Pregnancy and the postpartum period carry high risk of thromboembolic phenomenon which increases manifold postpartally. Multiple predisposing risk factors, usually acting in combination, add up to this risk. All three of the Virchow's triad conditions predisposing to vascular thrombosis, i.e., endothelial injury, venous stasis and hypercoagulability, are present in pregnancy. The deaths caused by pulmonary embolism may become the subject of medicolegal investigation due a sudden and unexpected fatal outcome. A thorough dissection, documentation and ancillary investigations pertaining to pulmonary emboli are important components of evaluating such fatalities. The investigation requires a dependable protocol that should include a proper evaluation of the timing of the embolus along with the residual and other thrombi, whenever discovered. Because of the pathophysiology and propagation of thrombus over a range of time period, one may see a broad histological range of thrombosis and organization. The present case pertains to the sudden death of a young female in the postpartum period who suffered a massive pulmonary embolism about 6 weeks after delivery. An extensive dissection of the lower limbs showed multiple deep venous thrombi in the calf and femoral regions while moderate to large coiled emboli were found in the pulmonary arterial vasculature. An additional histopathological dating of the thrombi as well as pulmonary embolus was performed that helped in elucidating the time range of their development.
Pulmonary embolism, Vein, Pregnancy, Puerperium, Dissection, Autopsy
Venous thromboembolism (VTE) encompasses pulmonary emboli (PE) and deep venous thrombosis (DVT), typically of the pelvic and lower-extremity veins. The incidences of pulmonary embolism and DVT are difficult to calculate and are commonly under-diagnosed [1]. The mortality rate of an acute untreated PE is 30-35% [2]. Virchow described three risk factors for thrombosis, which bear his eponym 'Virchow's Triad'. These include endothelial injury, vascular stasis, and hypercoagulability [3].
Females during pregnancy and in postpartum period are particularly at the higher risk of thromboembolic phenomenon. These risks are due to the normal or physiological changes of pregnancy to save the blood loss of parturition [4]. However, the hemodynamic disturbances may also lead to the pathological changes of Virchow's triad. Pregnancy related pulmonary embolism accounts for about 10% of maternal deaths throughout the world [5]. The incidence of pulmonary thromboembolism (PTE) is 10 times more common in the pregnant than the non-pregnant females in the same age group while the risk increases manifolds in the postpartum period which remains increased until approximately 12 weeks after delivery [5,6].
The sudden and unexpected nature of death from fatal pulmonary thromboemblism may become the issue of medico-legal concern and a forensic pathologist is then expected to perform a thorough autopsy with ancillary investigations as well as appropriate evaluation of the circumstances surrounding death [7]. In cases of sudden deaths from PTE, the necessary autopsy protocol involves: (a) Demonstration of the pulmonary emboli and a search for the residual and additional thrombi by detailed dissection, esp. of the lower limbs and pelvic region, (b) Evaluation of the probable duration of thromboembolus with the help of histological methods, (c) A sound evaluation of the underlying etiology and, (d) The manner of death, i.e., natural versus non-natural.
The present case relates to a 21-years-old female, gravida one, married since one year and a village resident who delivered her first child (male) about 6 weeks back by lower segment caesarian section. The child was healthy with a full term and uneventful delivery. The female was obese with a significant lack of ambulation or physical activity, mostly spending a lethargic routine, as narrated by the attendants. She had complaints of relapsing and remitting pain and discomfort in legs since last one week which was not given any particular attention. On one evening while she got up from the bed and went to the toilet, she had a sudden onset and escalation of symptoms in the form of abdominal discomfort, chest pain and dyspnea and was taken to the local primary health centre (PHC) where her general status was found to be poor and the case was referred to the tertiary care centre. During transportation, she became unconscious after about an hour of onset of symptoms along with a single episode of seizure and did not respond thereafter. On arrival at the referral centre, she was declared 'dead on arrival'. The dead body was brought for autopsy after about 4 hours of death.
The female was apparently obese with a body weight of 95 kg and 165 cm in length. The clothes did not reveal any abnormal stains or odor. Postmortem hypostasis was livid purplish over dorsum of the body, face and root of the neck and blanching on blunt digital pressure. Rigor mortis was well developed on head, neck and upper limbs and re-established after breaking. No external injury or any other finding suspicious of foul play was present.
Gross findings: No evidence of any internal injury was present. A suspicion of a fatal pulmonary embolism was already evident during autopsy from the history and circumstances of the case which became obvious during removal of the lungs Dissection of the each lung at the hilum lead to the extrusion of moderate to large snake-like emboli from the main pulmonary arteries that were extending into further divisions (Figure 1). A large coiled embolus was found entangled within the musculature of right ventricle, as well as in the pulmonary trunk. The emboli further revealed a tan-white granular to pink layered deposit over the dark purplish clot. This differential pattern was further evident in different layers of the sectioned clot thereby defining Lines of Zahn, confirming their true ante mortem nature.
 Figure 1: Long snake like embolus emerging out of right main pulmonary artery. Tan white to pinkish deposits over the clot may be seen (Zahn's Lines).
View Figure 1
Figure 1: Long snake like embolus emerging out of right main pulmonary artery. Tan white to pinkish deposits over the clot may be seen (Zahn's Lines).
View Figure 1
Following evisceration of thoracic, abdominal and cranial organs, the body was laid prone and lower limbs were dissected in search for deep venous thrombi. Serial sections and slight milking of each calf muscle extruded out paste-like and erect dark purplish thrombi from multiple veins while many of them were firmly adherent to the vascular walls (Figure 2). Upon proximal tracing and longitudinal opening, similar thrombi were found in femoral veins. Common iliac vein also depicted few adherent thrombi at random intervals. The viscera depicted changes of hypoxia and shock in the form of pallor and swelling with firm, pale and shiny cut surfaces.
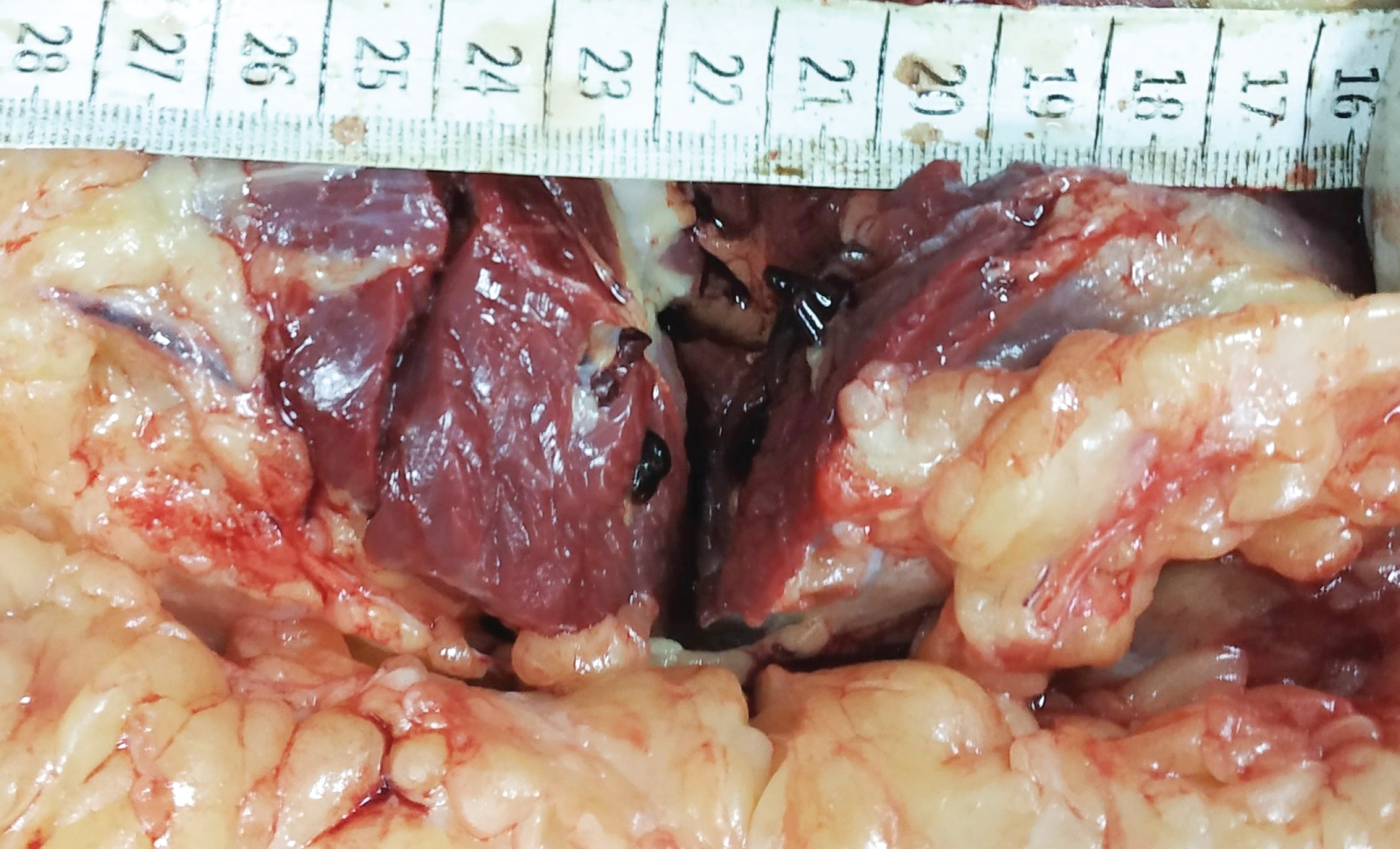 Figure 2: Sectioned right calf muscle depicting paste like and firm thrombi protruding out of the veins.
View Figure 2
Figure 2: Sectioned right calf muscle depicting paste like and firm thrombi protruding out of the veins.
View Figure 2
Sections of the calf muscles containing vascular thrombi as well as the pulmonary emboli were preserved for detailed histopathological examination including their probable durations (Figure 3).
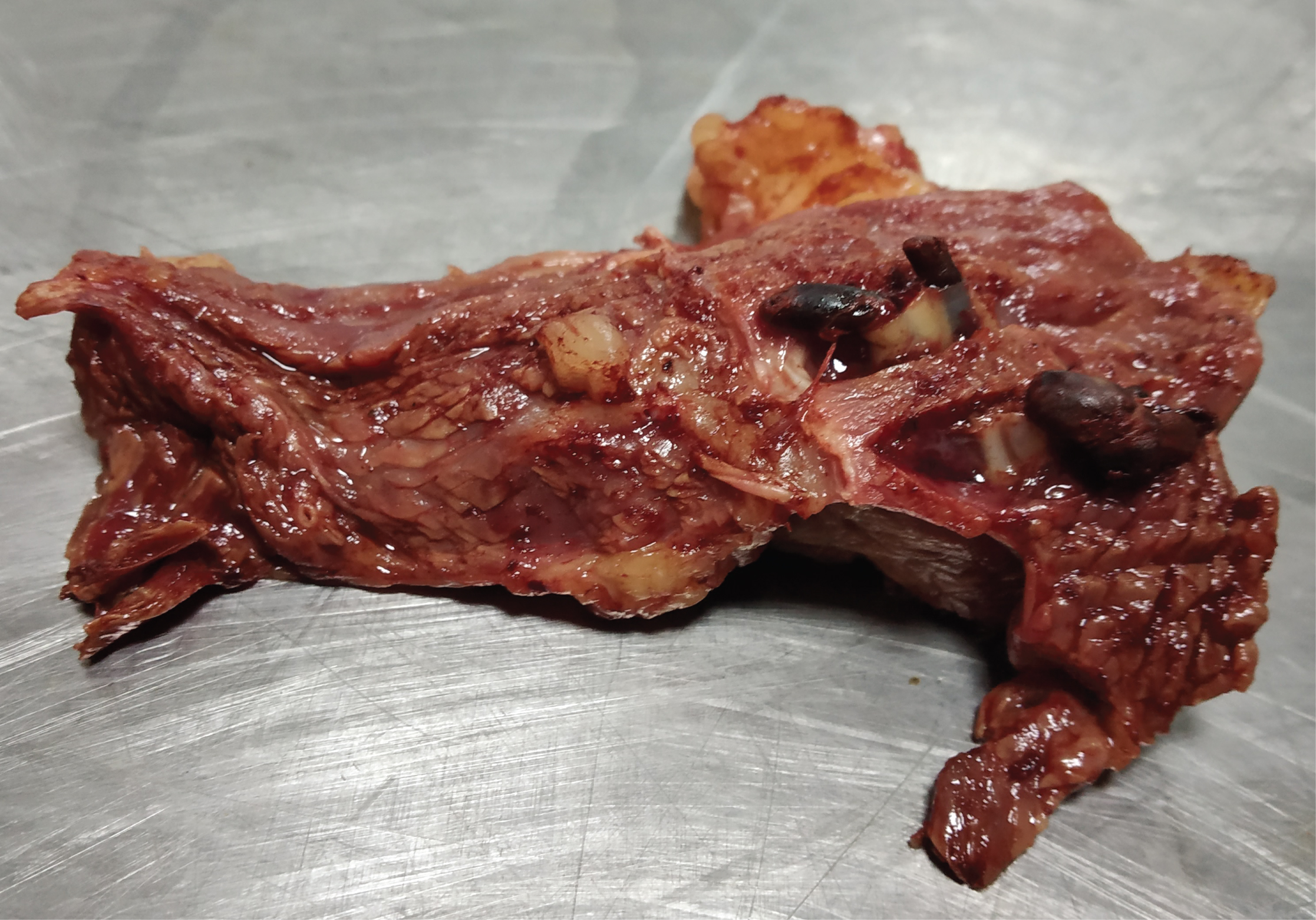 Figure 3: Calf muscle section along with contained thrombi preserved for histopathlogical evaluation.
View Figure 3
Figure 3: Calf muscle section along with contained thrombi preserved for histopathlogical evaluation.
View Figure 3
Histolopathological examination: Microscopic examination (H&E) of the thrombus's section taken from popliteal, femoral and pelvic veins resulted in different findings, varying from the acute changes of fibrin, RBCs and inflammatory cells' accumulations to peripheral organization in the form of fibrosis (Figure 4, Figure 5 and Figure 6). There was an apparent distal (popliteal) to proximal (femoral-pelvic) progression of the histological changes from an organizing to acute thrombi respectively. The pulmonary embolus revealed histological changes similar to femoral and pelvic vein thrombi, i.e., accumulation of fibrin, erythrocytes and leucocytes. Thereby, the proximally extended femoro-pelvic thrombi were the most likely source of pulmonary embolism in the present case.
 Figure 4: H & E stained section from the popliteal vein thrombus revealing organization, with RBCs located centrally while histiocytes and fibroblasts are in the periphery (100x magnification).
View Figure 4
Figure 4: H & E stained section from the popliteal vein thrombus revealing organization, with RBCs located centrally while histiocytes and fibroblasts are in the periphery (100x magnification).
View Figure 4
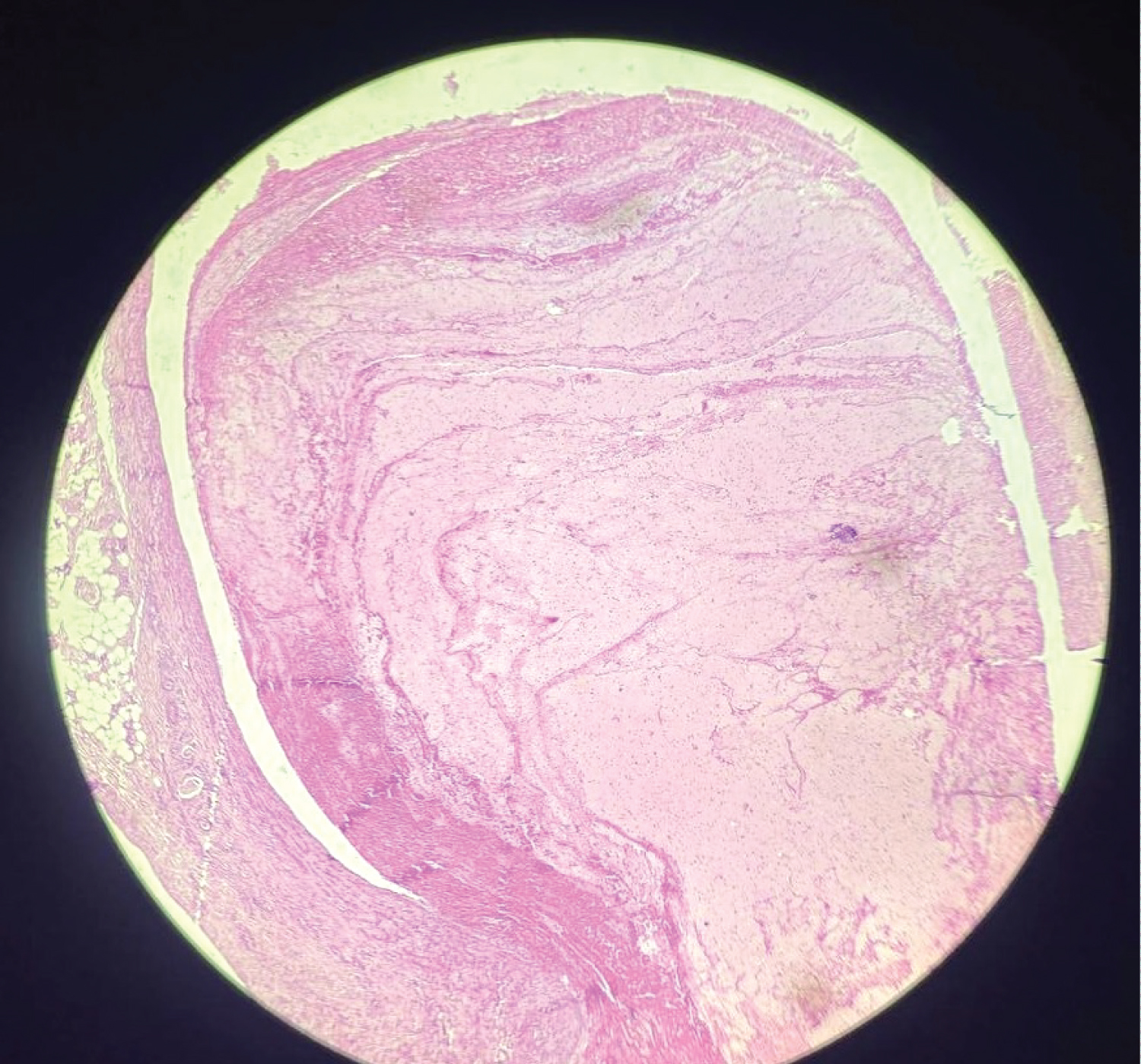 Figure 5: H & E stained section from the femoral vein thrombus with abundant fibrin, RBCs and interspersed WBCs (100x magnification).
View Figure 5
Figure 5: H & E stained section from the femoral vein thrombus with abundant fibrin, RBCs and interspersed WBCs (100x magnification).
View Figure 5
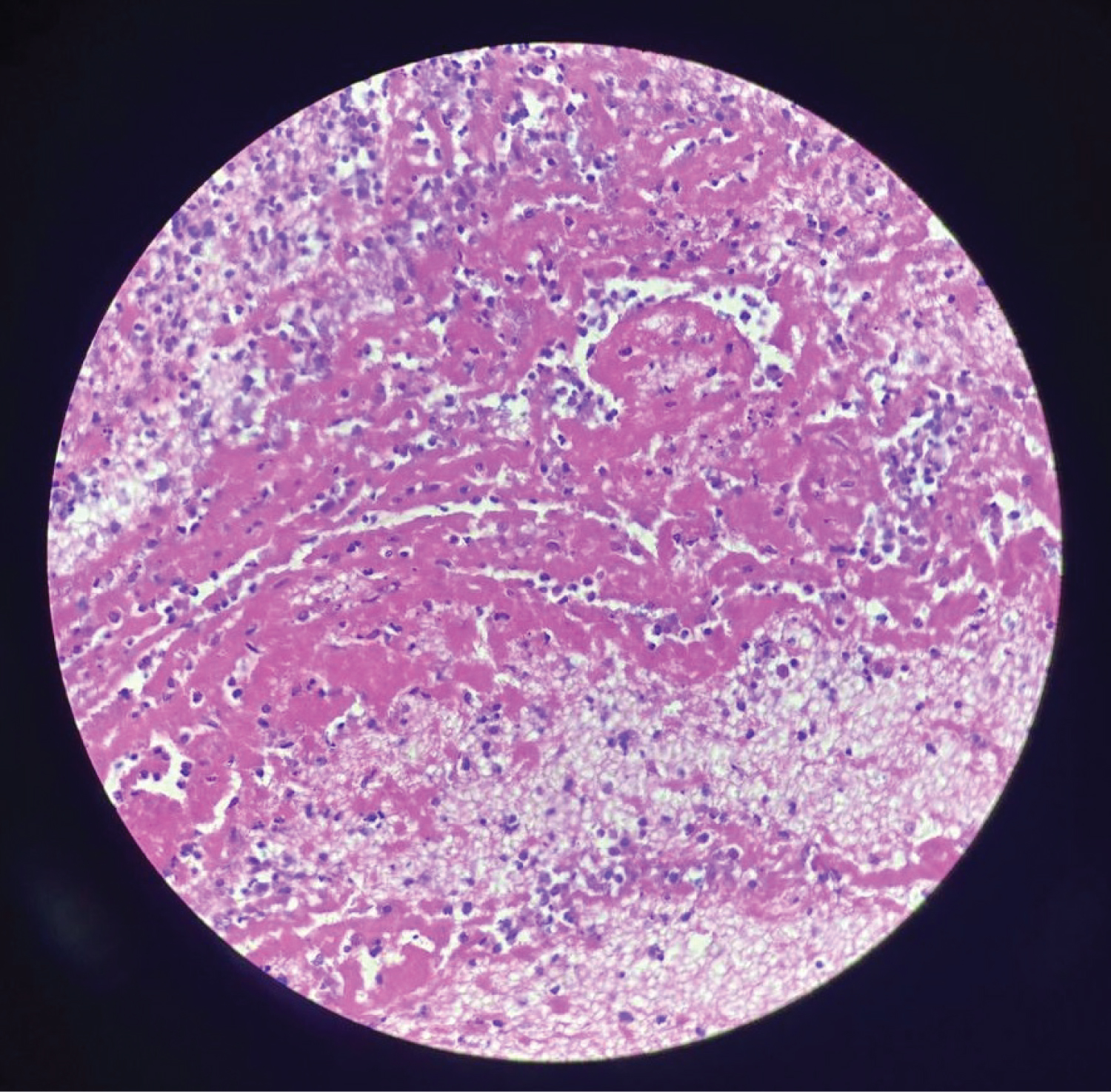 Figure 6: H & E section from the most proximal pelvic thrombus revealing fibrin and interspersed inflammatory cells (400x magnification).
View Figure 6
Figure 6: H & E section from the most proximal pelvic thrombus revealing fibrin and interspersed inflammatory cells (400x magnification).
View Figure 6
Toxicology results of viscera and blood sample were negative. The cause of death was given as acute pulmonary embolism due to deep venous thrombosis of the lower limb veins, leading to hypoxia and acute right heart failure. The manner of death was natural.
Pulmonary embolism is the most under-diagnosed cause of death where no autopsy is usually performed and it has been estimated that less than half of fatal pulmonary emboli are recognized clinically [8]. In clinical pathology, postoperative pulmonary artery embolism and embolism associated with clinically manifest leg and/or pelvic deep vein thrombosis are predominant. Pregnancy associated PE occurs suddenly and is not easy to diagnose promptly as the most common symptom of PE is dyspnea which may be the presentation of normal pregnancy [9,10]. The concurrence of typical symptoms and signs, i.e., dyspnea, chest pain and hemoptysis occurs in less than 25% of cases [11]. Forensic pathologists, however, also frequently see acute fatalities from PE without any previous symptoms of disease in seemingly healthy persons who die during routine activities. Usually, the deep veins of one or both legs still contain residual thrombi; the pelvic veins alone are rarely the origin of acutely lethal PE.
PTE is the leading cause of maternal mortality all over the world [4,12]. A woman's likelihood of venous thromboses is increased manifolds during pregnancy [5,6], due to the conditions satisfying Virchow's triad. Vascular trauma occurs to some extent during both vaginal and operative delivery. Venous stasis occurs during pregnancy because the venous system dilates, and blood flow in the leg and pelvic veins is slowed by compression by the gravid uterus [13]. This may be augmented by immobility associated with bed rest. A state of hypercoagulability is induced by increases in certain clotting factors (including fibrinogen and factors II, VII, VIII, and X) and decreases or variable changes in some components of the fibrinolytic and anticoagulant systems (factor XI and others) [14,15].
This increased coagulability is particularly significant if the woman has an underlying mutation in the prothrombin or factor V Leiden genes [16].
The risk of pulmonary artery thromboembolism is increased both in the antepartum and postpartum periods [6]. Overall, pregnancy initiates a hypercoagulable state that spans a period of approximately 10 to 11 months [13]. The state of hypercoagulation is only temporary, and usually normalizes approximately 2 months after delivery [14].
The overall risk of venous thromboembolism in pregnancy is estimated at 1:1000 to 1:2000 [17]. The risk of PTE is considerably increased in the postpartum period and is several times common following a cesarean section delivery than a vaginal delivery [17,18]. Risk factors of pregnancy associated with pulmonary embolism are history of previous surgery, preeclampsia, obesity, immobility, anemia, multiparity, age greater than 35 years and previous history of pregnancy associated pulmonary embolism [9].
A thrombus may form in the deep leg veins because of endothelial injury, stasis, and/or a hypercoagulable state [3]. There are three possible fates of the thrombus: lysis, organization with recanalization and resorption, or embolism. Before resolution, the thrombus may extend further in the veins. Proximal propagation is the most dangerous because the risk of embolism is greatest from thrombi in the inguinal/proximal leg veins [19]. Because of the pathophysiology of propagation, this proximal thrombus is the most recent and therefore least adherent to the vessel wall. In fact, treatment of a DVT with anticoagulation therapy is aimed to stop the progression of the thrombus to allow time for the body to naturally lyse or organize and resorb the thrombus [20].
Once the thrombus dislodges and travels to the lung, the embolism, depending on its size and coexisting cardiovascular diseases, interferes with hemodynamics [21]. Increased pulmonary vascular resistance will cause right heart strain or failure. Inadequate gas exchange occurs because of decreased perfusion, which results in increased dead space and ventilation-perfusion mismatches. This, combined with decreased cardiac output, leads to global hypoxia [22].
Larger emboli wedge in the main pulmonary artery, whereas smaller emboli occlude the peripheral arteries and may cause pulmonary infarction(s).
It may not be possible to always detect residual thrombus but dissection of the leg veins usually provides important information [23]. The chances of thrombus detection further depend upon the site and thoroughness of dissection and the technique(s) employed [1,7].
The vast majority of pulmonary emboli arise from the venous thrombi situated in lower extremities and pelvis; however, other locations exist as well. Indwelling central venous catheters, upper extremity veins, and the right heart have been described as possible locations of thrombi [24]. An even more rare source is from jugular vein thrombosis, sometimes seen as an extension of intracranial dural venous sinus thrombosis (commonly superior sagittal sinus) that may also occur in the females in peripartum period, causing cerebral infarction and stroke [1,4,12].
Axillary and subclavian vein thrombosis is equally unusual while the legs account for the vast majority of emboli. The origin of an embolus from the deep leg vein is usually suggested by its gross morphology, i.e., shape, diameter, and length.
The age assessment of a DVT carries special medicolegal significance. Various elaborative histological techniques, including special immunhistochemical methods, have been described to estimate the age of a thrombus [25-28]. Overall, one may determine whether a thrombus is acute, organizing, or remote. Because of the individual variability, it is often not possible to determine the precise age of a thrombus.
The histological appearance, however, can be compared and correlated with the clinical information for a corroborative evaluation. An opinion may be given on whether the age of a thrombus is consistent with a known history or not.
Once the thrombus forms, it can gradually enlarge as more fibrin-platelets are deposited and arranged in alternate layers with the erythrocytes, the so-called "Zahn's Lines" [26]. Zahn's lines are depictive of a typical antemortem embolus and its origin in an area of sufficient blood flow.
On a broader basis, the histological dating of the DVT may be summarized as follow [26]:
- During the first week, there are typical Zahn's lines characterizing platelet plug and fibrin deposition in a layered manner on the background of an eroded endothelium in the flowing blood. White blood cells and monocytes with enlarged nuclei are observed.
- During 2nd to 8th weeks, there is endothelial budding and proliferation of fibroblasts. Hemosiderophages as well as fibrinous transformation along with deposition of white cells' nuclear debris and endothelium over the surface of thrombus may be seen.
- From the 8th week, completely hyalinised thrombus with central sinuous cavities and more advanced recanalisation with fresh flowing blood are evident (older than 2 months).
Mansuto, et al. performed the following short-time ranged categorization of thrombus duration, involving evaluations at thromboendothelial junction along with a central progression (using H& E, immunohstochemistry (IHC), immunofluoroscence (IF) and additional staining for collagen/fibrosis) [28].
- Early thrombus (≤ 1 hour): Platelet aggregation with few or no erythrocytes. Presence of factor VIII and fibrinogen. Absence of lymphocyte (T cell) infiltration.
- Recent thrombus (> 1 h to 24 h): Lymphocytes (T-cells, CD3+) present. No evidence of fibroblasts and/or fibrosis.
- Recent-medium (24 h to 48 h) upto medium (48 h to 72 h) thrombus: Increase in inflammatory cells along with initial degradation with histiocytic and fibroblast cells that showed progressive recruitment. There is lysis of RBCs, WBCs and platelets with prevalent fibrin and cellular debris (homogenisation).
Fibroblast cells with fibrosis are located in peripheral vascular tissue upto third day while they are central and copious from the fourth day onwards.
- Old thrombus (˃ 72 h): Proliferation of endothelial cells, increase of fibrosis and recanalization phenomena.
Because of the pathophysiology and propagation of a thrombus, one may see a broad histological range of thrombosis and organization [7]. Histological examination of residual DVT gives the best opportunity to properly age the thrombus. The embolus typically appears recent and additionally helps in dating the initiating thrombosis.
Thorough sampling of the deep leg veins, with particular attention to the thromboendothelial junction, provides the most information. Endothelial proliferation, collagen deposition, hemosiderin, and neovascularization are common features to note. Because the maximum age of the thrombus usually is most important for medicolegal purposes, adequate and thorough tracing with sampling of the thrombi is needed.
Pulmonary embolism is the most under-diagnosed condition in most of the nations, frequently being unsuspected as the cause of death by clinicians. It is noteworthy that an originally non-lethal injury may end in death because of venous thrombosis and pulmonary embolism, converting what might be a simple accident or a common assault into a grave legal issue [1].
Victims of trauma-in-general: The victims of many forms of trauma are at risk from pulmonary embolism primarily due to factors such as injury-responsive hypercoagulability of blood, local venous thrombosis in the contused soft tissues and around fractured bones (esp. in the leg and pelvic regions) and the effects of general shock and debility that immobilize and confine the patients to the bed for an invariable period of time.
The various aspects of pulmonary embolism have been investigated in several retrospective autopsy surveys with somewhat different results depending on the era and the source of material [8,23]. Some studies have highlighted a complete lack of recognition and deficient autopsy techniques to ascertain pulmonary embolism [23]. In cases of trauma and immobility, the peak incidence of PE has been noticed after 1 or 2 weeks [1,29]. Most surgical teaching is that a DVT starts "on the table." The use of preoperative, operative, and postoperative prophylaxis usually is dictated by individual patient risk factors and the type and anticipated length of the surgical procedure [2].
Peripartum PTE: Pregnancy is accompanied by unique physiologic changes geared toward accommodating and delivering the growing fetus, followed by recovery and a return to the pregravid state. In most instances, the body is able to adapt to these changes, and the pregnancy from conception through the postpartum period is without major medical complications. However, severe life-threatening pregnancy-related complications do arise that may lead to death of the mother. In the postpartum period/puerperium, the chances of PTE are extremely high during first 6 weeks while in general increase until approximately 12 weeks after delivery [5,6,30].
Sudden maternal death due to PTE may rarely present as a suspicious death. In dealing with such a death, the pathologist should also keep in mind that the woman or her associates might not even have been aware that she was pregnant, as the incidence of DVT in general spans throughout the pregnancy [12,31]. As such, the pathologist may be the first doctor to become aware of the pregnancy.
The timing of VTE may have important medicolegal consequences. DVT may form in hours, and the resultant embolism often occurs after a sudden onset of movement after prolonged inactivity.
Both early diagnosis and treatment of VTE are crucial in order to prevent maternal deaths during the puerperal period. Due to lack of an appropriate and timely diagnosis, undetermined numbers of deaths occur each year because of thromboembolic events. A significant obstructive embolus may prove fatal within a very short period of time. Once a forensic pathologist is involved, a careful autopsy involving through dissection of the leg and pelvic veins along with careful evisceration of organs, evaluation of the probable duration(s) of deep venous thrombi and the pulmonary embolus, the manner of death and a possible exclusion of any medical malpractice must be made. Depending upon the particulars of an investigation, one may offer an opinion of the age and the possible etiology of a thrombus. However, it becomes more difficult to find or establish an initiating link when the interval increases beyond several weeks. Because of the medical, legal, and familial issues, deaths caused by pulmonary emboli need to be properly investigated and documented.
None.
Nil.
All the authors declare that they do not have any possible conflicts of interest.