Background: Physical activity (PA) is associated with positive health-related quality of life outcomes in children and young people (CYP). However, many CYP do not meet recommended PA guidelines set by the World Health Organization (WHO). Hospital admissions reduce PA levels in adults but no review has looked into CYP levels.
This review aimed to summarise existing evidence of PA levels of CYP in inpatient settings.
Method: The study was registered on PROSPERO CRD42023418334 and followed PRISMA reporting guidelines. A systematic review was conducted, with 18 studies included in final synthesis.
Results: CYP with physical health conditions had significantly lower PA levels than recommendations during inpatient admissions. In contrast, individuals with Anorexia Nervosa (AN) met WHO guidelines for Moderate to Vigorous PA. Fidgeting was observed to be higher in admitted AN patients compared to community, suggesting a potential compensatory mechanism for reduced step count. Notably, hospitalisation itself resulted in a decline in PA levels.
There is a lack of generalisability of this data due to limited conditions investigated. Additionally, the use of different assessment tools, such as questionnaires and accelerometers, introduced heterogeneity and limited comparability between studies.
Overall, the findings underscore the importance of promoting PA in CYP during hospitalisation, particularly in those with physical health conditions.
Conclusion: This review sheds light on disparities in PA levels among CYP with physical health conditions and AN in the inpatient setting. Addressing these discrepancies of PA during hospitalisation could lead to improved health outcomes and enhanced overall quality of life for these populations.
Mental health, Physical health, Step count, Physical activity
Physical Activity (PA) levels are associated with numerous health-related quality of life outcomes for children and young people (CYP), such as cognitive development, psychosocial and cardiometabolic health, motor development, fitness, and bone health [1-3]. The most benefit appears to be with moderate-to-vigorous (MVPA) and vigorous intensity (VPA) physical activity. Lower intensities of physical activity, described as light intensity (LPA) and moderate intensity (MPA), show less effect. There is also evidence to show that a variety of type of exercise, to include things like muscular strengthening and cardiovascular endurance exercise, are required to maximise benefits [2].
Sedentary behaviour, particularly excessive screen time, is associated with poor mental health, unfavourable behaviour, worse sleep, and poorer cardiometabolic health [2,4]. Although, certain sedentary activities, like reading and drawing, are important for childhood development. High sedentary time has been linked to increased all-cause mortality, reduced sleep duration and quality, musculoskeletal issues, depression, and cognitive impairment [5].
Current recommendations for CYP aged 5-17 include one hour per day of MVPA and three days of vigorous intensity muscle and bone strengthening exercises. The step count target for adults is commonly set at 10,000 steps per day [6], notably this is not for optimum benefit but as a realistic target to increase PA levels of the general population. For children and adolescents, 12,000 steps per day have been endorsed [7], while limiting sedentary screen time to no more than an hour per day [8]. The UK chief medical officer's recommendations for CYP with disabilities suggest 20 minutes of exercise per day with strength and balance activities three times per week [9].
Unfortunately, globally, the majority of children do not meet the recommended guidelines for PA, screen time, and combined sleep [10]. Hospitalised children should have ample opportunities for exercise due to the presence of healthcare professionals [11]; however, factors including illness, limited mobility, or reduced motivation in the hospital environment further hinder meeting guidelines [12].
Beyond reducing mortality, PA is employed as a treatment across adult and paediatric in physical and mental health populations. It has shown promise as an anti-depressive treatment for adolescents, a supportive intervention for airway clearance in people with cystic fibrosis, and has beneficial effects on reducing inflammation in people with asthma [13-15]. PA can also impact chemotherapy-induced peripheral neuropathy in people undergoing cancer treatments [16].
While there is clear evidence of the benefits of PA, little is known about PA levels in the paediatric inpatient setting. This review aims to compile evidence on CYP's inpatient PA levels, how these levels are monitored and explore potential differences between different patient populations.
- To quantify CYP's inpatient PA levels,
- To understand how these levels are monitored,
- To explore any potential differences between different patient populations.
The research question was developed using the patients-intervention-comparison-outcome (PICO) approach.
Question: Do Paediatric patients in an inpatient setting meet WHO guidelines for physical activity levels?
P - Paediatrics in an inpatient setting
I - No intervention
C - WHO guidelines
O - Physical activity levels
The 2020 PRISMA guidelines [17] were followed for this study and the protocol has been registered on PROSPERO (ID: CRD42023418334).
Inclusion Criteria
- Papers published in conferences and journals, as full research paper, short and position paper presenting new and emerging ideas, as well as doctoral symposiums,
- Paper explicitly measures PA,
- Paper includes or is specific to paediatric patients,
- The patient is in a hospital or acute setting.
Exclusion Criteria
Studies not published in English by title,
The patient is in an outpatient or home setting,
The population is exclusively adults (> 18),
The abstract is not available.
CINAHL, MEDLINE, SPORTDiscus, Amed databases were reviewed on 27 th January 2023 and rescreened on 15 th October 2023.
A manual search of the reference lists of included articles was also undertaken. All references were analysed by authors in Rayyan web software [18].
Searches are in Appendix 1.
Two investigators (KL/CH) performed screening all of the titles and abstracts independently. Studies deemed not relevant based on eligibility criteria were excluded at this point. A third reviewer (DY) helped to resolve conflicts through facilitated discussion with investigators. Two instigators (KL/CH) read all the full-text articles and reviewed against eligibility criteria. The third reviewer (DY) facilitated discussion to resolve conflicts.
Where data included participants over the age of 18, authors were contacted and data for participants 18 and under was requested. All studies were included in narrative synthesis. For statistical analysis, if data for participants under 18 was not available, only studies whose mean age was 18 or below were included.
Data items: One reviewer (KL), using a pre-determined template, agreed between the three authors, extracted data; this was then reviewed by a second reviewer (CH). The following data was extracted from included studies: Number of participants; Age of participants; Sex of participants; Diagnosis of participants; Type of inpatient setting; Outcome measure for assessment of PA; Make and position of accelerometer (if used).
Risk of bias: Appropriate CASP tools were used for each study design. Evaluation of the risk of bias for Cohort studies was assessed by the Newcastle-Ottowa scale [19], and for Cross-sectional studies a modified Newcastle-Ottowa scale described by Herzog, et al. [20] with minor adjustments to outcome measures (Appendix 2).
Figure 1: PRISMA flow diagram.
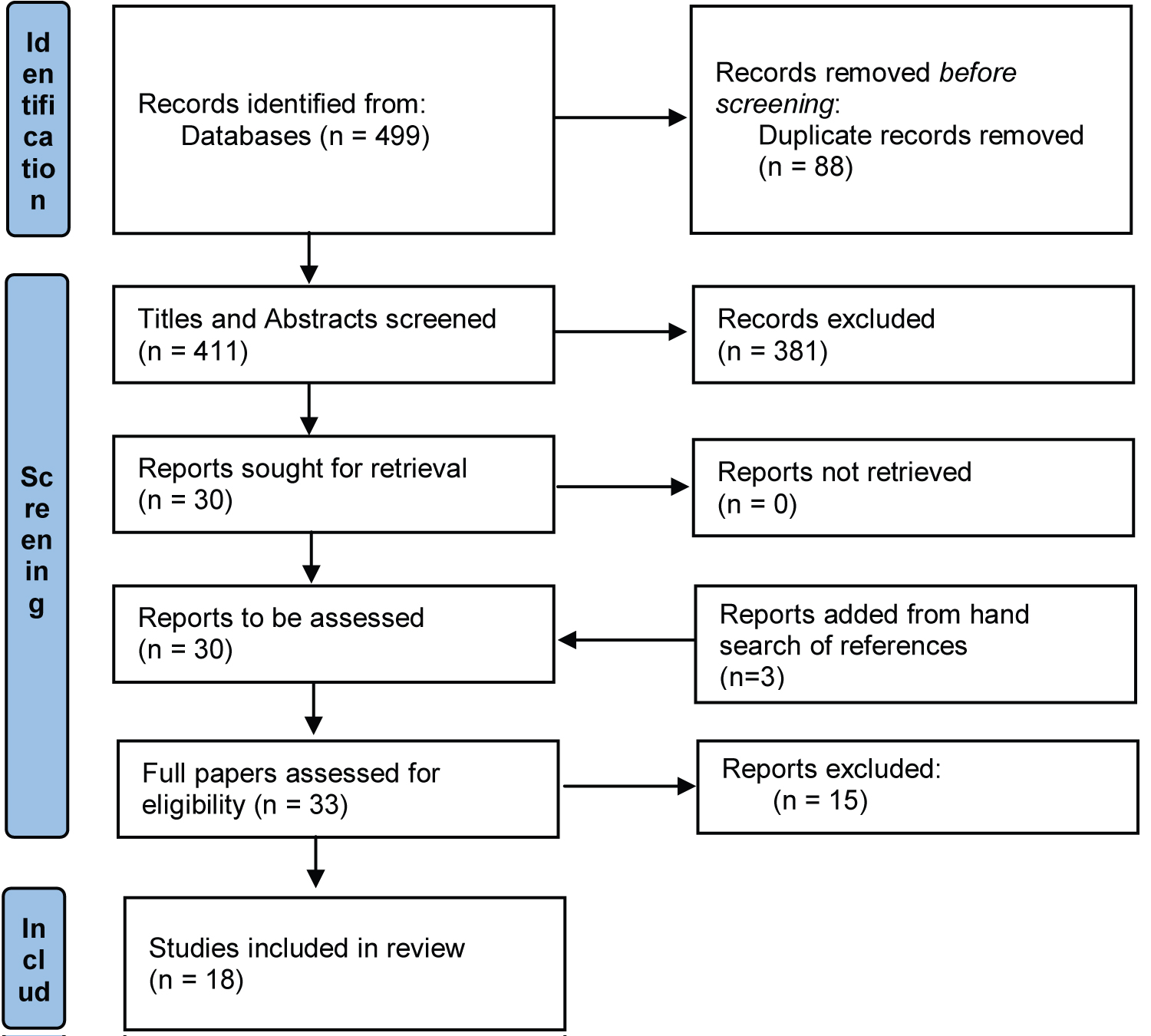 Figure 1: PRISMA flow diagram.
View Figure 1
Figure 1: PRISMA flow diagram.
View Figure 1
13 Cross-sectional and six case-control cohort were included. Of included studies 7 (36.8%) contained adults, all of which had participants with Anorexia Nervosa (AN). A variety of health conditions were studied, 8 (44.4%) AN, 4 (22.2%) Oncology, 2 (11.1%) post-operative, 1 (5.6%) neurological diagnosis, obesity, malnutrition and mental health requiring antipsychotic medication. Seven of eight (87.5%) articles assessing AN had female only participants. 8 (44.4%) included participants over the age of 18, all studied AN, the remaining 10 (55.6%) had exclusively participants 18 or younger.
Four studies had participants in a general paediatric inpatient ward, one in a specialist rehabilitation ward, one on a weight loss ward, four from an oncology ward and eight were based in specialist mental health units wards or units.
Six studies documented step count; nine studies documented an intensity of PA or sedentary time. Fifteen studies used accelerometers to measure PA levels in the population. Two studies measured accelerometer data from the hip. Seven measured accelerometery from the arm/wrist of which two measured on either arm, two measured on the non-dominant arm, one on the dominant arm and two did not discuss. One chest, two hip and wrist, one shoe and one chest, thigh, and feet. Questionnaires were used in five studies; each of these used a different PA questionnaire.
Seven studies investigated time spent sedentary. Five of these reviewed patients with AN [21-25] and two [26,27] assessed those with an oncology diagnosis. People with an oncology diagnosis ranged from 1219-1412 minutes sedentary per day. Another study reported, on average, patients with a bone tumour did not leave the bed for > 23 hours/day and 40% of patients with Leukemia reporting leaving the bed for < 1 hour/day [28]. Those with AN diagnosis ranged from 352-862 minutes sedentary per day, one of these studies included a control group and no significant difference was seen between groups (p = 0.118) [24].
LPA was high in three studies [23-25]. One had a control group, LPA was significantly higher in those admitted with AN than the healthy control group (p < 0.001) [24]. LPA ranged from 103-343.4 min per day in these studies. LPA ranged from 28-125 min/day in patients admitted to oncology wards [26,27], with one pre-operative inpatient group reporting 153 min/day [29].
Three of the four studies involving patients with AN measuring a mean MVPA above the WHO recommendations [23-25] vs. Langlet, et al. [21]; overall MVPA ranged from 26.3 to 99.5 min/day in AN. One study compared to a control group, MPA was significantly higher than controls (p = 0.009), however there was no significant difference for time spent in VPA [24]. People with AN self-reported significantly lower MVPA than accelerometer reported data (p = 0.019). People admitted with an oncology diagnosis had an MVPA of 0-4 min/day in one study [26] and 0 min/day of VPA in another study [27], with 20 min/day of MPA. Pre-operatively whilst an inpatient, MVPA was reported as 90 min/day by Ghomrawi, et al. [29].
One study mentioned an overall reduction in PA, though this was not broken down into intensity of PA, in patients with a mental health diagnosis. This was true for both those on antipsychotics and those who were antipsychotic naïve, compared to healthy controls (P < 0.05) [30].
Fidgeting was measured in two studies, one found that people with AN had a significantly higher volume of fidgeting compared to controls (p = 0.003) [31]. Another study reported no change throughout admission in fidgeting in people with AN, they did not directly compare admission to a control but one month following discharge there were no difference to controls [32].
While this review did not look to investigate interventions, there were two interventions of training programmes which fulfilled the inclusion criteria. Both increased PA levels, one in cancer treatment where step count was increased by 265 steps per day [33], the second in people with obesity where the PA level was increased (p < 0.001) and sedentary time decreased (p < 0.001) [34]. Key findings included due to “their frequent interactions with patients, nurses are a primary source of support and guidance about activity in the hospital” [33,35] p.12 and physical activity plays a role in improving HRQOL without substantial change in body composition in CYP with obesity.
Using the Newcastle-Ottowa Scale, three studies were considered “fair” and four were “good”. Using the modified Newcast-Ottawa Scale, one study was considered “satisfactory”, six were “good” and four “very good” (Supplementary File 1). CASP tool results can be seen in Supplementary File 2.
In view of a lack of studies using control or comparison groups, pooled means were calculated where this was possible rather than undertaking a meta-analysis. Due to the clear differences in levels achieved, Physical and Mental health papers have been reported separately. The papers included are stated next to each table (Table 1).
Table 1: Physical activity measures alongside the pooled mean and pooled variance for included studies in the physical health population. View Table 1
Where standard deviations (S.D) were reported, variance was calculated as the S.D 2 . The following equation was used to calculate pooled mean from the study mean and number of participants .
The pooled variance was calculated with following formula using variance :
CYP with a physical health diagnosis averaged 1652 steps, and a total of 34.6 minutes in MVPA compared to 131.04 in LPA and 1295 minutes sedentary (21.58 hours). There is large variance within this data which is likely to represent the large variances in each study. In populations with a mental health diagnosis there was a pattern of more activity with a mean of 210 minutes of LPA and 61.25 minutes of MVPA per day, exceeding the recommendations set out by the WHO. Due to lack of data, number of steps was not able to be assessed. Time spent in sedentary behaviours was considerably less in this population with only 597 minutes (9.95 hours) observed. There again is large variance within this data (Table 2).
Table 2: Physical activity measures alongside the pooled mean and pooled variance for included studies in the mental health population. View Table 2
Matrix for data collection can be seen in Supplementary File 3.
The present review aimed to summarise the existing evidence on physical activity levels of CYP in an inpatient setting. A pooled means analysis was undertaken to answer this aim and to provide a statistical value of PA from this review. It is acknowledged that there is a large, pooled variance within this data, which may limit the precision of the estimated values. However, some trends appear evident. The step count of 1,651.785 in the physical health group is significantly lower than the recommended 10,000-12,000 in the literature; this may well influence health outcomes in the population. Both studies measuring step count in AN reported between 8200 and 8500 steps, with one reporting a significant reduction compared to a matched control. Though there is a reduction compared to controls, this is still a considerably higher step count than in physical health, even with restrictions placed on PA of inpatients with AN. Even when documented as sedentary with wrist/hip accelerometer, patients with AN also demonstrated a significantly higher value of fidgeting compared to controls, so whilst step count is down, there may be a higher amount of skeletal muscle activity and energy expenditure [31]. The difference in PA between physical health and AN groups extended to time in MVPA and LPA, people with AN reached WHO guidelines for MVPA averaging 1.25 minutes higher than the recommended 60 minutes per day, with physical health reaching almost half of this at 34.58 minutes per day of MVPA which may have been skewed higher by pre-operative data used in Ghomrawi, et al. 2018 [29]. People with physical health were also spending over 21.58 hours per day sedentary in an inpatient setting. People with physical health were also spending over 21.58 hours per day sedentary in an inpatient setting. In adult populations we have moved well past “Bed is Best” principles and increased sedentary times is associated with increased length of stay and negative outcomes [36]. The amount of sedentary time should therefore be questioned in this population too. Interestingly none of the physical health papers reported limitations put on the patient’s mobility by the medical teams. We can therefore hypothesise that the sedentary time was not clinically necessitated but may be clinically induced by factors such as health beliefs, patient symptom or environmental factors.
In this field of research, two populations have received considerable attention: individuals with AN and those with an oncology diagnosis. Notably, people with AN displayed higher PA levels and lower levels of sedentary time even when restrictions were placed with an aim to reduce their levels of activity. Conversely, Vancampfort, et al. [30] observed significantly lower PA levels in psychiatrically stable individuals with mental health conditions compared to healthy controls. While PA is recognised as a crucial element of recovery in all these conditions, the desired outcomes differ. Kern, et al. [22] reported a reduction in PA dependence during the inpatient stay for individuals with AN, possibly indicating a normalisation of PA levels as a person's mental health improves. On the other hand, in the field of oncology, studies have sought to enhance PA levels, often utilising training programs and interventions led by nurses, who have frequent interactions with patients and play a pivotal role in this regard [33,34]. Despite successful interventions, PA levels in these populations remain significantly below the WHO recommendations. Additionally, Ambuehl, et al. [37] highlighted the significance of understanding variations in PA intensities, particularly among people with neurological disorders, with different therapies eliciting different intensities of PA. Although nurse involvement in physical activity training programs is an interesting proposition, it raises concerns about staff training, confidence, and the potential diversion of staff from other duties. Such interventions would require assessments of effectiveness and overall impact on HRQoL.
Despite the overall low level of PA in individuals with physical health conditions, two studies demonstrated a significant reduction in PA levels following surgery, with participants failing to regain pre-surgery activity levels even after discharge [29,35]. Complexity of surgery was also associated with decreased PA [35] likely due to potential time on bed rest, levels of pain, medications and number of attachments. Similar trends were observed in other studies where hospitalisation led to a substantial decrease in PA compared to the patients' activity levels at home [26]. Additionally, two studies reported a notable reduction in PA during chemotherapy induction, with one study documenting a 91% decrease [27,30]. This was hypothesised as being due to factors such as fatigue, and health beliefs of parents and caregivers [26].
The reduction in PA upon admission to the hospital setting may partially be attributed to the interventions performed on patients and the resulting secondary side effects. However, the comparison between home and hospital treatment suggests that the hospital environment itself may contribute to the limited PA in individuals with physical health conditions. A number of factors may effect PA in the hospital environment, such as pain, fatigue, attachment to medical devices, and environmental constraints (e.g., lack of access to toys or appropriate seating) [38]. Additionally, time constraints and limited staffing of nurses to encourage play and PA may further affect these levels among hospitalised individuals. Interestingly, pre-operative PA levels in a separate study indicated that the hospital environment might not exert as significant an influence [29]. This finding suggests that factors specific to the surgical or treatment phase may play a more substantial role in determining the decline in PA than the setting itself. This variability in PA documented in home vs hospital setting in oncology and pre vs post-operative PA in an inpatient setting indicates an impact of condition, environment and intervention on PA. Differences in ward set up, beliefs and ideals may play a role in PA, as well as staff or parental beliefs surrounding a condition. What is clear is that quantity and severity of intervention impact PA levels significantly
Although this review did not set out to identify this, the striking disparity in PA levels between CYP with physical health conditions and those with AN emerged as a noteworthy finding in this study. Initially, the focus of this review was centred on PA levels among children with physical health conditions. However, during the literature review, a substantial body of evidence surrounding PA in individuals with AN and its significance became apparent. Understanding the wide array or conditions that CYP can experience, how they interact and how to influence them is key to being a holistic practitioner. Andrew, et al., surmised limited confidence is a major barrier in treating people with chronic mental health issues and whilst most physiotherapists are aware of the role they can play and consider it a part of their job, systemic changes are needed to fill in this training gap [39].
The review identified notable research gaps, including the lack of evidence for specific populations such as those with cystic fibrosis (CF), traumatic or acquired brain injuries, and depression. Addressing these gaps is essential to provide a more comprehensive understanding of physical activity levels in different inpatient populations. The significance of PA in these populations is well known and understanding inpatient PA may allow for more efficacious inpatient stays. Post-operative studies were also lacking, with only two studies with varying populations [29,35]. Further evidence in this area may support a transition to enhanced recovery after surgery programmes, under researched and underutilised in paediatrics but routinely carried out in adults.
The review acknowledged the challenges associated with measuring PA levels, including the use of different assessment tools. The placement of the accelerometer on dominant or non-dominant wrist may have impacted the results between these studies, with the CDC, recommending use of the non-dominant arm to monitor accelerometer data in clinical research [40-42]. These methodological differences can limit the comparability of data and underscore the need for standardisation in future research.
In the studies included in this review, a variety of PA questionnaires were employed, with each of the five studies using a different questionnaire. This practice aligns with the validation of various questionnaires across diverse populations. However, the use of PA questionnaires introduces certain limitations that may affect their comparability to accelerometry data. The heterogeneity resulting from the diverse questionnaires used in these studies poses challenges in comparing and synthesising data across the literature.
The authors acknowledged that a meta-analysis was not conducted due to the absence of control groups in the included studies. This limitation is crucial to acknowledge since control groups are essential for establishing causality and drawing stronger conclusions about the effects of physical activity in an inpatient setting. Most studies were cross-sectional cohort studies, which limit the generalisability of the data, cross-sectional studies provide a snapshot of data at a specific point in time, but they cannot establish causality or account for changes over time. The authors noted that there was varying quality within the study designs and methodologies, which can affect the reliability and validity of the results. Future research should aim for more rigorous study designs to enhance the quality of evidence in this area. In studies with a good rating with regards to risk of bias there were often still several confounding variables, which though documented could often not be accounted for due to the above-described lack of control groups. There was considerable heterogeneity of populations and only limited conditions. The review highlighted that the pooled variance of physical health values are likely skewed due to the high inclusion of oncological diagnosis and pre-operative physical activity levels in the inpatient setting. There is therefore a considerable risk of bias and more work is needed in this area.
The heterogeneity of populations is also a challenge, with only AN and oncology having multiple studies that could compare a specific condition, physical health conditions were grouped but some diagnoses may have greater or worse PA than others. The inclusion of over 18s in the data synthesis may have impacted the overall results, though there was an attempt to mitigate this by only including studies where the mean age was under 18.
1) Further research in other inpatient conditions, including cystic fibrosis and depression, particularly with case-control studies.
2) Creating guidelines on monitoring PA in paediatric patients, especially inpatients.
3) Further evidence on ways to improve PA levels in people admitted with physical health problems.
4) Work directly comparing mental health and physical health PA levels in inpatient environments.
5) The ongoing development, refinement and rigorous evaluation of interventions to promote PA in inpatient settings.
CYP with a physical health diagnosis do not reach WHO guidelines for PA, increasing PA levels should remain a key priority for health care providers. CYP with AN do have a reduction in step count but continue to meet WHO guidelines for MVPA even with restrictions of PA placed upon them. Pathological exercise occurs at both ends of the spectrum between physical and mental health. There are a number of PA measurements which require standardisation, and identifying appropriate, acceptable and widely agreed methods of measuring and promoting PA could open avenues for care improvement moving forwards.
The authors declare that they have no conflicts of interest.
There was no funding for this project.
Mr Kieren Lock: Conceptualised and designed the study; joint led study selection; led data collection; led narrative synethsis; drafted initial manuscript; reviewed and revised the manuscript; and approved the final manuscript as submitted.
Mr Colin Hamilton: Supervised the conceptualisation and design of the study; joint led study selection; supervised data collection; completed statistical analysis; critically reviewed and revised the manuscript; and approved the final manuscript as submitted.
Mr David Young: Critically reviewed and revised the manuscript; supported in study selection; and approved the final manuscript as submitted.