Background: Apnea of prematurity is one of the most common challenges in preterm infants, contributing to the U.S.’s high infant mortality rate. There is a growing need for alignment of remote patient monitoring techniques and at-home care for infants diagnosed with apnea of prematurity, especially in the South where accessibility to health services is lacking.
Methods: A comprehensive literature review of journal articles and research studies published between 2018 and 2023 was conducted through searching reputable databases including PubMed, Google Scholar, Scopus, New England Journal of Medicine, and ScienceDirect. All articles were assessed for relevance and English language. The analysis adhered to a robust set of inclusion criteria to establish credibility, validity, reliability, and transferability.
Results: This analysis revealed health care accessibility challenges associated with living in the South, which contribute to excess infant deaths above the national average. It also revealed that standardized guidelines for apnea of prematurity management through at-home cardiorespiratory monitors are lacking, and the scarcity of clinical and emotional support post-discharge causes parental worry and unnecessary visits to the hospital. Because of the current requirement for meeting with providers in person, excess clinic visits also occur. Additional strategies to improve accessibility and coordination of care are greatly needed.
Conclusion: The further integration of remote patient monitoring into the home setting for infants diagnosed with apnea of prematurity is recommended to reduce costly hospital visits, increase accessibility of care, diminish parental stress, and increase parental emotional support. Future research should investigate how the incorporation of a trained clinical care team into the interpretation of cardiorespiratory monitors provides benefits to infants and their families.
Infant, Apnea of prematurity, Premature/prematurity, Remote patient monitoring, AOP, RPM, cardiorespiratory monitor
AOP: Apnea of Prematurity; BPM: Beats per Minute; NICU: Neonatal Intensive Care Unit; RPM: Remote Patient Monitoring; SIDS: Sudden Infant Death Syndrome; THP: Transition Home Plus
Although infant mortality across the U.S. has declined in recent years, the rate of decline has been slower in the South when compared to the New England and Pacific regions [1]. According to recently available data, an extra 1.2 infants per 1,000 births in the South die when compared with other regions in the U.S., resulting in over 1,600 excess infant deaths each year [2]. Racial and regional disparities exist that contribute to excess deaths for certain races, ethnicities, and locations above the national average. Southern states in particular have consistently higher infant mortality rates than the rest of the country [1], with various social determinants of health widening this gap [2]. Mississippi, for example, had the highest rate of infant mortality in the U.S. in 2020 at 8.12 per 1,000 live births. Louisiana had the second highest rate of infant mortality in 2020, as evidenced by 435 infants dying before the age of one and a corresponding death rate of 7.59 per 1,000 live births [3].
Preterm birth complications are the leading cause of death among children under 5 years of age [4]. In 2019, 16.5% of all infant deaths in the U.S. were a result of prematurity and/or resulting low birthweight [5]. Among these preterm infants, the most common cause of death was respiratory distress or failure [6]. One example of a respiratory complication, apnea of prematurity (AOP), occurs when the newborn pauses breathing for more than 15 to 20 seconds, or pauses breathing for less than 15 seconds and has a slow heart rate or low oxygen level [7]. Combined with hypoxia and/or bradycardia, AOP is one of the most common and recurrent problems in preterm infants, due to their underdeveloped respiratory systems [8]. Because AOP is associated with respiratory immaturity and leaves infants susceptible to decreased oxygen levels in the blood and slow heart rate, AOP poses a significant risk for respiratory failure and death [9].
There are three main categories of apnea: central, obstructive, and mixed. Central apnea accounts for approximately 10%-25% of all cases and involves complete cessation of chest movements and airflow [10]. Obstructive apnea accounts for another 10%-25% of all cases and specifically refers to restriction of airflow, with or without respiratory effort [10]. Finally, mixed apnea is the most common, accounting for 50%-75% of all cases, and it includes components of both central and obstructive apnea [10].
Discharge of the infant after an apnea-free interval of 2-10 days is accepted, although this timeframe is widely debated among physicians [11]. In most instances, the infant will arrive home without the need for additional AOP monitoring, but in more severe cases, parents must monitor AOP through the use of an at-home apnea monitor. High risk infants include those who underwent apparent life-threatening events, those with siblings who died from sudden infant death syndrome (SIDS), and premature infants who are still mildly or moderately symptomatic but otherwise ready for hospital discharge [12].
In both the neonatal intensive care unit (NICU) and home environments, monitors serve the purpose of alerting the caretaker to potentially life-threatening events [13]. Apnea monitors are comprised of a belt with sensory wires worn around the infant’s chest, as well as a monitoring unit with an alarm, which sounds if changes in the infant’s breathing or heart rate are detected [7]. Once the alarm sounds to alert the parents to a possible AOP episode, tactile stimulation may be required, and the cardiorespiratory monitor will record the event in an electronic log [14]. While the monitors record each AOP event electronically, parents must also manually document details, such as visible signs of distress or color change [7,15]. This reliance on manual logging and interpretation can cause undue stress and excess mental burden for parents [15].
Remote patient monitoring (RPM) allows for providers to monitor a patient’s health status remotely and away from a traditional clinical setting. Specifically, RPM harnesses the power of digital technologies to collect health data from patients and electronically transmit health information directly to the provider for review and assessment [16,17]. Smartphones, wearable devices, and other computerized systems are methods of data collection and transmission in a noninvasive manner, all of which reduce patient burden and human error associated with manual data entry [16]. As technology advances, the hope is that pediatric RPM will become more automated and integrated into the clinical care setting overall.
This review aims to provide a set of recommendations for further integration of RPM into the care of infants diagnosed with AOP discharged to home in the southern U.S., where accessibility of clinics is lacking.
This review uses existing, current, and peer-reviewed research. A comprehensive literature review and established search strings assisted in identifying articles for inclusion that were evidenced-based and community and culturally appropriate, including: PubMed, George Washington University’s Himmelfarb Health Sciences Library, Google Scholar, Scopus, New England Journal of Medicine, and Science Direct. This review also consulted outside sources of infant-specific experts, including National Institutes of Health’s Eunice Kennedy Shriver National Institute of Child Health and Human Development, American Academy of Pediatrics, and Children’s Hospital Association.
In order to conduct informed and precise searches, search strings included terms related to AOP, RPM, health information technology, home monitoring, infant supervision, rural locales, cardiorespiratory waveforms, and others (Table 1). This review excluded articles that failed to mention AOP or premature infants, as well as studies that focused on countries outside the U.S. This review also excluded articles about AOP treatment, including caffeine therapy, supplemental oxygen inhalation, and positive airway pressure, since treatment with stimulants is outside the scope of this analysis. Additionally, only articles in English were included, and precedence was given to articles published within the last five years (2018-2023), although articles outside of this timeframe were considered depending upon applicability.
Table 1: Full search criteria. View Table 1
After a preliminary screen of title and abstract to indicate relevance, the initial literature review yielded 225 articles. Supplementary searches were conducted as the analysis took place to identify articles outside of the predetermined search strings. 36 articles were excluded due to duplication and 9 were excluded due to accessibility challenges and non-English language. The text was then consulted more fully to determine relevance, where 136 articles were eliminated. 44 articles remained for inclusion (Figure 1), 16 of which were discovered through the search strings (Table 2).
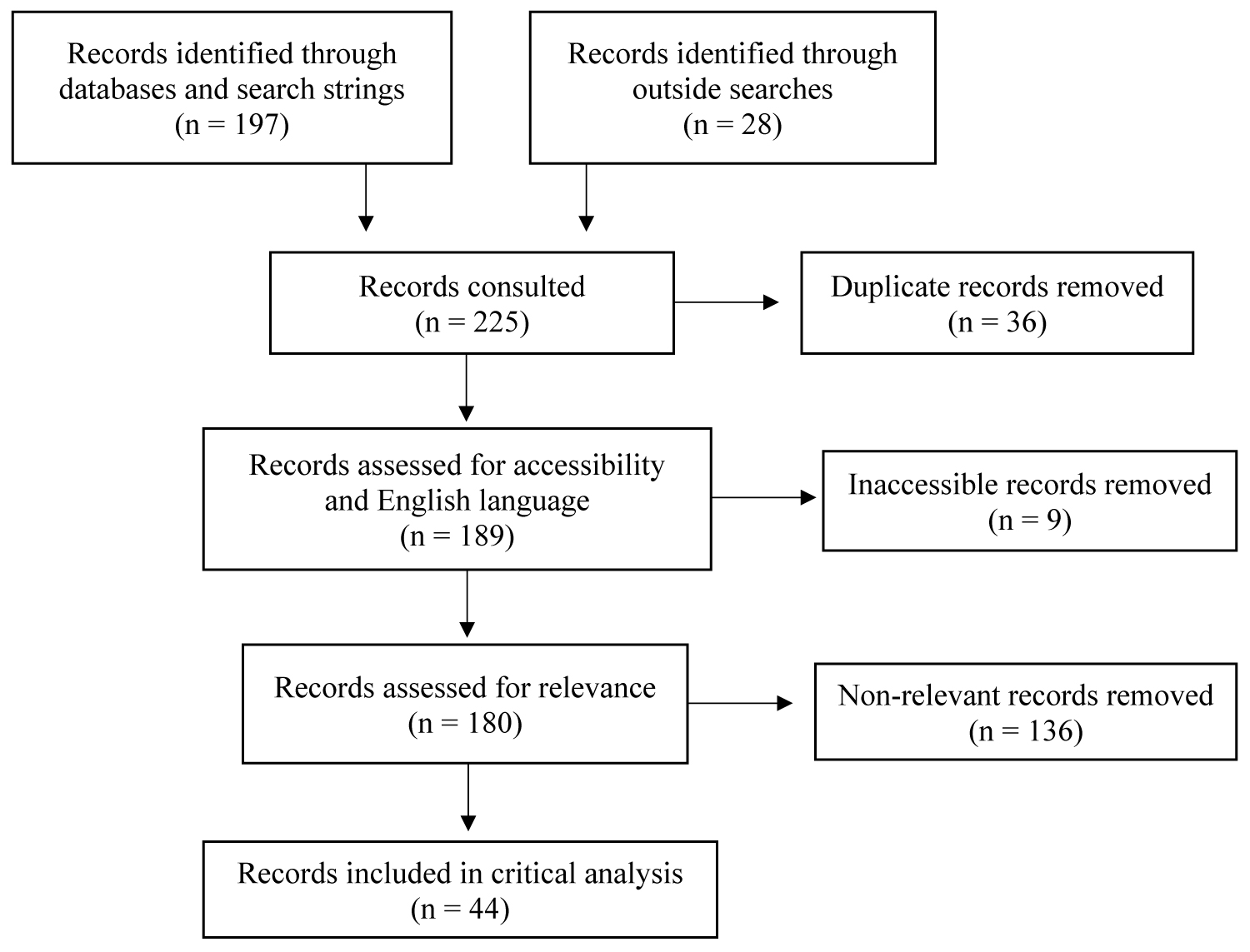 Figure 1: Articles yielded for literature review - This figure shows the flow for how all articles were systematically included and excluded.
View Figure 1
Figure 1: Articles yielded for literature review - This figure shows the flow for how all articles were systematically included and excluded.
View Figure 1
Table 2: Literature review articles. View Table 2
The majority of articles included were evidence-based research studies and journal articles focused on infant mortality, southern and rural health outcomes, AOP, home monitoring, cardiorespiratory monitors, RPM, and care coordination for NICU infants. Randomized trials, cohort studies, evidence-based programs and interventions, survey research, and quasi-experimental studies were consulted. All records were peer-reviewed, set in the U.S., and adhered to the following criteria: credibility, dependability, confirmability, transferability, and reflexivity criteria for qualitative studies [18] and validity, reliability, and generalizability for quantitative studies [19].
Infant mortality is caused by a variety of factors, but the leading cause of mortality for infants under the age of five years is prematurity [20]. As such, AOP remains one of the most common diagnoses in the NICU [21], with AOP emerging as a significant predictor of neonatal death [22]. Because of the frequency with which NICU infants are diagnosed with and die from AOP and AOP-related complications, targeting interventions towards infants diagnosed with AOP can serve as a meaningful way to reduce infant mortality overall.
Lower socioeconomic status and lack of access to high-quality maternity care and clinics are two major factors that contribute to high infant mortality in the rural South [23-26]. In the Deep South specifically, these factors play a major role in creating and sustaining child health disparities [27]. Given the South’s disproportionately high poverty rate and low median incomes compared to the rest of the country [28], mothers and families living in this region can experience a concentrated lack of resources and increased economic distress. This lack of stable income can affect families’ ability to afford necessary medical care and health services, and the high poverty rate means that there are few opportunities for funding medical improvements and interventions. Educational attainment is also lacking in the South, which contributes to challenges in promoting positive health and wellbeing, further perpetuating the cycle of socioeconomic hardship [28].
Rural poverty is unique in that southern states are historically agricultural, so basic infrastructure including clinics and running water may be lacking [28]. Specifically in the rural South, there are insufficient numbers of obstetricians, as women in areas of Alabama reported having to travel up to 70 miles to deliver at a hospital [29]. This barrier means that mothers may not seek out proper prenatal and maternal care, and infants may not receive adequate or timely medical attention.
While region of residence is statistically related to children’s health measures and is among one of the most powerful predictors of child health outcomes, very few policies identify Southern children as a high-risk group [27]. The challenges associated with accessing care for this demographic, therefore, have persisted and worsened over time. It is imperative to focus on interventions that take lack of brick-and-mortar buildings and socioeconomic strain into account when establishing recommendations for this population.
Although manual data collection is the most widely used method in pediatric care today, it has the greatest potential for error [17]. In fact, a 2013 American Journal of Perinatology study recorded that only 7.7% of apnea events recorded by nursing documentation were confirmed as true apnea when compared to 50.4% of apnea recorded by monitors [30]. As such, home monitors play a significant role in identifying AOP complications. Studies have shown that an overwhelming majority of preterm infants who have persistent AOP or other respiratory complications at the time of discharge will likely continue struggling with AOP at home [13]. In an Advances in Neonatal Care study, it was revealed that 78% of discharged preterm infants experienced cardiorespiratory events, with 31% of preterm infants experiencing events classified as extreme [31]. While data from event-recording monitors are crucial, it should serve as a supplement to visual observation [32], as the most accurate results occur when combining data with the expertise of trained clinicians.
Clinicians often struggle with the decision to discontinue cardiorespiratory monitoring before discharge or prescribe home monitoring for continued surveillance [13]. Because there is no standard definition of what duration of apnea classifies as clinically significant [13], the decision to discharge infants diagnosed with AOP also varies by geographic location and hospital preference. There are also vast irregularities with nursing and electronic documentation policies by state [33]. For example, monitors in California use a heart rate threshold of 80 beats per minute (bpm), whereas Massachusetts sets their alarms to 100 bpm [34]. These discrepancies introduce uncertainty for the infant and family at onset, further contributing to the ambiguity surrounding infant care.
Although monitors contain memory capabilities to document episodes [13,14], parental inexperience with monitor operation and confusion regarding interpretation of electronic logs means that a great deal of apnea episodes may be addressed in an inefficient manner. For example, parents may underreact to serious complications that should require clinician contact, or they may overact and perpetuate avoidable hospital admissions. Even if there is an agreed-upon approach to documenting and charting, human variation in manual entries makes discharge decisions and care coordination extraordinarily difficult [33]. This lack of clarity is true not only for nurses [35], but also for parents who adopt a clinical role in the at-home care of their infant.
The speed and preciseness with which parents must respond is demanding, especially given the fact that guidelines are not standard across hospital systems. Clinicians should therefore be mindful to provide parents with instructions for how to operate and interpret the monitor and act in an appropriate manner [13,15]. In all cases, parents are encouraged to document narrative information and severity of the intervention separately in a logbook. Examples of notes include: “light tactile stimulation”, “significant tactile stimulation”, or “cardiopulmonary resuscitation” [15]. The act of documenting is important because it provides context to supplement the episode data. The trouble, however, exists in the delay between the time that the event is documented and the time that the physician is seen. Especially for residents in the South, travelling to these appointments can be time consuming and costly [29]. While visits to the primary care physician or pediatrician are still recommended, appointments for the explicit purpose of facilitating this logbook information exchange could be spared.
In addition to the added responsibility of caring for an infant with respiratory complications, the uncertainty regarding monitor interpretation may cause parents to endure excess worry and negative impacts on family life [13]. The financial stress following a prolonged period of NICU hospitalization may also cause excess mental distress, as the amounts of partner, family, and community support vary greatly [36]. Consequences of this mental suffering are far-reaching, as caregiver burnout can negatively impact maternal recovery and wellbeing; Irritability and extreme fatigue can contribute to delays in maternal post-partum recovery, maternal role gratification, and role attainment [37]. Continued social and clinical support is therefore necessary to keep parental anxiety and depression at bay.
A concrete support system, however, is neither provided nor facilitated by hospitals. While some external clinics offer periodic evaluations when arranged, the majority of NICUs do not have their own follow-up clinics [14]. In general, only a small number of outpatient clinics provide a full workup of follow-up services. Separate subspecialty appointments are instead scheduled, and the responsibility for coordination of care typically falls upon the primary care physicians [14]. This lack of continuity places the successful transition home in jeopardy, as consistency for the infant and family is essential for positive health outcomes [36].
Through continuous monitor data and self-upload of patient data, infants’ cardiorespiratory data can be automated and sent to a care center for analysis [38]. The transmission of data to a qualified care team can allow for more timely and accurate analysis of data and more prompt responses, as immediate contact can be facilitated via internet, phone call, and/or text message [38]. In addition to allowing for quicker response times, RPM upholds data interpretation accuracy through its collaboration with qualified specialists and subspecialists. A Garingo, et al. study compared the quality of care received through RPM to care received by onsite physicians and discovered that neonatologists who were able to physically examine patients in the NICU had “good or excellent agreement for most assessments of patients” when compared with remote physicians [39]. This agreement demonstrates alignment in interpretation, communication, and execution of remote clinical strategies.
Integration of a clinical care team is not a new concept for helping families transitioning home post-NICU. One successful example of this enhanced support is the Transition Home Plus (THP) program, which connects families to resources such as social workers and family resource specialists [40], working in tandem with the medical team to ensure families settle in well at home. The THP evaluation found that infants in the intervention group had significantly lower Medicaid spending, fewer emergency department visits, and fewer readmissions to the hospital than the comparison group [40]. In fact, during the 90-day period where the intervention group infants were followed, they experienced a quarterly Medicaid savings of $4,591 per infant [40]. This example showcases the psychological and monetary benefits associated with utilization of a broader care team post-discharge, for which support is often lacking.
Underscoring the importance of a care team is RPM’s ability to connect infants with physicians more frequently. In fact, RPM has shown to decrease the average number of days that patients spend without contacting a physician, lessening the possibility of an undetected medical complication remaining unaddressed [38]. Infants on a 6-month follow-up regimen could experience a temporal gain of 134 days of physician contact, and infants on a 3-month follow-up timeline could gain 44 days [38]. In situations such as these, however, reliable technology and internet connectivity is crucial for success, which may prove to be challenging for patients without access to the internet.
By allowing physician interactions to be conducted remotely and away from the traditional clinical setting [38], RPM also decreases unnecessary hospitalizations. Benefits noted in a Leoni, et al. study included an average of 72 prevented visits to the emergency department for infants with implantable cardiac devices [41]. The electronic transmission of data also decreased the median time between data gathering and viewing by a trained professional, and it procured a more accurate interpretation of infants’ cardiorespiratory data [38].
Another challenge faced by rural parents with apneic infants is the time and cost associated with traveling to their physicians. RPM, however, eliminates the need for these visits and replaces them with more efficient electronic communication to qualified cardiac specialists [42]. A Leoni, et al. study included an average savings of 2 hours and 35 minutes of transportation time [41], and a Respiratory Care study found that a team-based monitoring system has the potential to save anywhere between $62 and $2,422 per infant [43], as the clinical care team provides guidance and reassurance for families before they arrive at the hospital. Enhanced support and care coordination for the infants and their families is therefore extremely beneficial not only to the families themselves, but also to the larger healthcare system at large.
Finally, there is a psychological benefit for parents, as their quality of life and emotional wellbeing can increase once they are in their own homes with additional support [20]. When integrated through RPM strategies, securing a trained and qualified care team can provide a sense of reassurance and security for parents and families. During a time of great uncertainty and worry, parents may feel overwhelmed by the thought of operating a new piece of technology. A 2011 Clinical Cardiology study, however, found that 87% of patients and families who underwent RPM rated the process as “very easy to perform” [42]. This statistic is especially important because parental buy-in and ease of use is critical to RPM’s successful implementation. It is therefore clear that a team-based approach can contribute to better health outcomes for infants and families alike.
The classification of improvement in infant mortality as a Healthy People 2030 objective coupled with telehealth’s recent rise in popularity makes the current policy landscape for integrating RPM favorable. Especially in the wake of the COVID-19 pandemic, the benefits of telehealth are even more apparent, as it allows for convenient and timely communication, increased accessibility that can overcome geographic barriers, and burden reduction for medical infrastructure and workforce [44]. The health landscape has forever changed to allow for more convenient forms of health management, and RPM is a logical next step.
Overall, further integration of RPM into the at-home care of infants diagnosed with AOP is recommended to provide increased quality and efficiency of care, essential process improvements for parents and caretakers, and reductions in unnecessary hospital admissions and infant mortality. In addition to being more convenient for southern communities, RPM is also less costly to the families, hospitals, and healthcare system overall. Parental worry and fatigue can also improve as parents receive vital support from a clinical care team, all in the comfort of their own homes.
Although the breadth of literature identified was considerable, accessibility and payment restrictions emerged as obstacles for certain journal articles. It must also be noted that zero studies reported precisely on this topic, so direct transferability may not hold without additional considerations. While technology solves physical accessibility limitations, it also establishes new connectivity challenges. Reliable broadband connection is vital to transmitting information electronically, but households of lower socioeconomic statuses may not have access to high-speed internet [44]. Additional solutions for implementing wireless RPM strategies may need to be considered.
No acknowledgements to list.
Self-funded by the study author.