycine sulphate. The TH17 enrichment kit (Miltenyi Biotech) was used for enrichment of TH17 cells in CD4 cell culture. The cocktail used in the experiment is: major cytokines like, mouse recombinant IL2 (20 ng/ml); mouse IL6 (10 ng/ml); human TGFβ (5 ng/ml) along with mouse IL23 (10 ng/ml) and anti-IL4 (10 µg/ml), anti-IFNγ (10 µg/ml) antibodies for 72 hours in a CO2 incubator at 37 ℃. The cells in each well were stimulated by 1 µg/ml dose of plate bound antiCD3 antibody (BD Bioscience) and 1 µg/ml of plate bound antiCD28 antibody for a period of 72 hours.
The NO donor nitrosoglutathione (GSNO) (Mol. Weight 336.323) was purchased from CalBio Chem (USA). The one Molar fresh stock solution of GSNO was prepared in dimethyl sulfoxide (DMSO) and aliquoted in several microfuge tubes. The CD4 cells (5 × 105 cells per well) were incubated with 200 µM of GSNO in the presence of cytokine cocktail as mentioned above for 72h in a CO2 incubator. After appropriate time of incubation, all the cells were harvested and processed for Flow cytometry analysis.
The flow cytometry analysis of CD4 cells were performed following the guidelines of BD Bioscience for intracellular and extracellular staining. Briefly, the viable cells (5 × 105 cells) (the unstained cells examined by trypan blue exclusion method using BioRad Tc20 automated cell counter) were resuspended in BD stain buffer (BD Bioscience) and incubated with Fc receptor blocking antibody kit for 30 minutes at 4 ℃. Following the harvest and a brief wash, the cells were processed for extracellular cell surface staining for CD4- BUV395 and CD25-APC and/or CD25- BV786 (BD Biosciences) for 40 minutes in BD staining buffer (BD Bioscience). Afterwards, cells were harvested by centrifuging at 300 g at 4 ℃ for 5 minutes and incubated the cell pellets in BD permeabilization and fixation buffer for an hour at 4 ℃. After a brief wash using BD wash buffer, cells were incubated with different antibodies and attached with fluorochrome conjugates including mouse RORγt-PerCPCy5.5 (BD Bioscience) (1:100), mouse FOXP3-PECF-594 (BD Bioscience) (1:100) for an hour at 4 ℃ on a rocker in dark conditions. The controls like isotype antibody conjugated with fluorochrome stained cells, unstained as well as unstimulated cells were processed at the same time for flow cytometry experiments. The stained cell samples were analyzed in a flow cytometer (MoFlo) by the extent of their fluorescent intensities under normalized condition with respect to controls (the unstained cells were used to set up background control for unspecific fluorescence. The isotype controls for fluorochromes were prepared by staining unstimulated normal CD4 cells with corresponding immunoglobulin isotypes conjugated with same fluorochrome in RPMI 1640 medium (IgG1κ- BUV395 conjugate antibody). These isotype control cells were used to set up base level of fluorescent intensity for CD4 specific immune staining using antiCD4 IgG1-κ BUV conjugate antibody. The experimental fluorochrome intensities in X- and Y-axes were automatically adjusted above the base cut up value. About 20,000 cells were gated in the flow cytometer for data acquisition and analysis. The fluorochromes used were, BUV395 (UV excitable): 348/395 nm (moderate intensity); APC: 650/660 nm (bright intensity); PerCPCy5.5 (moderate intensity): 482/678 nm; PECF594: 488/578 nm (brightest intensity); Alexafluor488: 495/519 nm (moderate intensity). The filters for the fluorochromes were selected based on their excitation and emission wavelengths: UV 355 nm band pass filter (379/28) for BUV395; FL1 (533/30); FL2 (585/40); FL3 (670 LP), FL4 (675/26) and violet 405 nm band pass filter (780/60) for BV786. Three independent experiments were performed using spleen from 5 normal mice in each set of experiments. The fluorochrome intensities in cell populations were expressed as Mean ± s.d. of percentage of stained cells for each set of experiment.
The CD4 cells (1 × 106 cells per well) were incubated with plate bound antiCD3 antibody and anti CD28 antibody (1 µg/ml final concentration) in the presence and absence of GSNO (200 µM) (daily application in cell culture) for 70 hours as mentioned in cell culture section in the Materials and Methods section. The apoptotic condition of CD4CD25 cells was assayed in flow cytometry by analyzing CD4 cells stained with FITC-tagged AnnexinV apoptosis detection kit following the instruction manual supplied with the kit (Catalogue No. 556547, BD Bioscience). The PI positive CD4-BUV395 (348/395 nm wavelength) and CD25-BV786 (407/786 nm wavelength) stained cells were gated for data acquisition and analysis in a flow cytometer. Data represented as Mean ± s.d. of percentage of apoptotic CD4 cells from three experiments.
The flow cytometry results (Figure 1) stated the effect of NO(.) carrier GSNO on CD4 cells in cell culture. The 20,000 cells (CD4CD25) were gated for analysis of stained cell population in presence and absence of GSNO as described in Materials and Methods. The isotype antibodies and corresponding fluorescent conjugates were used as controls for the flow cytometry experiments (Figure 1A and Figure 1B). The fluorescent intensities over the baseline were recorded as positive for the cells. In the experiments, control double stained CD4CD25 cells were incubated with DMSO-saline in the RPMI1640 culture condition without any treatment in vitro (Figure 1C, R14). However, incubation of CD4 cells in presence of IL2 along with plate bound antiCD3/antiCD28 antibodies increased the CD4CD25 cells to 5% (Figure 1D, R14). Further, the increase in percentage of CD4 cells to 7% were also noted when cells were stimulated in presence of cytokine cocktail IL2, IL6 and TGFβ in cell culture (Figure 1E, R14). But, incubation of CD4 cells in presence of 200 µM GSNO in similar cell culture condition was found to decrease the CD4CD25 cells to 3% (Figure 1F).
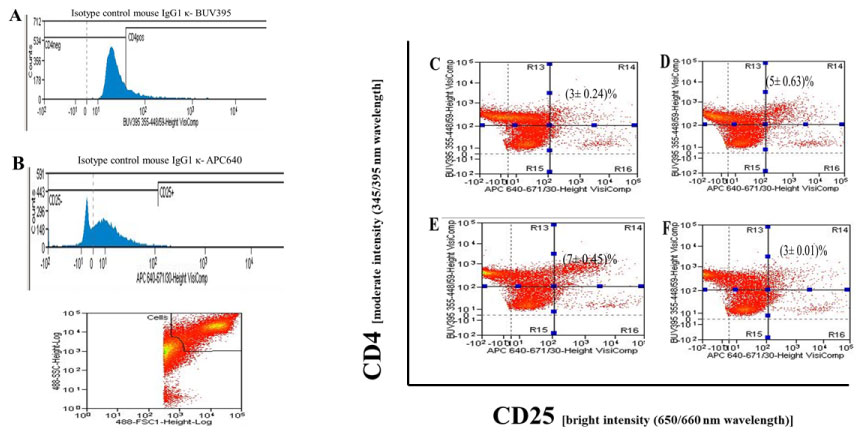 Figure 1: Nitric Oxide Carrier s-Nitrosoglutathione (GSNO) decreases CD4CD25 cells in inflammatory culture condition.
Figure 1: Nitric Oxide Carrier s-Nitrosoglutathione (GSNO) decreases CD4CD25 cells in inflammatory culture condition.
The CD4 cells were isolated from normal 4-6 weeks old female C57BL/6 mice (n = 5) and single cell suspension was prepared as described in Materials and Methods. A) The 5 × 107/ml cells were resuspended in RPMI 1640 (supplemented with GlutaMax, 10% FBS and antibiotics) and distributed in each well of 48 well tissue culture plates (5 × 106/well); B) The triplicate wells of CD4 cells were incubated for 72 h with C) DMSO saline; D) Plate bound antiCD3/antiCD28 antibodies + IL2; E) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ; F) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ + GSNO (200 µM). The isotype controls (A and B) for corresponding fluorophores were used as basal fluorescent intensity for flow cytometry analysis. The gated cells (20,000) were analyzed for data acquisition purposes. The double positive CD4CD25 cells were in right upper side R14 quadrant of flow cytometry. The results were obtained from mean ± s.d of triplicate wells from three different experiments.
View Figure 1
We wanted to determine whether in vitro treatment with GSNO has any impact on transcription regulator FOXP3 and RORγt positive mouse spleen CD4 cells.
The flow cytometry analysis for isotype controls were presented in (Figure 2A, Figure 2B and Figure 2C) showing the basal intensity of corresponding fluorochromes (PECF594, PerCPCy5.5, BUV395) used to detect specifically FOXP3, RORγt and CD4 cells under experimental conditions. The stained cells of FOXP3 (2% CD4FOXP3 cells) (Figure 2D) and RORγt (2.3% CD4RORγt cells) (Figure 2G) respectively in unstimulated control CD4 cells. The incubation of cells in presence of plate bound antiCD3/antiCD28 antibodies with cocktail cytokines IL2 + IL6 + TGFβ did not show any change in CD4FOXP3 level (Figure 2E). However, a decrease in FOXP3CD4 cells to 1% was noted in presence of reactive NO(.) carrier 200 µM GSNO in cell culture under similar condition (Figure 2F). On the other hand, level of RORγtCD4 cells showed a marginal increase (3%) in presence of plate bound antiCD3/antiCD28 antibodies with cocktail cytokines IL2 + IL6 + TGFβ (Figure 2H). Though, the presence of reactive NO(.) decreased level of RORγtCD4 cells to 2.1% (Figure 2I). The ratio of CD4FOXP3 to CD4RORγt in presence of NO(.) carrier GSNO shows overall reduction of CD4FOXP3 cells compared to control (p ≤ 0.05) (Figure 2J).
 Figure 2: Incubation with GSNO down regulated activated FOXP3CD4 but not RORγtCD4 cells.
Figure 2: Incubation with GSNO down regulated activated FOXP3CD4 but not RORγtCD4 cells.
The CD4 cells were isolated from normal 4-6 weeks old female C57BL/6 mice (n = 5). The single cell suspension (5 × 107/ml cells) were resuspended in RPMI 1640 (supplemented with GlutaMax, 10% FBS and antibiotics) distributed in each well of 48 well tissue culture plates (5 × 106/well). The triplicate wells of CD4 cells were incubated for 72h with D and G) DMSO saline; E and H) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ; F and I) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ + GSNO (200 µM) for 72h. The fluorochrome specific isotype control cells (A, B and C) Were used to determine basal level of fluorescent intensity for corresponding experimental conditions. The stained gated cells (20,000) were used for data acquisition and analysis purposes. The presence of double positive CD4FOXP3 and CD4RORγt are shown in R14 and R23 quadrant of flow cytometry analysis, respectively; J) The quantitative plot showing ratio of %CD4FOXP3 to %CD4RORγt cells in presence and absence of GSNO. The level of significance was shown in the figure with respect to control (p ≤ 0.05). The results were expressed as mean ± s.d of triplicate wells of three different experiments.
View Figure 2
The intracellular cytokine staining for detection of IL10 and IL17A positive CD4 cells were performed and the results were presented in Figure 3. An increase in %IL17A(+)CD4 cells (5.1%) (R65) were demonstrated in presence of plate bound antiCD3/antiCD28 antibodies and cocktail cytokines IL2 + IL6 + TGFβ (Figure 3B) than unstimulated control (3%) (Figure 3A). However, presence of NO(.) carrier GSNO was found to reduce IL17A(+)CD4 cells to 3.55%, comparable to control (Figure 3C). On the other hand, there was no remarkable change in IL10(+) CD4 cells (1.45%) (R68) found as compared to control (0.83%). The ratio of NO(.) carrier GSNO treated %IL10(+)CD4 to %IL17A(+)CD4 cells demonstrated only insignificant alterations as compared to control (p ≥ 0.05) (Figure 3D).
 Figure 3: Effect of GSNO on IL10+CD4 and IL17A+CD4 positive cells.
Figure 3: Effect of GSNO on IL10+CD4 and IL17A+CD4 positive cells.
The single cell suspension (5 × 107/ml) of CD4 cells (isolated from C57BL/6 mice) were resuspended in RPMI 1640 (supplemented with GlutaMax, 10% FBS and antibiotics) and distributed in each well of 48 well tissue culture plates (5 × 106/well).
The triplicate wells of CD4 cells were incubated for 72 h with A) DMSO saline; B) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6+TGFβ; C) Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6+TGFβ + GSNO (200 µM). The presence of IL10+CD4 (lower right panel R68) and IL17A+CD4 (Upper left panel R65) in stained gated cells were analyzed by flow cytometry. The results were obtained from mean ± s.d of triplicate wells from three different experiments. The quantitative bar diagram showing ratio of %IL10+CD4 to %IL17A+CD4 cells in presence and absence of GSNO and level of significance was shown with respect to control (p ≥ 0.05). The results were expressed as mean ± s.d of triplicate wells of three different experiments.
View Figure 3
We incubated the CD4 cells to grow in cell culture in presence of 200 µM GSNO along with plate bound antiCD3/antiCD28 antibodies and cocktail cytokines IL2 + IL6 + TGFβ for 70 h. The CD4 cells were then harvested and stained with CD25-BV786 to obtain CD4CD25 cells. These cells were further stained with Annexin- FITC and counterstained with propidium iodide (PI) for detection of apoptosis. The PI stained cells were gated for data acquisition and analysis by flow cytometry. The flow cytometry results presented in Figure 4 demonstrated an increase in late apoptosis (upper right quadrant) of AnnexinV-FITC and PI double stained CD4+CD25- cells following incubation with GSNO as compared with control.
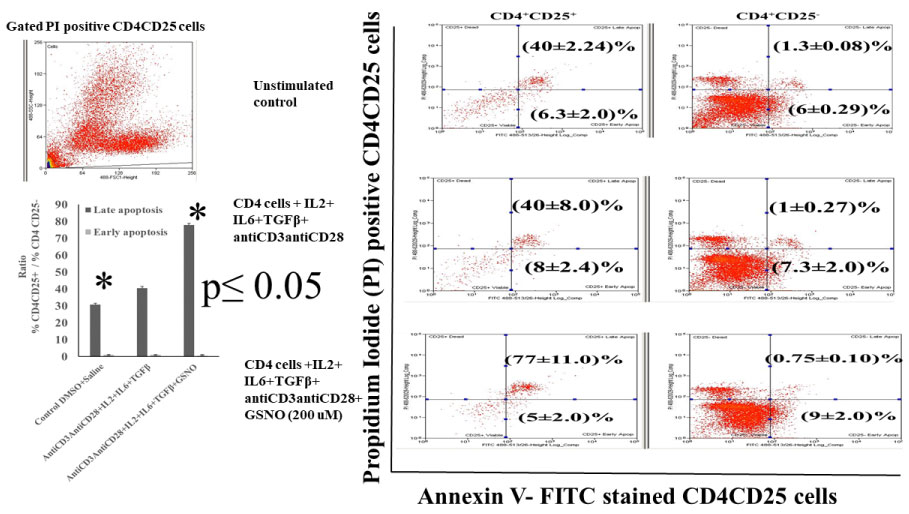 Figure 4: Incubation with GSNO induces late stage apoptosis of CD4CD25 double positive cells in presence of plate bound antiCD3/antiCD28 and IL2 + IL6 + TGFβ in culture.
Figure 4: Incubation with GSNO induces late stage apoptosis of CD4CD25 double positive cells in presence of plate bound antiCD3/antiCD28 and IL2 + IL6 + TGFβ in culture.
The single cell suspension of C57BL/6 CD4 cells (5 × 106/well) were distributed in each well of 48 well tissue culture plates. The triplicate wells of CD4 cells were incubated with DMSO saline (unstimulated control); Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ treated CD4 cells; Plate bound antiCD3/antiCD28 antibodies + IL2 + IL6 + TGFβ + GSNO (200 µM). After 70 h incubation, the CD4 cells were harvested from each well. After two brief wash with sterile PBS+ 2% FBS, cells were stained with antiCD4 and antiCD25 fluorochrome conjugated antibodies and then stained with AnnexinV- FITC and Propidium Iodide (PI) apoptosis detection staining solution following instructions of the apoptosis detection staining kit. The flow cytometry analysis was performed for PI positive CD4CD25 gated cells. The apoptosis of CD4CD25 cells in presence and absence of GSNO were analyzed in PI and AnnexinV stained CD4 CD25+ and CD4+CD25- cells. The quantitative ratios of CD4+CD25+ cells to CD4+CD25- apoptotic cells were shown with respect to control in the figure as bar diagram. The results were expressed as mean ± s.d. from three different experiments. The level of significance was shown in the figure (p ≤ 0.05).
View Figure 4
The flow cytometry analysis showed 1.3% of unstimulated normal CD4+CD25- cells were positive for PI-AnnexinV-FITC stain, the presence of cytokine cocktail and plate bound antiCD3/antiCD28 antibodies did not alter extent of late apoptosis as compared to corresponding control. Interestingly, incubation with NO(.) carrier GSNO (200 µM) in similar cell culture condition caused early (lower right quadrant) but not late apoptosis of these CD4+CD25- cells (0.75% late apoptosis : upper right quadrant). A modest increase in apoptosis was noted in the activated CD4+CD25- cells under similar conditions (6% in control to 9% in GSNO treated CD4+CD25- cells; lower right quadrants of analysis). The quantitative estimation of early and late stage apoptosis was determined by analyzing the ratios of PI- AnnexinV stained %CD4CD25+ to %CD4CD25- cells. The ratio between %CD4+CD25+ and CD4+CD25- Annexin V-PI positive cells demonstrated a significant increase during late apoptosis (bar diagram) (p ≤ 0.05) in the presence of NO(.) carrier GSNO.
Nitric oxide (NO) is a well-recognized bioactive gaseous molecule, which, in presence of super oxide radical, generates reactive nitrogen species (RNS) [12]. The long presence of RNS aggravates a wide range of diseases e.g. arthritis, systemic lupus, multiple sclerosis, and Sjogren's Syndrome [13-18]. Apart from inflammatory and tissue degenerative conditions, reactive nitric oxide is also reported to play a role in different physiological processes [19-22]. The critical role of RNS is found during thymic selection process of T cells. Thymus raises endogenous nitric oxide concentration thus initiates negative selection of T cells via apoptosis. Recent investigations demonstrated role of RNS in thymic epithelial cells by altering GSNO/GSNOR balance in maintenance of central tolerance in the thymus [10,23,24].
In the experiments, we used AnnexinV which binds with exposed apoptotic marker phosphatidyl serine. The DNA intercalating dye propidium iodide was used to counters tain apoptotic cells. Our findings showed a limitation of activated CD4 cells to commit towards CD4CD25 in presence of NO(.) in cytokine cocktail containing cell culture. We also found presence of NO(.) caused downregulation of FOXP3CD4 cells more than RORγtCD4 cells in vitro. Furthermore, we found that NO(.) carrier GSNO at 200 µM optimum concentration leads apoptosis of activated CD4+CD25+ than CD4+CD25- cells. In the cytokine cocktail, interleukin 2 (IL2) is a key cytokine to regulate expression of its receptor chain CD25 on early stage of activated T cells via ras- dependent signal pathway [25]. The other cytokines IL6, TGFβ and anti-IL4, anti-IFNγ antibodies facilitates a condition to enrich CD4RORγt cells.
Our observations demonstrated presence of both transcriptional regulators FOXP3 and RORγt in unstimulated CD4 cells. We found an increase in CD4RORγt than CD4FOXP3 cells when activated CD4 cells were incubated in presence of cytokine cocktail. However, reactive NO(.) was found to decrease CD4FOXP3 more than RORγtCD4 in ratio of CD4FOXP3 to CD4RORγt indicated a process of commitment from CD4 towards RORγtCD4 cells by eliminating FOXP3CD4CD25 cells.
The lack of direct relationship in production of IL10 and IL17A cytokines with transcription regulators FOXP3 and RORγt raises question whether activation of these transcription factors regulates expression of IL10 and IL17A cytokine genes? The activated CD4 cells in presence of IL2, IL6, TGFβ cytokine cocktail demonstrated moderately elevated level of IL17A positive CD4 cells but only minimum expression of IL10 than controls. However, the presence of NO(.) carrier GSNO was found to reduce IL17A+CD4 cells to the level of control cells with only a modest increase in IL10+CD4 level. It has been reported by different laboratories that IL17 cytokine is also produced by CD4CD25FOXP3 cells [26,27]. Thus, reduction of CD4CD25 and CD4FOXP3 cells influences overall production of IL17A under a conditioned milieu. The influence of FOXP3 on IL10 gene expression is not known. As found in our experiment, the ratio of IL10+CD4 to IL17A+CD4 (Figure 3D) in presence of NO(.) carrier GSNO showed a modest increase than the ratio of FOXP3CD4/RORγtCD4 (Figure 2J). It appears from the result that, expression of IL10 and IL17A genes has no relation on activation of FOXP3 and RORγt transcription factors. Altogether, our findings indicated that presence of reactive nitric oxide NO(.) facilitates commitment of activated CD4 towards inflammatory CD4RORγt and eliminates tolerogenic CD4CD25FOXP3 cells by apoptosis independent of expression of IL10 and IL17A.
Our observations demonstrated nitric oxide carrier S-nitrosoglutathione (GSNO) caused marked apoptosis of CD4+CD25+ but not CD4+CD25- cells following activation with plate bound antiCD3 and antiCD28 antibodies in presence of cocktail cytokines IL2, IL6, TGFβ , IL23 and anti-IL4, anti-IFNγ antibodies in conditioned cell culture. We also found a decrease in ratio of FOXP3CD4/RORγtCD4 under similar culture condition. We found low insignificant rise in the ratio of IL10CD4/IL17CD4 cells is not dependent on FOXP3 or RORγt in CD4 cells.
I acknowledge Medical University of South Carolina (MUSC) for limited resources to develop the project, "Study effect of nitric oxide carrier GSNO on T cell" (PI, SDG). The research work did not receive any specific grant from US Gov't or Non-Gov't organizations yet. I am thankful to graduate student and technicians in the Pediatrics, MUSC. I thank Jacob Kendrick, Flow Cytometry Facility Manager, Medical University of South Carolina for data acquisition and analysis.
Author have no conflict of interests.