Introduction: IgD myelomas constitute about 1% of myelomas, have worse prognosis, have higher association with lambda light chains, and are associated with excess free involved light chains. Standard immunofixation protocols stain only for IgG, IgM and IgA heavy chains. Proper attribution of a monoclonal gammopathy as having a delta heavy chain requires additional testing that is not available in most clinical laboratories. Retrospective review of protein electrophoresis and other laboratory results showed characteristic findings in IgD myelomas.
Methods: Retrospective review of monoclonal gammopathies at one institution revealed four cases of IgD multiple myeloma. Serum protein and immunofixation electrophoreses, and other laboratory and clinical findings were reviewed.
Results: All four cases revealed multiple bands of monoclonal immunoglobulins. The multiple bands were due to excess free monoclonal light chains producing ancillary bands in addition to the intact monoclonal immunoglobulin. Monoclonal bands were preferentially in anodic locations. All multiple myeloma cases met criteria for light chain predominant multiple myeloma. Circulating plasma cells were noted in all IgD myelomas.
Discussion: Due to the difficulties in properly diagnosing IgD myelomas, the true incidence may be higher than reported. All IgD monoclonal gammopathies examined in this study have a characteristic pattern of multiple bands on protein electrophoresis, preferentially in anodic location, along with excess free involved light chains, predominant association with lambda light chains, presence of circulating plasma cell and suppression of IgA, IgG, and IgM immunoglobulins allow a presumptive diagnosis of IgD myeloma. Worse prognosis in IgD MM warrants a reevaluation of treatment and an improved algorithm for detection.
Multiple myeloma, Light chain predominant multiple myeloma, Diagnostic criteria for IgD myeloma, Multiple bands in IgD myeloma, Plasma cell myeloma, eGFR, Survival
Neoplastic disorders of plasma cells are the commonest hematological malignancy in adults, with the exception of heterogeneous group of non-Hodgkin lymphomas [1,2]. In increasing order of severity/malignancy, the neoplastic lesions of plasma cells consist of monoclonal gammopathy of undetermined significance (MGUS), smoldering multiple myeloma (SMM) and the malignant disorder, multiple myeloma/plasma cell myeloma (MM) [3,4]. About 83-85% of multiple myeloma plasma cells produce and secrete intact immunoglobulin, about 13 to 15% secrete light chains only, while 2-3% of plasma cell dyscrasias consist of bi-clonal, tri-clonal or non-secretory immunoglobulins [5]. IgD producing multiple myelomas constitute about 1% of all myelomas and are often difficult to diagnose, in part due to lower levels of monoclonal IgD as compared to monoclonal immunoglobulin in IgG and IgA myelomas [6]. In 60-90% of IgD myelomas the associated light chain is of lambda type, in contrast to the 2:1 ratio of kappa and lambda light chins in IgG and IgA myelomas [6,7]. IgD myelomas usually produce a far greater excess of free homogenous light chains than is usual with IgG and IgA myelomas. The excess free light chain production may be misdiagnosed as biclonal multiple myeloma producing IgD and light chains [8-11].
Diagnosis of IgD multiple myeloma, in addition to the usual criteria for multiple myeloma, requires verification of the monoclonal immunoglobulin being IgD. Usual immunofixation electrophoresis only tests for IgG, IgA and IgM. Thus, immunofixation electrophoresis and/or quantification for IgD are usually employed to confirm the diagnosis, generally by sending the serum specimen to a reference laboratory.
Due to the difficulty in accurately diagnosing IgD myelomas, the true incidence of these cases may be higher than reported. It is conceivable that there are patients who are incorrectly diagnosed with light-chain myeloma due to the lack of available laboratory assays for IgD immunoglobulins in most clinical laboratories. Ordering IgD immunofixation or IgD quantification requires an appropriate level of clinical suspicion by the pathologist and/or clinician. A retrospective review of laboratory findings revealed characteristic findings that may allow a presumptive diagnosis of IgD myeloma and prompt additional confirmatory laboratory testing. A critical appraisal of criteria for presumptive diagnosis of IgD multiple myeloma is presented in this report.
This investigation was carried out at 480-bed tertiary care, medical school affiliated medical center in South-Eastern United States. The hospital, in addition to usual tertiary care oncology services provides stem cell transplantation for hematological malignancies. The study protocol was reviewed and approved by the Augusta University institutional review board (Protocol # 657783). Being that this was a retrospective review of medical records, the IRB waived the requirement for informed consent. This study is a continuation of a report on light chain predominant multiple myelomas and light chain myelomas, and some the patients addressed in this report were included in the earlier reports [8-10].
Briefly; all patients who had a serum protein electrophoresis (SPEP) and/or serum immunofixation electrophoresis (SIFE) since year 2000 were reviewed. Among the more than 600 cases of multiple myeloma, four cases were identified that met criteria for IgD multiple myeloma. The diagnoses were based on SPEP, SIFE, serum free light chain (SFLC) measurement; bone marrow examination by morphology, flow cytometry, cytogenetics, bone x-ray survey, and routine laboratory tests. All of the patients demonstrated monoclonal IgD and exhibited CRAB criteria (Hypercalcemia, renal disease, anemia and lytic bone lesions) or components of CRAB criteria.
SPEP, SIFE and SFLC quantification were carried out as described earlier [5,8,12-14]. Briefly SPEP and SIFE were carried out on Helena SPIFE Touch instrument. (Helena Laboratories, Beaumont, TX) SIFE was performed with antisera obtained from Helena and by following the protocol recommended by the manufacturer. SIFE was also carried out after reducing the serum with DTT and staining with antiserum to free lambda light chains in one patient according to a protocol reported earlier [14]. Antiserum to IgD was also procured from Helena. Serum levels of IgD were obtained from ARUP Laboratories (Salt Lake City, UT), a reference laboratory. SIFE with antiserum to IgD was not usually employed, but was carried out retrospectively in two patients on whom sera were available. In these two patients FLC-Modified SIFE was also carried out to ascertain the presence of free monoclonal light chains according to the method described earlier [15]. Antisera to kappa and lambda free light chains were procured from Sebia Inc. (Norcross, GA). SFLC were measured with kits and Optilite analyzer procured from The Binding Site (Birmingham, UK).
Peripheral blood smears were examined as a routine in the evaluation of MM patients and percentage of plasma cells as a proportion of nucleated cells were recorded. Additional data reported in Table 1 were extracted from the medical records as was described for light chain predominant multiple myelomas (LCPMM) [8-10]. Briefly, eGFR was calculated by the CKD-epi equation from serum creatinine values; survival in months was estimated from the time of diagnosis to death or last known contact with the patient [8-10].
Length of survival for IgD MM was compared to similar data for LCPMM by T-test using one tailed analysis and assumption of unequal variance. Since survival in IgD MM was significantly shorter than that for LCPMM, a comparison against usual MM, with longer survival, was not conducted.
Review of data from January 2000 to August 2020 revealed multiple myeloma cases of various types in the following numbers: IgG K -246, IgG L -123, IgA K -77, IgA L -49, Kappa light chain - 57, Lambda light chain 41, Bi- and Tri-clonal -21, Non-secretory 6, IgD - 4 for a total of 624 cases.
SPEP and especially SIFE revealed multiple "monoclonal" bands in all IgD MM patients (Figure 1). The SIFE gels show the staining pattern with antiserum to the involved light chain. All of the IgD Lambda lesions showed one or more bands in the alpha and/or beta region. The prominent band in the IgD kappa myeloma (Figure 1, Lane 4) was in the anodal gamma region. The multiple bands represented an intact IgD and excess free light chains. The excess free monoclonal light chains migrated anodal to the intact immunoglobulin IgD bands. Representative results of staining for IgG, IgA, IgM, IgD, kappa and lambda light chains and free kappa and free lambda light chains for patient 1 are shown in Figure 2. The lane labelled DTT represents serum reduced with DTT and stained for free lambda light chains. The two bands of lambda light chains seen in unreduced samples converted to a single band revealing that the darker, anodal lambda chain band consisted of dimeric monoclonal lambda chains.
 Figure 1: SPEP and SIFE patterns for patients with IgD MM, are shown. The top row represents SPEP in the four patients. The bottom row displays staining in SIFE with Helena antiserum to the involved light chain, namely anti-lambda in the first three and anti-kappa in the fourth case. All patients have at least two bands, corresponding to IgD and free light chain bands. In lane 3 no band is noted on SPEP as the IgD band overlaps transferrin band and the light chain bands noted on SIFE are at too low a concentration to be detectable on SPEP for the specimen presented in the figure. The highest level of free lambda light chains was in the range of LCPMM (Specimen with the highest light chain concentration was not submitted for SPEP, SIFE for patient 3). The IgD bands in the first three cases were associated with lambda light chains and are in the alpha and beta regions. The IgD kappa band is in anodal gamma region, in the 4th case, though the kappa chain band is still anodal to the faint IgD kappa band discernible on SIFE.
View Figure 1
Figure 1: SPEP and SIFE patterns for patients with IgD MM, are shown. The top row represents SPEP in the four patients. The bottom row displays staining in SIFE with Helena antiserum to the involved light chain, namely anti-lambda in the first three and anti-kappa in the fourth case. All patients have at least two bands, corresponding to IgD and free light chain bands. In lane 3 no band is noted on SPEP as the IgD band overlaps transferrin band and the light chain bands noted on SIFE are at too low a concentration to be detectable on SPEP for the specimen presented in the figure. The highest level of free lambda light chains was in the range of LCPMM (Specimen with the highest light chain concentration was not submitted for SPEP, SIFE for patient 3). The IgD bands in the first three cases were associated with lambda light chains and are in the alpha and beta regions. The IgD kappa band is in anodal gamma region, in the 4th case, though the kappa chain band is still anodal to the faint IgD kappa band discernible on SIFE.
View Figure 1
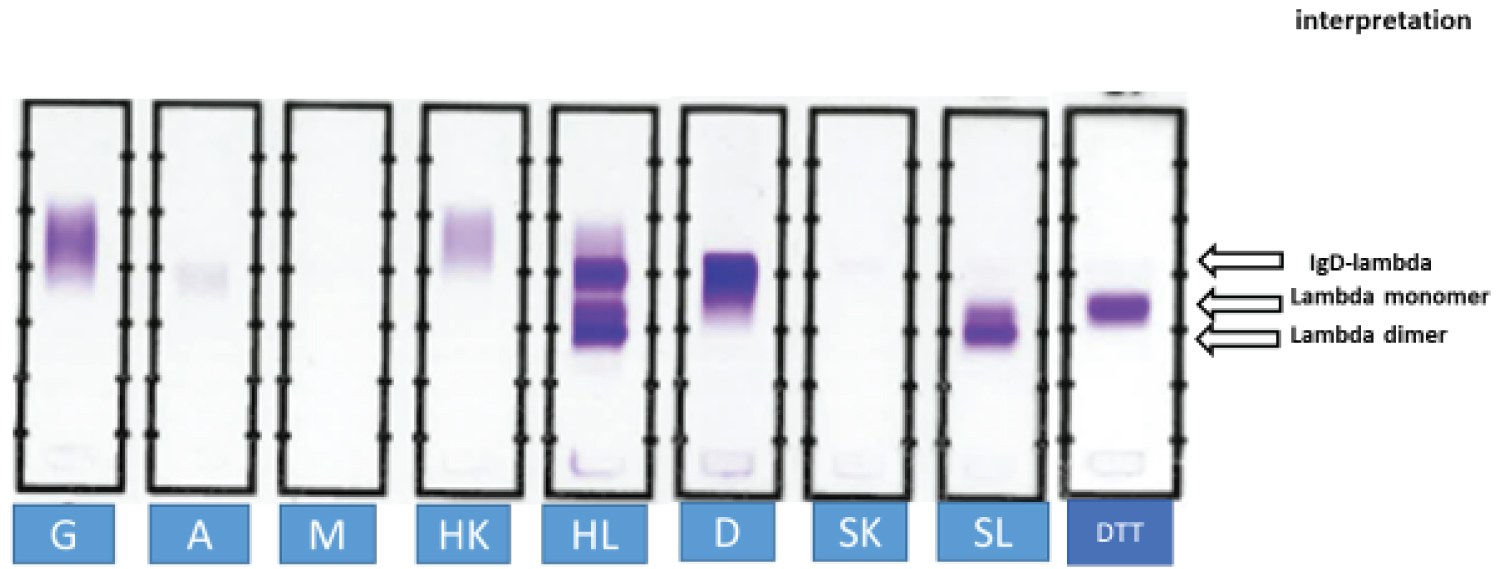 Figure 2: Stored serum from patient 1 was subjected to SIFE. Antisera against free kappa and lambda light chains were procured from Sebia Inc. (SK, SL). All other antisera were obtained from Helena Laboratories. G: anti IgG; A: anti IgA; M: anti IgM; HK: antiserum to kappa light chains; HL: antiserum to lambda light chains; D: antiserum to IgD. The antisera stained the IgD band in anodal gamma region and the two lambda light chain bands in beta and alpha regions. The identity of the cathodal band in lane HL was confirmed to be monoclonal IgD by staining for antiserum to delta chains, lane D. No staining was noted in SK. SL revealed the two bands of free monoclonal lambda light chains. Reduction of serum with DTT (DTT) converted the two lambda light chain bands into one band of monomeric light chains. The dominant light chain band consisted of dimeric lambda light chains (lower band in lanes HL and SL).
View Figure 2
Figure 2: Stored serum from patient 1 was subjected to SIFE. Antisera against free kappa and lambda light chains were procured from Sebia Inc. (SK, SL). All other antisera were obtained from Helena Laboratories. G: anti IgG; A: anti IgA; M: anti IgM; HK: antiserum to kappa light chains; HL: antiserum to lambda light chains; D: antiserum to IgD. The antisera stained the IgD band in anodal gamma region and the two lambda light chain bands in beta and alpha regions. The identity of the cathodal band in lane HL was confirmed to be monoclonal IgD by staining for antiserum to delta chains, lane D. No staining was noted in SK. SL revealed the two bands of free monoclonal lambda light chains. Reduction of serum with DTT (DTT) converted the two lambda light chain bands into one band of monomeric light chains. The dominant light chain band consisted of dimeric lambda light chains (lower band in lanes HL and SL).
View Figure 2
As reported previously, all IgD myelomas displayed high enough concentrations of serum free light chains to be classified as light chain predominant multiple myelomas (LCPMM) [8,9]. Serum free light chain concentrations, IgD concentrations, and other findings are presented in Table 1. It should be noted that patient no. 3 presented with IgD Lambda SMM and was treated for SMM, though the patient progressed to plasma cell leukemia. As expected, the concentrations of IgA, IgG and IgM were suppressed in all IgD MM patients.
Table 1: Laboratory findings and clinical information for four patients with IgD multiple myeloma: All IgD MM patients had high levels for SFLCs, and circulating plasma cells. Only the highest levels of free light chains are recorded in the table. All patients had multiple "monoclonal" bands, including patient 3 in whom IgD band overlapped transferrin band in beta region. The patient numbers are the same as in Figure 1. View Table 1
An additional noteworthy finding was the presence of circulating plasma cells in all IgD MM patients. Only patient 3 reached circulating plasma cells high enough to be called plasma cell leukemia. The IgD band in patient 3 overlapped the transferrin band and monoclonal lambda light chain band was in the beta region.
The average length of survival from the time of diagnosis was 22 months. Even when the one outlier, patient 1, with only one month's survival was excluded, the average length of survival of 29 months was still markedly shorter than that noted for IgG and IgA- intact immunoglobulin-producing light chain predominant multiple myelomas. The average survival in LCPMM was 43 months and the corresponding figure for conventional intact immunoglobulin producing MM was 65 months [8,9]. The average survival for IgD MM at 22 months was significantly lower than that for even LCPMM (P = 0.04), despite the low number for IgD MM cases.
Patients with IgD MM tend to have extra-osseous lesions more frequently, are predominantly associated with lambda light chains, have organomegaly and amyloid more frequently and tend to have worse outcomes [6,7,16-18]. IgD myelomas constitute about 1% of all myelomas. IgE myelomas are the rarest type with < 50 reported cases [18]. Thus in patients with monoclonal gammopathy and no detectable heavy chain on SIFE, the possibilities include, in order of frequency, light chain only myeloma or IgD or IgE myeloma. It is proposed that a presumptive diagnosis of IgD myeloma could be made if the following characteristics are noted:
1. Multiple bands on SPEP, and SIFE with usual staining for GAM, kappa and lambda. SPEP band representing intact immunoglobulin, in the beta or anodal gamma region.
2. One or more bands in the alpha or beta region or anodal gamma region on SIFE representing monoclonal light chains.
3. High enough concentration of serum free light chains to meet criteria for LCPMM [8,9].
4. Circulating plasma cells.
5. Lambda light chain as the involved light chain.
6. Suppressed levels of IgG, IgA and IgM.
It is by no means implied that confirmation by quantification of IgD or SIFE using antiserum to delta heavy chain is not needed.
The limitations of this study include the small sample size from a single institution and the retrospective observational design of the study. However, the remarkably consistent findings of multiple bands on SPEP/SIFE, especially bands being present in beta and alpha region, high enough concentration of SFLC for the lesion to qualify as LCPMM, and presence of circulating plasma cells allow for the criteria listed above to be used for a presumptive diagnosis of IgD MM. The small sample size reflects the low incidence of IgD MM.
It is worth reiterating that the presence of very high serum free light chains in patients with IgD myeloma does not warrant a diagnosis of biclonal myeloma [8,9]. The short survival in IgD MM is likely to be due, in part, to excess free monoclonal light chains causing light chain nephropathy and renal failure. All four patients had lower than normal estimated glomerular filtration rate (eGFR) and three of the four had marked reduction in eGFR. The markedly shorter survival, even shorter than that for IgG and IgA LCPMM, warrants a reevaluation of the treatments in current use. As suggested for usual LCPMM, aggressive efforts at reducing the concentration of free monoclonal light chains may be useful for preventing light chain nephropathy and renal disease [10,15].
While standard hospital clinical laboratories lack the ability to definitively provide evidence of IgD monoclonality, a presumptive diagnosis of IgD MM can be made on the basis of a constellation of laboratory findings: Multiple bands on SPEP and SIFE, especially bands anodal to the gamma region, presence of circulating plasma cells, high concentration of free light chains, and suppression of IgG, IgA and IgM. IgD MM has poor prognosis with survival even shorter than that for LCPMM. The need for reevaluation of treatment for LCPMM is further highlighted by the short survival in IgD MM.
No funding was received for this research.
The author serves as a consultant to Diazyme Inc. Sebia Inc. and Health Tap, however, this did not pose any conflict of interest in this research.
The study protocol was reviewed and approved by the Augusta University institutional review board (Protocol # 657783). Being that this is a retrospective review of medical records, the IRB waived the requirement for informed consent.