Objective: To determine the expression of IMP3 (Insulin-like growth factor-II mRNA binding protein 3), which has been reported to be associated with poor prognosis in many organ malignancies, in thyroid malignant tumors and to determine its relationship with prognostic factors such as age, gender, tumor size, and histological type of tumor.
Materials and methods: The study included 60 cases diagnosed with thyroid carcinoma in the pathology department of Haseki Hospital. As the control group, two follicular adenomas, three Hurthle cell adenomas, and five nodular hyperplasia cases were included in the study. Immunohistochemistry was performed using the IMP3 polyclonal antibody.
Results: 50 cases were diagnosed with papillary thyroid carcinoma, five cases with medullary carcinoma, one case with anaplastic carcinoma, and four cases with well-differentiated tumors with uncertain malignant potential.
When IMP3 staining results were evaluated, 48 (80%) of 60 thyroid carcinoma cases had positive staining with IMP3. Of the 48 cases with positive staining, 26 (43.3%) had 1+, 16 (26.7%) 2+, 6 (10%) 3+ stainings. Positive staining was present in 7 (70%) cases in the control group. No 3+ staining cases were detected in the control group.
Conclusion: When the prognostic parameters were evaluated, no statistically significant correlation was found between IMP3 expression and age, gender, tumor size, tumor histological type, vessel invasion, lymph node metastasis, multifocality/multicentricity. All of the cases whose IMP3 expression was evaluated as 3+ were papillary thyroid carcinoma.
Thyroid carcinoma, IMP3, Prognosis
Thyroid carcinomas constitute 1% of all carcinomas in the world and are the most common malignancy of the endocrine system [1]. Due to the similarities between benign and malignant thyroid lesions and the subjective criteria, there may be differences in the evaluation between pathologists. Although immunohistochemistry markers such as HBME-1, Galectin-3, CEA, calcitonin, thyroglobulin, and TTF-1 are currently used to support the diagnosis, the need for ideal markers still continues [1-4]. IMP3 (Insulin-like growth factor-II mRNA binding protein 3) affects cell proliferation by regulating the transcription of insulin-like growth factor-II and plays an important role in RNA stabilization, cell growth, and migration during embryogenesis [5-7]. While Imp3 expression is not expected in normal tissues, it has been positively detected in malignant tumors of organs such as the colon and bladder [7]. It is associated with a poor prognosis in many organ malignancies, and IMP3 expression is high in metastases [8,9]. Our aim with this study is to determine the expression of IMP3 in benign and malignant thyroid tumors diagnosed in our clinic and to determine its relationship with prognostic factors such as age, gender, tumor size, histological type of tumor, vessel invasion, thyroid capsule invasion, surgical margin involvement, and lymph node metastasis.
Two pathologists evaluated 464 cases who underwent thyroidectomy or lobectomy in the pathology department of Haseki Training and Research Hospital between January 2011 and June 2012, and 60 cases diagnosed with thyroid carcinoma were included in the study. Hematoxylin-Eosin (HE) stained preparations of the patients included in the study were retrospectively analyzed and re-evaluated for histological type and histological subtype, thyroid tissue around the tumor, tumor capsule invasion, vessel invasion, thyroid capsule invasion, surgical margin, soft tissue invasion around the thyroid, perineural invasion, presence of calcification and necrosis. The cases were divided into two groups according to age as ≤ 45 and > 45. According to tumor size, the cases were divided into three groups as ≤ 1 cm, 1-4 cm and > 4 cm. Tumors were examined under 4 main groups according to their histological type: Papillary carcinoma, medullary carcinoma, anaplastic carcinoma, and well-differentiated tumor with uncertain malignant potential. Papillary carcinoma was divided into six groups according to histological subtype as classical type, follicular type, microcarcinoma, macrofollicular type, Warthin-like type and oncocytic type.
IMP3 polyclonal antibody (dilution: 1/100, rabbit polyclonal antibody, Code: GTX115459, Lot No. 40674, Genetex, USA) was used for immunohistochemical staining. Immunohistochemical staining was performed in a humidified, wet environment with a temperature up to 24 °C. As a result of immunohistochemical staining, cytoplasmic expressions of IMP3 were evaluated in the study and control groups. When evaluating the staining intensity; 0 negative; 1+ as weak positive; 2+ as moderately strong positive and 3+ was rated as highly strong positive.
Statistical analysis of the obtained data was performed by using SPSS 16.0. Data in the study were presented as mean value and percentage. Prognostic parameters and expression of IMP3 were compared using chi-square test, One-Way ANOVA test and Independent T-test.
The mean age of 60 thyroid carcinoma patients included in the study was 47.65 ± 13.64 years. There were 26 (43.3%) cases aged 45 years and younger, and 34 (56.7%) cases over 45 years of age. 14 (23.3%) of the cases were male and 46 (76.7%) were female. When the cases were grouped according to histopathological type, 50 cases (71.4%) diagnosed with papillary thyroid carcinoma, 5 cases (7.1%) diagnosed with medullary carcinoma, 1 case diagnosed with anaplastic carcinoma (1.4%), 4 cases diagnosed with well-differentiated tumor with uncertain malignant potential (5.7%) were in the study. In the control group, there were 2 cases (2.9%) diagnosed with follicular adenoma, 3 cases (4.3%) diagnosed with Hurthle cell adenoma, and 5 cases (7.1%) diagnosed with nodular hyperplasia. Among the cases with papillary carcinoma, 13 cases (26%) classical type, 17 cases (34%) follicular type, 13 cases (26%) papillary microcarcinoma, 1 case (2%) macrofollicular type, 3 cases (6%) Warthin-like type, 3 cases (6%) were evaluated as oncocytic type.
When the IMP3 staining results in the study group were evaluated, 48 (80%) of 60 thyroid carcinoma cases showed positive staining with IMP3, while 12 (20%) did not. Of 48 patients with positive staining, 26 (43.3%) were 1+ (Figure 1), 16 (26.7%) were 2+ (Figure 2), 6 (10%) were 3+ (Figure 3) staining was observed. Positive staining was observed in 7 (70%) cases in the control group, and 4 (40%) were evaluated as 1+ and 3 (30%) as 2+. There were no detected 3+ staining cases in the control group and no staining was observed in 3 (30%) cases.
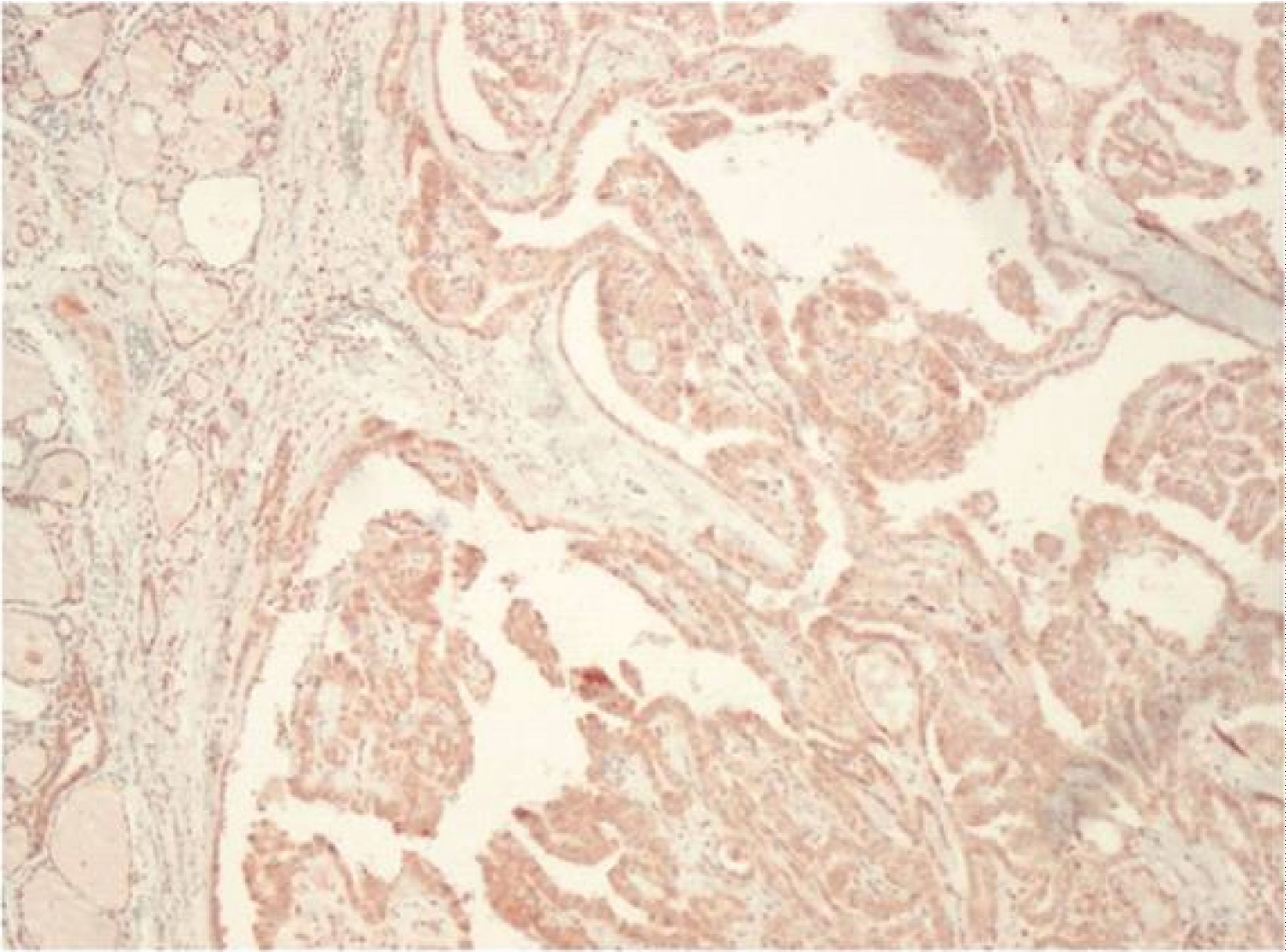 Figure 1: IMP 3 expression, 1+ staining (x100).
View Figure 1
Figure 1: IMP 3 expression, 1+ staining (x100).
View Figure 1
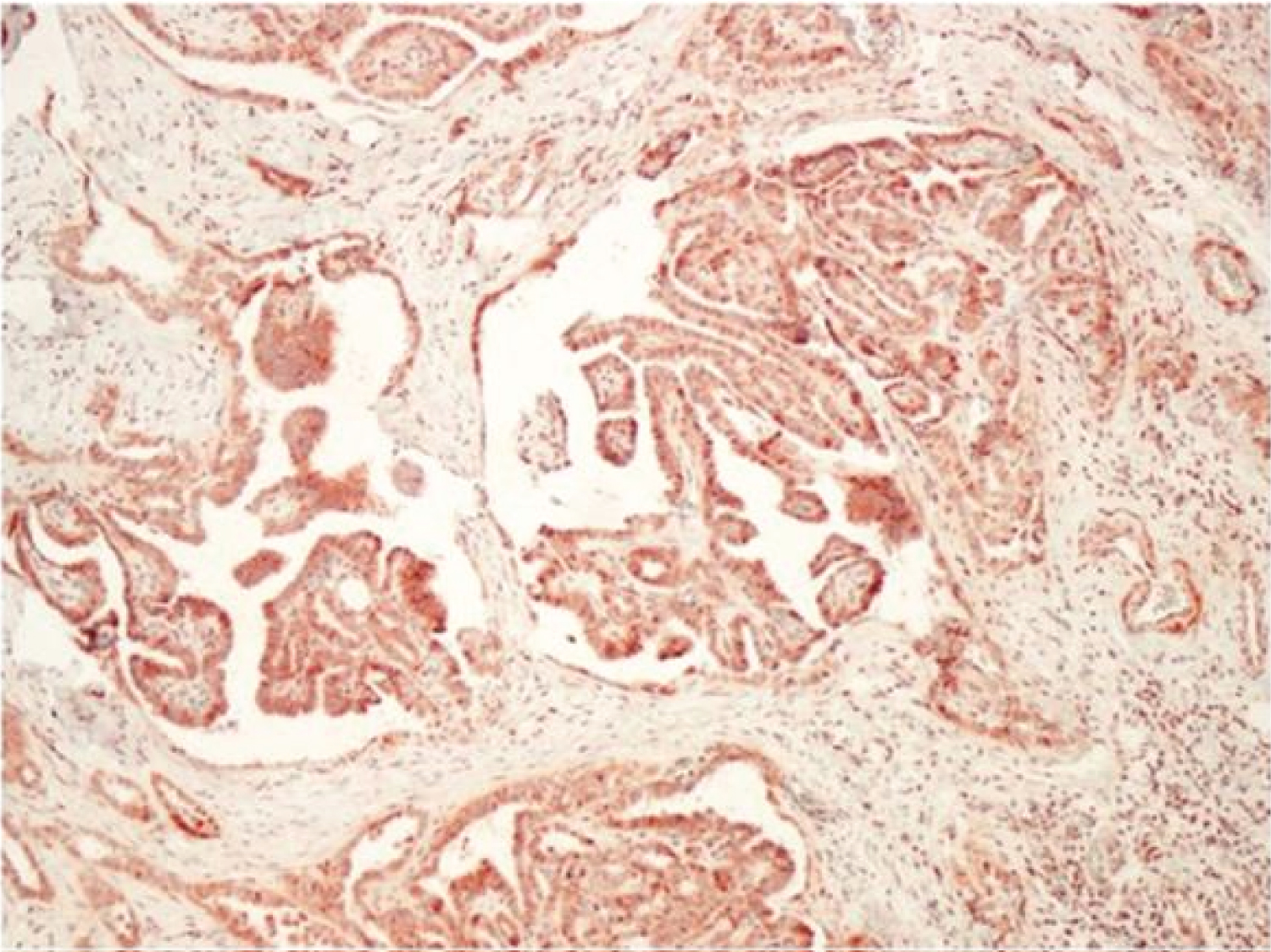 Figure 2: IMP 3 expression, 2+ staining (x100).
View Figure 2
Figure 2: IMP 3 expression, 2+ staining (x100).
View Figure 2
 Figure 3: IMP3 expression, 3+ staining (x100).
View Figure 3
Figure 3: IMP3 expression, 3+ staining (x100).
View Figure 3
While there was 1+ staining in 7 (46.7%) and 2+ staining in 4 (26.7%) of 15 cases with tumor size ≤ 1 cm, no staining was observed in 4 (26.7%) of these. Out of 34 cases with tumor diameter between 1 and 4 cm, 15 (44.1%) staining was 1+, 9 (26.5%) 2+ and 5 (14.7%) 3+ staining. No staining was observed in 5 of these patients with tumor size between 1 and 4 cm. Of 11 cases with tumor diameter > 4 cm, 4 (36.4%) staining was 1+, 3 (27.3%) 2+, 1 (9.1%) 3+ staining, 3 (27%), 3) no staining was observed in the case. There was no significant difference in terms of IMP3 staining score between cases with tumor diameters of ≤ 1 cm, 1 cm to 4 cm, and > 4 cm (p > 0.05). There was no significant difference in terms of IMP3 staining score according to gender between the cases aged ≤ 45 and > 45 years (p > 0.05). In the study group, 21 (42%) of 50 cases diagnosed with papillary carcinoma were 1+, 14 (28%) 2+, 6 (12%) 3+, while no staining was observed in 9 (18%) cases. While 1+ staining was observed in 3 (60%) of 5 cases diagnosed with medullary carcinoma, staining was not observed in 2 (40%). In cases diagnosed with medullary carcinoma, 2+ and 3+ staining was not observed with IMP3. 2+ staining was observed in one case in the study group diagnosed with anaplastic carcinoma. While it was 1+ in 2 (50%) and 2+ in 1 (25%) of 4 patients diagnosed with Well Differentiated Tumor with Uncertain Malignant Potential (WDTUMP), staining was not observed in 1 (25%) patient.
When 50 patients diagnosed with papillary carcinoma were evaluated according to carcinoma subtypes, 5 (38.5%) of 13 patients diagnosed with classical papillary carcinoma were 1+, 3 (23.1%) were 2+, 2 (While there was 3+ staining in 15.4% and no staining was observed in 3 (23.1%) cases. Of 17 cases diagnosed as follicular type papillary carcinoma, 9 (52.9%) had 1+ staining, 4 (23.5%) had 2+ staining, 2(11.8%) had 3+ staining, and no staining was observed in 2 (11.8%) case.
1+ staining was observed in 1 (33.3%) of 3 cases diagnosed with Warthin-like type papillary carcinoma, and 2+ staining was observed in 2 (66.7%) cases. While 1 (33.3%) of 3 cases diagnosed with oncocytic papillary carcinoma had 1+ staining, 2 (66.7%) had 3+ staining, and there were no cases that were not stained with IMP3 and stained 2+. 2+ staining was observed in the only macrofollicular type papillary carcinoma case in the study group. There was no significant difference between papillary carcinoma subtypes in terms of IMP3 staining score (p > 0.05) (Table 1 and Table 2).
Table 1: Comparison of papillary carcinoma and other carcinoma types with IMP3 staining percentages. View Table 1
Table 2: Relationship between prognostic parameters and IMP3 expression. View Table 2
Due to the very high p value, grouping was made between histological types and the results are given in Table 1.
Thyroid carcinomas constitute 1% of all cancers in the world and it is estimated that there are 122,000 new cases of thyroid carcinoma each year. 5-year survival is around 90% [1]. IMP3 is an oncofetal protein and belongs to the IGF-II m-RNA binding protein family like IMP1 and IMP2 [10]. Members of the IMP family are implicated in RNA movement and stabilization, cell growth, and cell migration in the early stages of embryogenesis [6,7]. The IMP3 gene is located on chromosome 7p11.2 [11]. It is a protein equivalent to KOC (K homologous domain-containing protein over expressed in cancer) protein that clones from pancreatic tumors [12]. IMP3 is secreted from the growing epithelium, muscle and placenta during human and mouse embryogenesis. It is reported that it is produced too little to be detected in adult tissues [6,10].
While IMP3 is secreted in many malignant tumors, it is seen that it is not expressed in benign tissues adjacent to the tumor [10,13]. While IMP3 is expressed in pulmonary and extrapulmonary high-grade neuroendocrine carcinomas, its expression is not observed in carcinoid tumors [14]. In many studies, it has been shown that IMP3 is associated with advanced stage and aggressive tumor behavior in tumors in which IMP3 is secreted [6,10,13]. They found a higher rate of IMP3 expression in metastatic renal cell carcinomas than the primary tumor, and concluded that IMP3 expression is an independent poor prognosis factor in primary renal cell carcinoma [14].
In a study investigating the expression of IMP3 in the extrahepatic biliary system, the expression of IMP3 in high-grade dysplasia was found to be significantly higher than the expression in normal, inflamed and low-grade dysplasia cases. In carcinoma cases, IMP3 expression was found to be associated with significantly reduced survival [15].
In a study conducted by Slosar, et al. in 2009, 219 thyroid lesions were examined and it was investigated whether IMP3 expression played a role in the differentiation of thyroid follicular tumors. IMP3 was positive in 38% of 60 follicular type papillary carcinomas, and 3+ staining was observed in 22% of them. Again, in this study, 22% of the follicular carcinomas had positive staining, of which 69% were 3+, while 89% of the classical type papillary thyroid carcinomas were positive and this staining was 1+/2+. IMP3 expression was not observed in hyperplastic colloidal nodule, Hurthle cell adenoma, follicular adenoma, Hashimoto's thyroiditis and Graves' disease. In the study conducted by Slosar, et al. although it was stated that IMP3 may be useful in the differentiation of malignant and benign thyroid tumors, no correlation was found between pathological tumor characteristics and IMP3 expression [16].
In a study conducted with poorly differentiated thyroid carcinomas, IMP3 was found to be positive in 61 (59%) of 103 poorly differentiated thyroid carcinomas, and the cases considered negative were completely negative or had a score less than 2+. No relationship was found between tumor characteristics such as age, gender, tumor size, vascular invasion, oncocytic features, growth pattern, necrosis and IMP3. In this study, it was stated that IMP3 was associated with overall survival, disease-free survival, and metastasis-free survival, and that it could be used as a prognostic parameter to identify types with more aggressive behavior compared to calmer poorly differentiated thyroid carcinomas [17].
Twenty-six (43.3%) of the subjects included in our study, whose ages ranged from 20 to 77 years, were 45 years or younger, and 34 (56.7%) of them were over 45-years-old. While 20 (76.9%) of the cases under 45 years of age were diagnosed with papillary carcinoma and 6 of them were diagnosed with other type of thyroid carcinoma, 30 (88.2%) of the cases over 45 years of age were diagnosed with papillary carcinoma, and 4 (11.8%) of them were diagnosed with other types of thyroid carcinoma. There was no statistically significant difference between age and other prognostic parameters. Although inconsistent with the literature, it was thought that this conclusion was reached due to the small number of cases included in the study.
In the study conducted by Jukkola, et al. with 499 differentiated thyroid carcinomas, it was concluded that male patients had a worse prognosis and gender was an independent risk factor [18]. In the study conducted by Loh, et al. with 700 thyroid carcinoma cases, tumor recurrence was found to be 2 times higher in men than women, and gender was thought to be an independent risk factor in predicting recurrence [19]. In our study, 14 (23.3%) of the cases were male and 46 (76.7%) were female.
Considering the tumor size in female cases, tumor size was smaller than 1 cm in 13 (28.3%) patients, between 1 and 4 cm in 29 (63.0%) and larger than 4 cm in 4 (8.7%) patients while in male cases the tumor size was less than 1 cm in 2 (14.3%) cases, between 1 and 4 cm in 5 (35.7%) and larger than 4 cm in 7 (50.0%) of them. Tumor size, which is an important prognostic parameter, was found to be larger in men than in women, and the relationship between gender and tumor size was found to be statistically significant (p = 0.002). Surgical margin involvement was found positive in 3 (6.5%) female cases, while it was detected in 4 (28.6%) of men. Surgical margin involvement was found to be more common in males, and the relationship between gender and surgical margin involvement was found to be statistically significant (p = 0.024).
In another study, it was reported that the tumor size ranged from 1-4 cm in most of the cases, and the risk of recurrence was higher in tumors larger than 4 cm [18]. Of the cases in our study, 15 (25%) were 1 cm or less, 34 (56.7%) were between 1 and 4 cm, and 11 (18.3%) were larger than 4 cm. Vascular invasion was detected in 7 (63.6%) cases with tumor size larger than 4 cm, and the relationship between tumor size and vascular invasion was statistically significant (p = 0.001).
Histological type of tumor is one of the most important prognostic parameters that affects survival. Although the 10-year survival rate is over 90% in well-differentiated thyroid carcinomas except medullary carcinoma, it is known that follicular carcinomas have a more aggressive course. In anaplastic carcinomas, the 10-year survival rate is extremely low [20,21]. Mortality risk was found to be higher in follicular carcinomas compared to papillary carcinomas [20]. In a study, it was reported that 82.1% of malignant thyroid tumors were positive with IMP3. Again, according to this study, no significant difference was observed between benign and malignant tumors [22].
When the prognostic parameters were evaluated, vascular invasion was found in 18% of papillary carcinomas (9 cases), 60% of medullary carcinomas (3 cases) and 100% of anaplastic carcinomas (1 case), and that was found a statistically significant (p = 0.024). Lymph node metastases were present in 10% of papillary carcinomas, 40% of medullary carcinomas, and 100% of anaplastic carcinomas, and these findings also were statistically significant (p = 0.014).
Multicentricity causes an increase in local recurrence and mortality, especially in papillary carcinoma [23]. In a study conducted with 700 thyroid carcinomas, it was reported that tumor recurrence was 1.7 times higher in multifocal tumors than unifocal tumors, but no correlation was found between multifocality and mortality [19]. In our study, multifocality/ multicentricity was detected in 23 (38.3%) cases, but not in 37 (61.7%) cases. Of the cases with multifocality/multicentricity, 20 (87%) were papillary carcinoma, 2 (8.7%) medullary carcinoma, 1 (4.3%) anaplastic carcinoma. This finding was consistent with the literature.
As a result; It has been shown that IMP-3 plays a role in carcinogenesis and prognosis in many malignant tumors including hepatobiliary system, esophagus, stomach, pancreas, urothelial tumors and melanocytic lesions. There are few studies indicating that IMP-3 is useful in the differentiation of follicular tumors in thyroid carcinomas and that it can be an effective parameter in determining the prognosis in thyroid carcinomas with an aggressive course. According to our study, all of the cases whose IMP3 expression was evaluated as 3+ were papillary thyroid carcinoma. However the findings of our study show that IMP3 expression does not fully differentiate between thyroid carcinoma types and is not effective as a prognostic indicator. These findings show that more studies should be done in large series to determine the status of IMP3 expression in thyroid carcinoma cases and its importance in the prognosis, as it is not compatible with the small number of literature.
MG and GÖ conceived and designed the study. MG, GÖ, SPB supervised the conduct of the study and data collection. MG, GÖ, SPB managed the data, including quality control. GÖ and MG analyzed the data. GÖ, MG, SPB performed the literature review. MG and GÖ drafted the article, and all authors contributed substantially to its revision. MG takes responsibility for the paper as a whole.
No conflict of interest was declared by the authors.