Although angiosarcoma and squamous cell carcinoma represent completely different tumor entities their histologic features in some cases might be similar, especially in the head and neck region. Interestingly the clinical features might be also identical.
We report a case of an acantholytic squamous cell carcinoma, observed in a male individual in his 7th decade of life. It presented as a polyp-like, ulcerated mass in the oral cavity. The histologic findings revealed diffusely infiltrative malignant tumor composed of the clusters of ovoid to spindle neoplastic cells with hyper chromatic nuclei and densely eosinophilic cytoplasm and with anastomosing vessel-like channels lined by a single layer of atypical epithelioid and hobnail-like cells on slightly myxoid background. Some atypical cells also had the tendency of forming pseudoglandular structures. Few keratin globules were also observed in sub epithelial connective tissue. The surface mucosal epithelium showed dysplastic changes. No mucin was identified. Because of these morphologic features seen on H&E stained slides the diagnosis of squamous cell carcinoma and angiosarcoma was considered. Immunohistochemical analysis led to a conclusive diagnosis of acantholytic variant of squamous cell carcinoma-the tumor cells expressed an epithelial marker cytokeratin 5/6 and a nuclear stain - p63, confirming the squamous differentiation.
Acantholytic squamous cell carcinoma is an uncommon variant of squamous cell carcinoma which is characterized by a combination of typical squamous cell carcinoma and pseudovascular structures, dyskeratotic cells and prominent acantholysis - loss of coherence between epithelial cells due to breakdown of intercellular bridges. This cancer is known to have an aggressive nature, especially when it occurs intraorally.
In conclusion, careful attention should be paid to the histological features of this rare variant of squamous cell carcinoma because tissue biopsy samples can easily be mistaken for angiosarcoma due to the presence of pseudoangiomatoid patterns. Immunohistochemical analysis is generally helpful in establishing an accurate diagnosis.
Acantholytic, Squamous cell carcinoma, Oral cavity
Acantholytic Squamous Cell Carcinoma (ASCC) is a very rare variant of the most common form of cancer found in the oral cavity which is Squamous Cell Carcinoma (SCC). It occurs mostly after the age of 60. UV radiation significantly increases the risk of ASCC. A great deal of evidence points to the significance that acantholytic squamous cell carcinoma of the oral cavity has an uncanny resemblance to that of an angiosarcoma under the pathological microscopic view. The similarity is due to the separation of layers between cells known as acantholysis. It can present as a fast locally growing mass that has a poorer prognosis compared to conventional SCC of the oral cavity. The acantholytic squamous cell carcinoma and angiosarcoma both have pseudovascular spaces, however on immunohistochemistry they can be distinguished. Hence it is important to know the differences between these two as treatment options are separate.
The formalin-fixed paraffin-embedded (FFPE) tissue samples with the material from the oral cavity of a 72-year-old male individual has been provided to the pathology laboratory of High Technology Medical Centre-University clinic for the consultation. On routine microscopic examination, the biopsy showed diffusely infiltrative malignant tumor composed of the clusters of ovoid to spindle neoplastic cells with hyperchromatic nuclei and densely eosinophilic cytoplasm and with anastomosing vessel-like channels lined by a single layer of atypical epithelioid and hobnail-like cells on slightly myxoid background. Some atypical cells also had the tendency of forming pseudoglandular structures. Few keratin globules were also observed in subepithelial connective tissue. The surface mucosal epithelium showed dysplastic changes. No mucin was identified. Because of these morphologic features seen on H&E stained slides the diagnosis of squamous cell carcinoma and angiosarcoma was considered. Immunohistochemical analysis led to a conclusive diagnosis of a moderately differentiated acantholytic variant of squamous cell carcinoma as the atypical cells showed positivity on cytokeratin 5/6 and p63. Vascular markers like CD34 and smooth muscle actin showed to be negative and only remained positive in internal control - true vessel channels.
In this article, we are trying to emphasize how similar acantholytic variant of squamous cell carcinoma and angiosarcoma is on histopathology. Currently, there is a lot of literature about ASCC in head and neck region available that describes the similarities between the two pathologies and why it is important to distinguish them. But, there are not many intraoral cases of ASCC.
Squamous cell carcinoma (SCC) is the most common cancer in the oral cavity. Acantholytic squamous cell carcinoma (ASCC) is a rare variant of SCC [1]. Acantholysis is the loss of adhesion molecules like E-cadherin, catenin, syndecan-1 and others that bind the cells tightly to each other resulting in separation of layers of cells [2-4]. Loss of these adhesion molecules also aid in the invasion of malignant cells in the tissue architecture [2]. In ASCC, these spaces are large enough to mimic pseudovascular or pseudoglandular spaces [1]. Angiosarcoma on histology has similar findings - irregularly anastomosing vascular channels with endothelial and stromal infiltration. Sometimes, reactive fibrosis within the tissue along with artifactual clefts lined by malignant cells create angiomatous pattern [5]. Hence, it is difficult to distinguish the two based on histology only and requires the neeartifactual clefts lined by malignant cells create angiomatous pattern [6,7] we can see and compare how closely both mimic each other in histology. Other characteristics on H&E staining that can be seen are neoplastic cells, hyperchromatic nuclei, eosinophilic cytoplasm, atypical eiptheloid cells and keratin pearls, like what the biopsy fragments in this case report revealed.
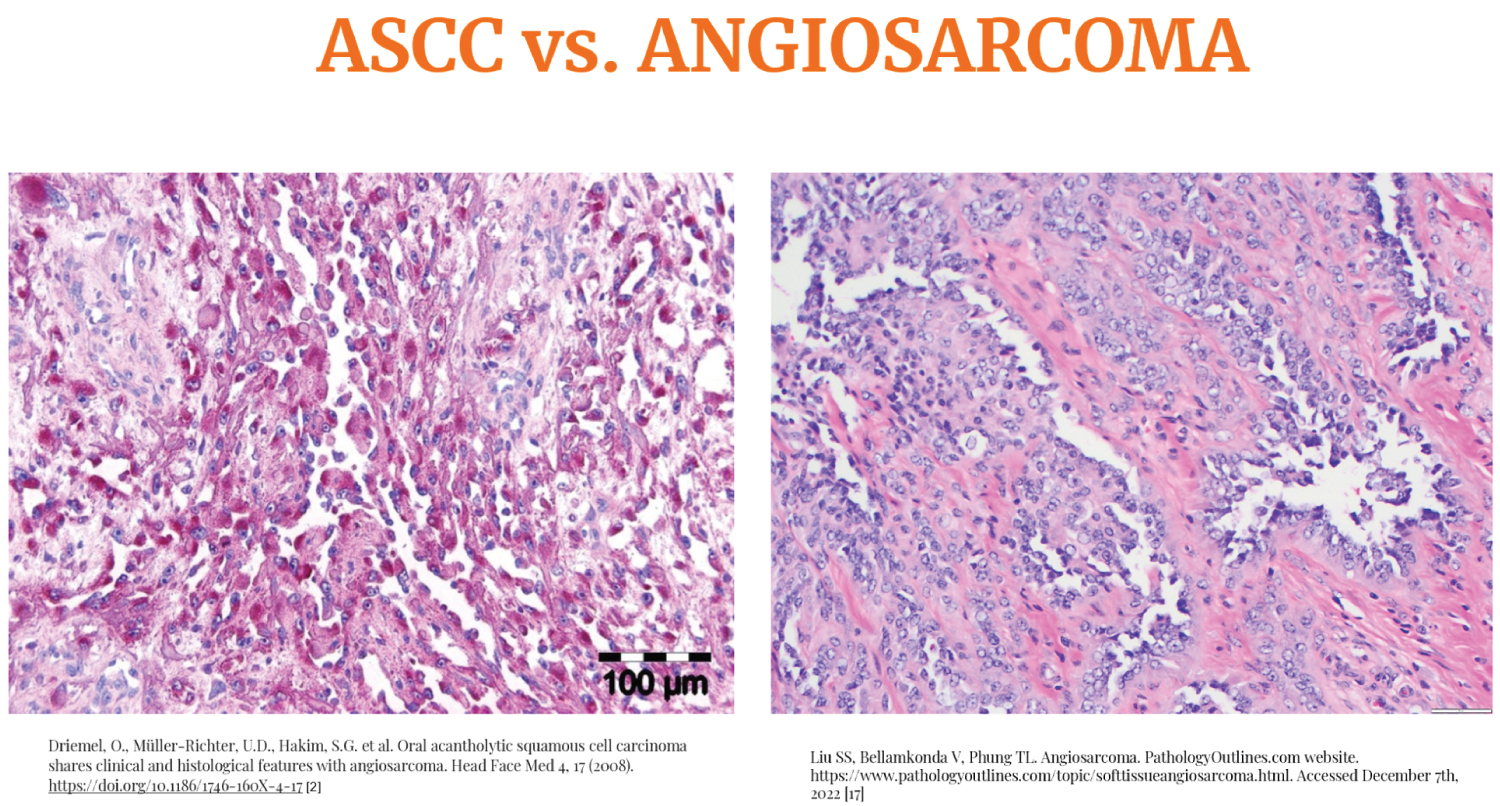 Figure 1: Comparison of ASCC and Angiosarcoma on H&E biopsy samples [6,7].
View Figure 1
Figure 1: Comparison of ASCC and Angiosarcoma on H&E biopsy samples [6,7].
View Figure 1
The clinical presentation of both are usually the same in the oral cavity. They are locally aggressive and rapidly growing lesions with a poor prognosis. They can present as a polyp, mass or ulcer with irregular borders [8,9]. Both ASCC and angiosarcoma can have hemorrhagic regions [8]. Regional lymph nodes may be enlarged. Both present in the late stages of life with average incidence of ASCC and Angiosarcoma in age 60 and 70, respectively [9]. UV radiation is thought to have a role in the etiology of cutaneous ASCC because most tumors grow on sun-exposed regions with signs of actinic keratosis [2,8,10]. In contrast to oral angiosarcoma, ASCC has a tendency to present 1 to 3.5 times more common in males [8]. According to some articles, ASCC is more aggressive than classic SCC but some studies deny this claim as current literature does not have enough number of cases reported to make conclusions [11,12].
Not to be confused, ASCC term is interchangeably used with a lot of other terms - adenoid SCC, pseudoglandular SCC, angiosarcoma-like SCC, and pseudoangiosarcomatous carcinoma [8,9].
In our case, after H&E staining two differentials were considered-Acantolytic SCC and Angiosarcoma. Immunohistochemistry was planned to confirm diagnosis. This test is specific and can distinguish between tumors by checking for cell markers. Carcinoma has epithelial origin and will stain positive for epithelial markers whereas sarcoma has mesenchymal origin and will stain positive for mesenchymal markers (Figure 2 and Figure 3).
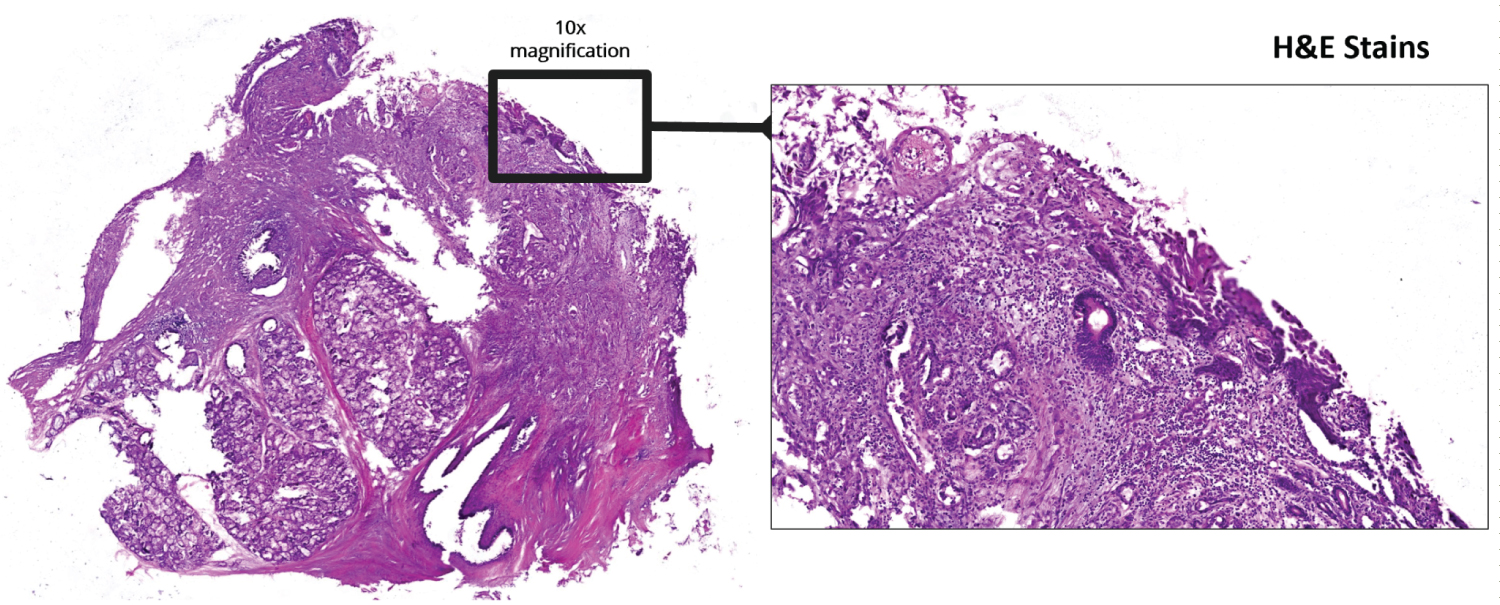 Figure 2: Low power magnification H&E stains.
View Figure 2
Figure 2: Low power magnification H&E stains.
View Figure 2
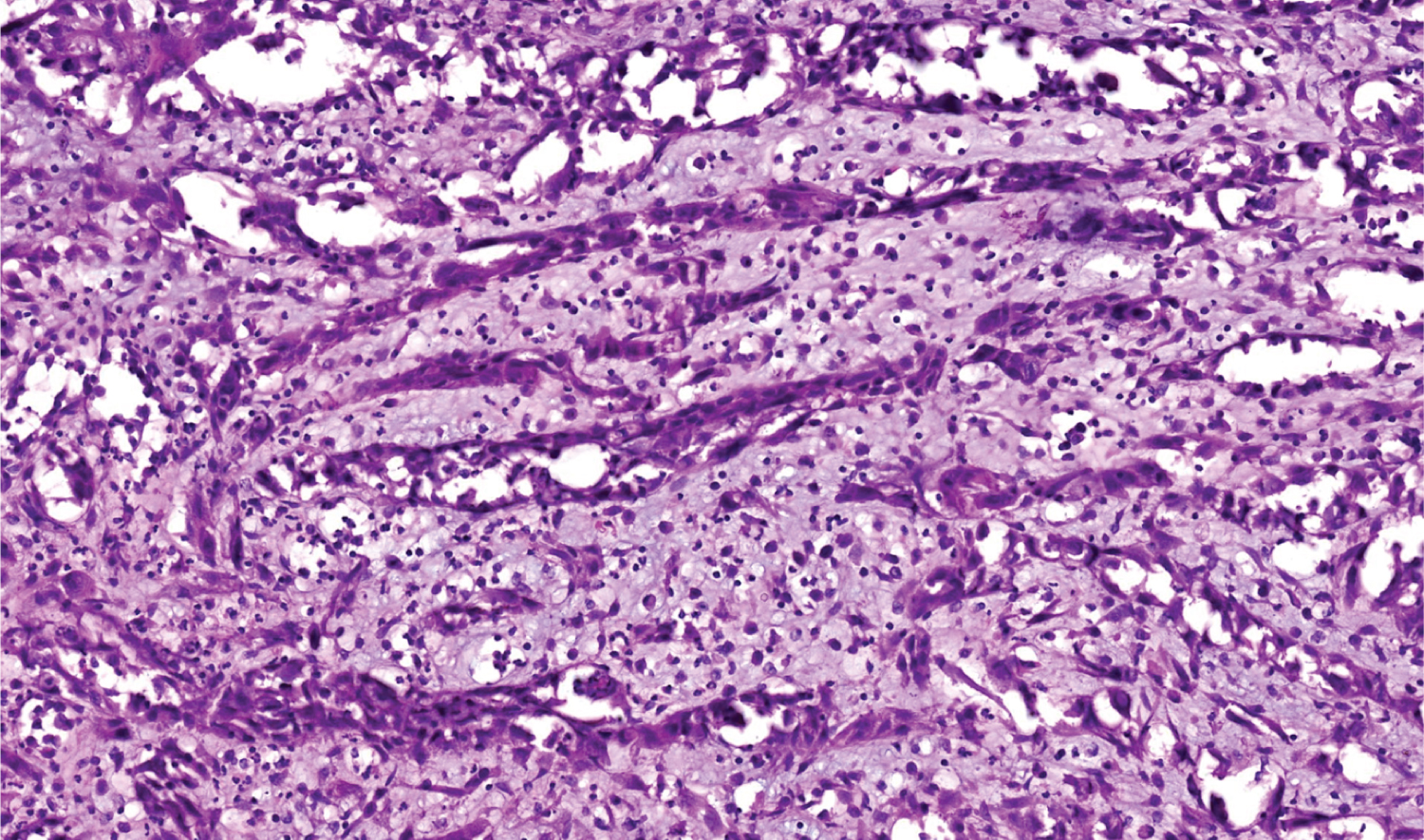 Figure 3: H&E stain 20x power magnification.
View Figure 3
Figure 3: H&E stain 20x power magnification.
View Figure 3
In immunohistochemistry, angiosarcoma will stain positive for vascular antigens like CD 31, CD 34, von willebrand factor (vWF) and smooth muscle actin [1,13]. A novel vascular differentiation marker, Fli-1 protein, a DNA binding transcription factor from the ETS family is positive in angiosarcoma [1,13]. ASCC will stain positive for cytokeratin, p63 and epithelial membrane antigens. Laminin-5 is a new marker that stains positive in the cytoplasm of ASCC. It is also known as a biological indicator for the poor prognosis of ASCC [1]. In a study, it was discovered that both ASCCs and angiosarcoma were cytokeratin-positive but angiosarcoma had less cytokeratin-positive tumor cells than ASCC did [8,14-17]. Pictures of H&E staining and immunostains cytokeratin 5/6 (Figure 4), p63 (Figure 5), CD 34 (Figure 6) and SMA (Figure 7) from our patient are added here. The pictures were received from Dr. Manana Jikurashvili, High Technology Medical Centre-University Clinic, CSD-Georgia. In these images, diffuse acantholysis (white spaces) and malignant infiltrative cells staining brown in color are noted.
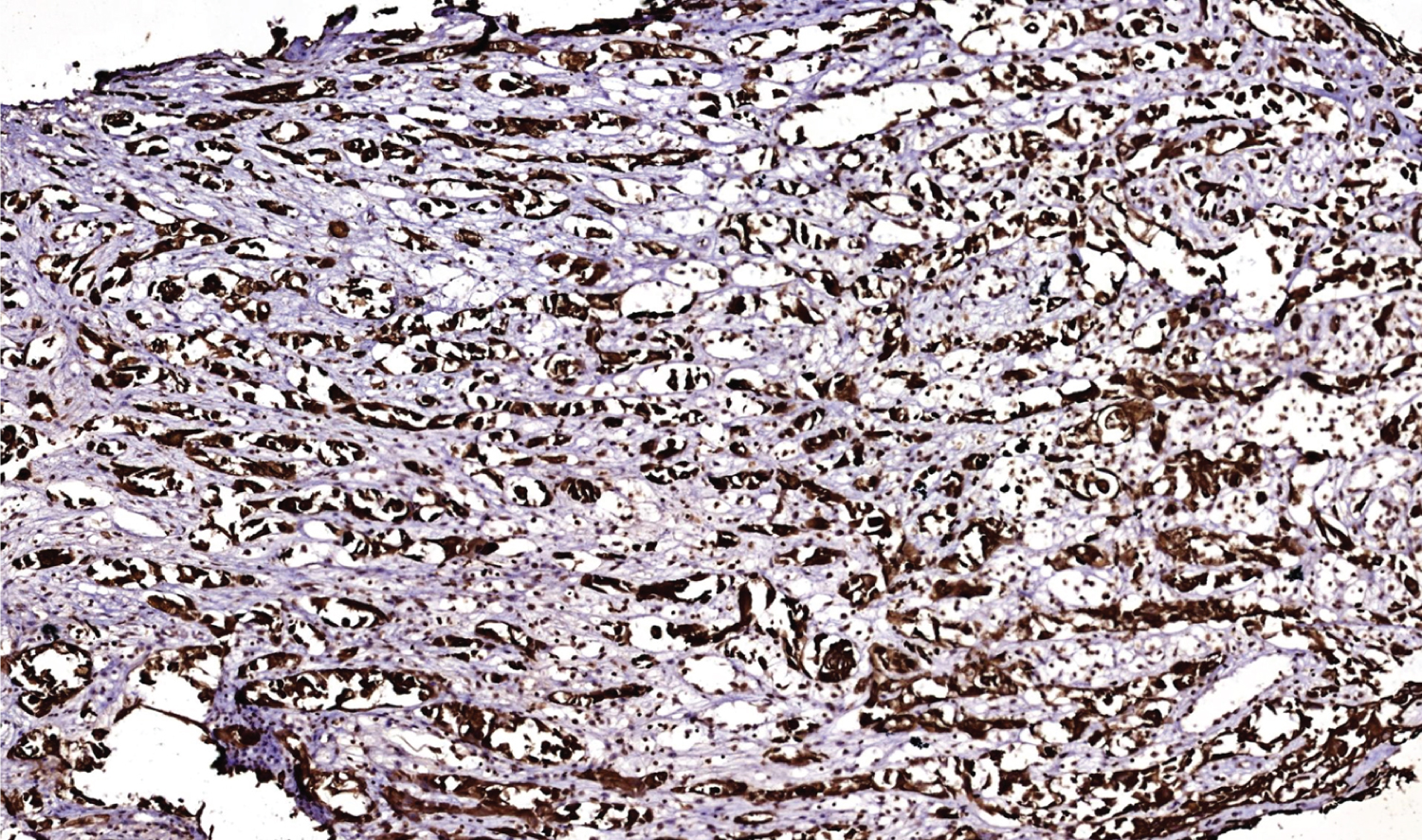 Figure 4: Cytokeratin 5/6 stain, 10x power magnification.
View Figure 4
Figure 4: Cytokeratin 5/6 stain, 10x power magnification.
View Figure 4
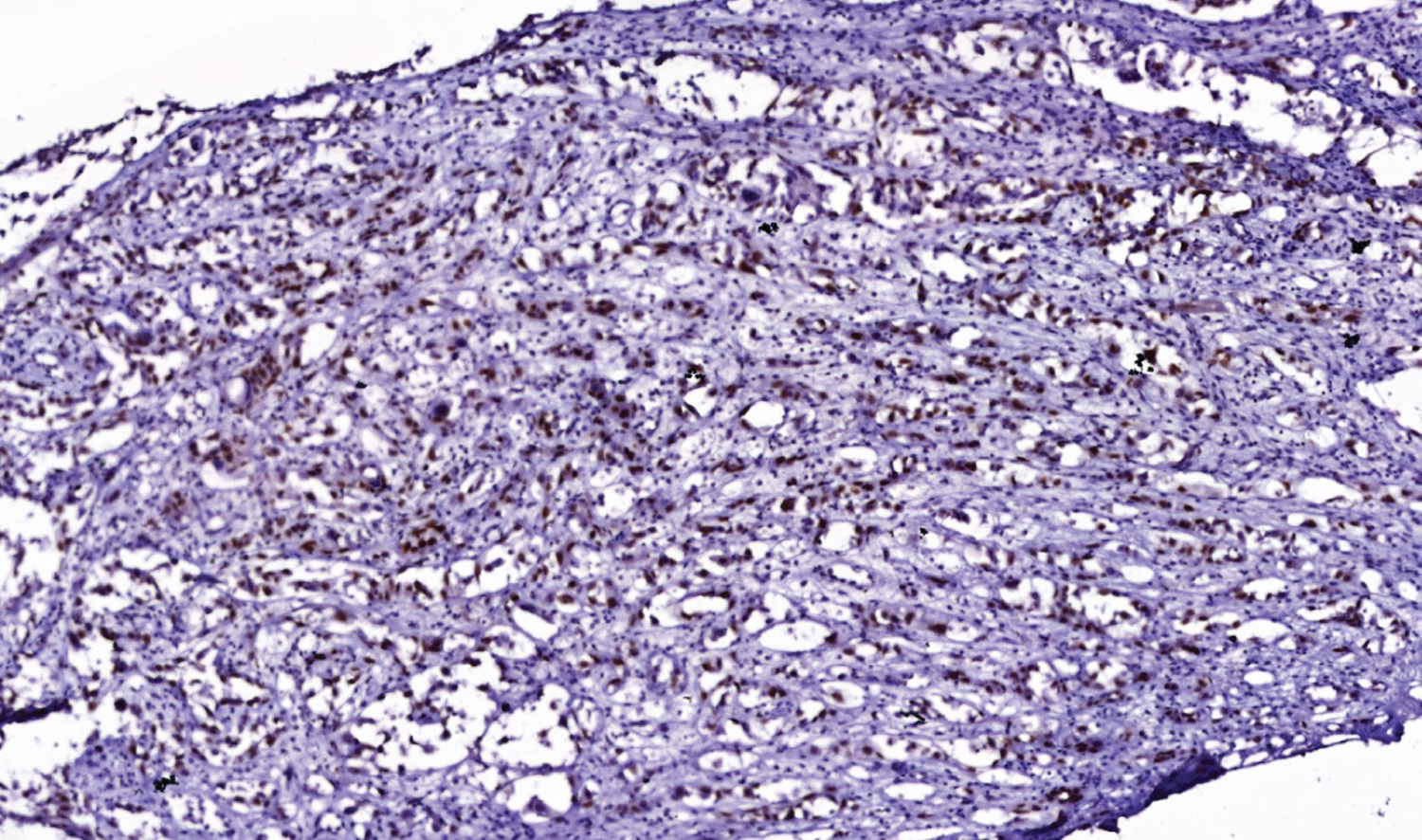 Figure 5: p63 immunostain, 10x magnification stain.
View Figure 5
Figure 5: p63 immunostain, 10x magnification stain.
View Figure 5
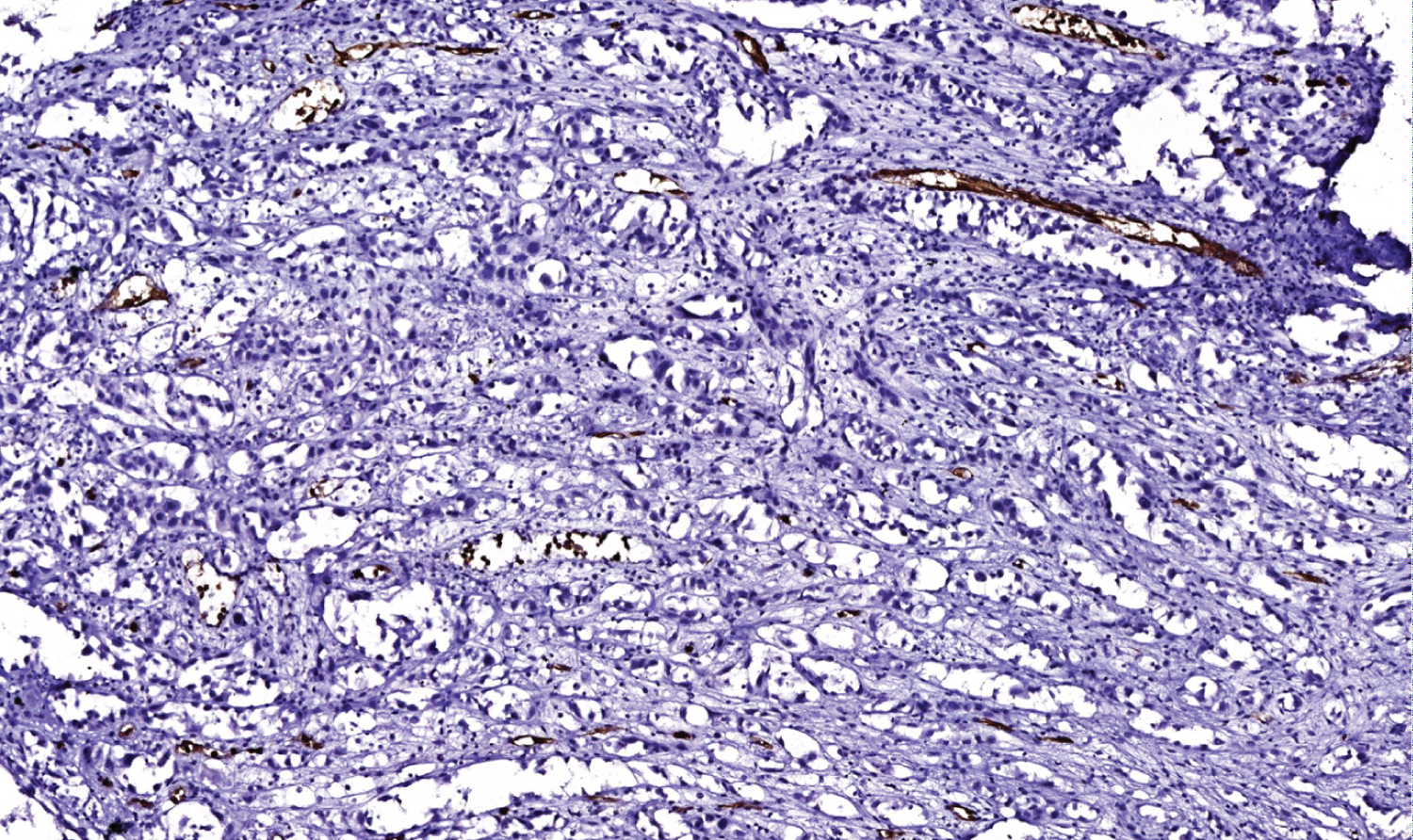 Figure 6: CD 34 stain, 10x power magnification.
View Figure 6
Figure 6: CD 34 stain, 10x power magnification.
View Figure 6
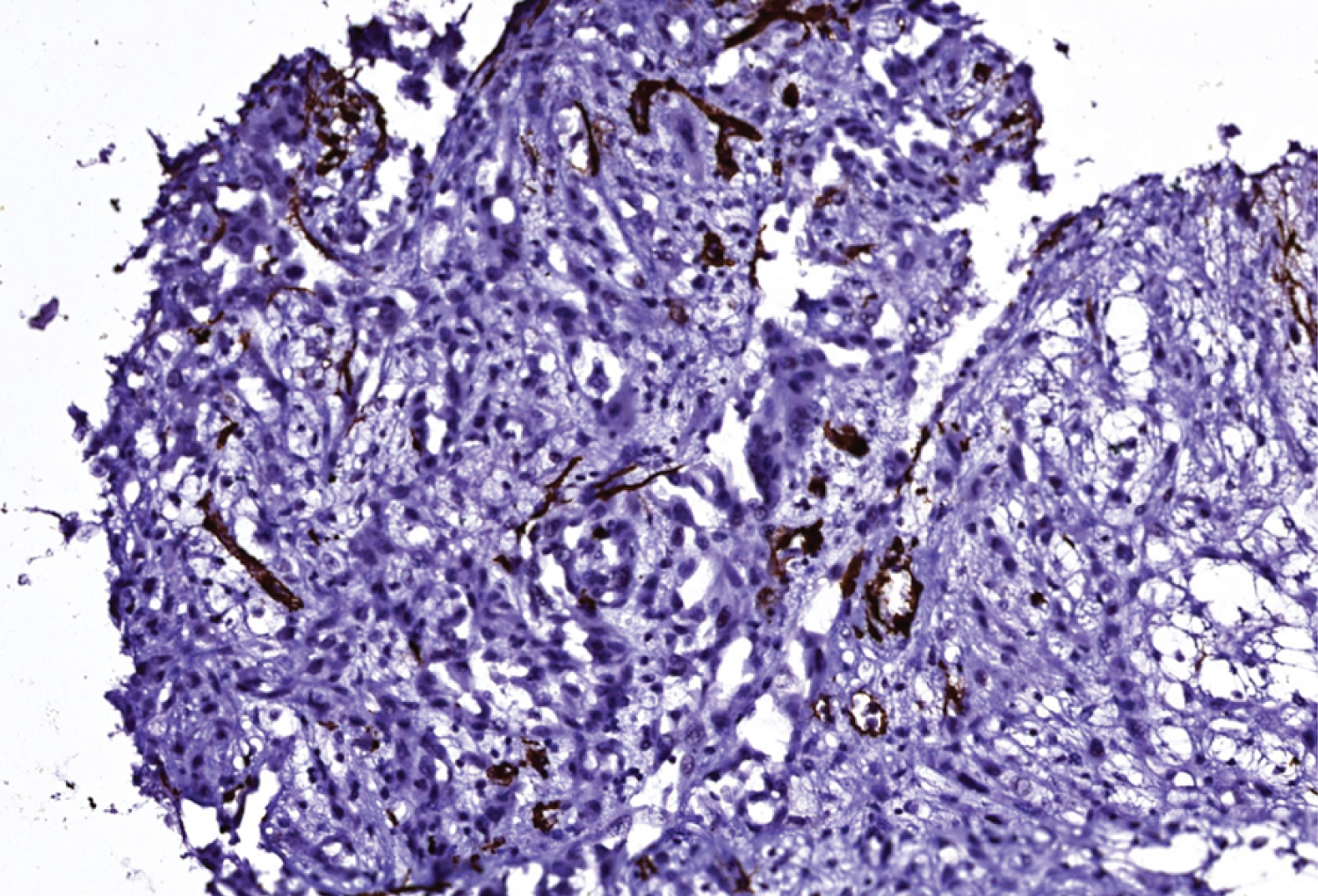 Figure 7: Smooth muscle actin stain (SMA), 20x power magnification.
View Figure 7
Figure 7: Smooth muscle actin stain (SMA), 20x power magnification.
View Figure 7
After immunohistochemistry, Acantholytic Squamous Cell Carcinoma was confirmed as the diagnosis. From the figures attached we can also confirm it by looking at the brown colored areas in immunostains. There is brown discoloration in cytokeratin 5/6 and p63 stain which verifies cell origin is epithelial and confirms carcinoma. There is no brown discoloration in CD 34 and SMA stain which verifies cell origin is not mesenchymal and rules out sarcoma. There is a small brown discoloration in Figure 6 (CD 34) and Figure 7 (SMA). This is the internal control smooth muscle in the blood vessel wall, the area which is normal and healthy. Internal control also tells us that the staining method was performed correctly.
The patient was consulted with the oncology department and is currently on radiation therapy.
This concludes our study that tissue biopsy H&E samples of ASCC can be confused for angiosarcoma due to the presence of pseudovascular or pseudoglandular patterns arising from acantholytic areas. Hence, extra caution should be made to the histological characteristics of this uncommon type of squamous cell carcinoma and confirmed with immunohistochemistry to avoid misdiagnosis.
We sincerely thank Dr. Manana Jikurashvili, Pathologist, Department of Pathology, Tbilisi State Medical University for her help and support in this report as our scientific supervisor.
The authors declare that they have no conflict of interest.
No funding was required.