Background: Peripheral T-cell lymphomas (PTCLs) are a rare and heterogeneous group of aggressive malignancies derived from mature (postthymic) T lymphocytes. Currently, there are no published data describing the clinical characteristics and outcomes of Algerian patients with PTCL.
Objectives: To describe the clinical characteristics and treatment outcomes of patients included in an Algerian PTCL registry.
Methods: Multicenter, non-interventional, cohort registry study of all patients with a diagnosis of PTCL attending four specialist oncology clinics in Algeria. For patients newly diagnosed after the study initiation date (June 2017), data were collected prospectively for 2 years, whereas for existing patients, retrospective data on diagnosis and treatment were obtained from medical records, and these patients were then followed prospectively for 2 years.
Results: A total of 60 patients were recorded in the registry. The majority were male (56.7%) and mean age at diagnosis was 55.1 years (range 21.2 to 82.1). Sixty percent had at least one comorbidity at diagnosis. At diagnosis, the majority of patients already had advanced disease (81.3% in stage 3 or 4). The most frequently reported PTCL types were nodal (45.8%) and leukemic (20.0%), and most frequently reported subtypes were adult T-cell leukemia/lymphoma stage 4 (16.9%) and peripheral TCL - not otherwise specified (NOS) stage 4 (13.6%). The most commonly used first-line treatments were CHOP-21 (18/44; 40.9%), CHOEP-21 (11/44; 25.0%) and COP (5/44; 11.4%). Compared with baseline, white blood cell, neutrophil and platelet counts were significantly lower at the final visit (p = 0.001, 0.043 and 0.017, respectively). Median progression-free survival (PFS) was estimated at 10.8 months (95%CI 5.3-16.3) and median survival was 45 months, with no significant differences between PTCL subtypes. One-year and 3-year survival were 57.3% (95%CI 43.7%-70.9%) and 49.3% (95%CI: 34.9%-63.7%), respectively.
Conclusions: In Algeria, the majority of patients presenting with PTCL are young, and they present with advanced disease. The prognosis is poor.
Peripheral T cell lymphoma, Registry, Cancer, Chemotherapy, Outcomes
Peripheral T-cell lymphomas (PTCLs) are a rare and heterogeneous group of aggressive malignancies derived from mature (postthymic) T lymphocytes, and which arise in peripheral lymphoid tissues such as lymph nodes, spleen, gastrointestinal tract and skin [1]. Based on various factors, including clinical presentation, morphology, immunohistochemistry, flow cytometry, cytogenetics and molecular biology, PTCLs are divided into different subtypes, which include cutaneous, extranodal, nodal and leukemic forms [2,3].
The incidence of PTCL is geographically variable and, even in the same country, the incidence of, and survival with, PTCL subtypes differs among different racial and ethnic groups [4-6]. In a large international survey, including several European countries, South Africa and the USA, PTCLs comprised between 5% and 10% of all non-Hodgkin lymphomas (NHLs) [5]. In Europe and the USA, the most common subtype is PTCL not otherwise specified (NOS), which accounts for approximately one third of PTCL cases [6]. However, in Asia, Africa and other parts of the world, the incidence of PTCL may be higher and, at least in part because of exposure to endemic oncogenic viruses, such as the Epstein-Barr virus (EBV) and the human T-cell leukemia virus-1 (HTLV-1), different subtypes predominate [3,5,6]. In Hong Kong, 18% of NHLs were a PTCL [5]. In a registry study including patients with PTCL from 9 Asian countries, extranodal natural killer/T-cell lymphoma (NKTL) and angioimmunoblastic T-cell lymphoma (AITL) were the most common forms, together accounting for 52% of cases [7]. The International NHL Classification Project showed that in Algeria, although the overall frequency of PTCL was similar to those in Western Europe and North America, NKTL accounted for 3% of NHLs, in comparison to 0.5% and 0.0% in the latter two countries, respectively [8].
With the exception of anaplastic large cell lymphoma (ALCL) and anaplastic lymphoma kinase (ALK)-positive subtypes, patients with PTCL have a poor prognosis [9-11]. The most commonly used first-line chemotherapy strategies are CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) and CHOP-like regimens, which have remission rates of 50-65% [10]. However, approximately 75% of patients with T-cell lymphoma will relapse after initial therapy, which is associated with a poor outcome [11]. Five-year overall survival rates with CHOP range from 32% to 43% for PTCL-NOS and 32% to 36% for AITL [9,10]. Data from the Swedish and Czech Lymphoma Registries showed that addition of etoposide to CHOP (CHOEP) improved progression-free survival (PFS) in patients younger than 60 years [12,13]. However, data are contradictory, and treatment outcomes may also differ by ethnicity or racial group. In an analysis of data from the Korean National Health Insurance Service and National Cancer Registry, in comparison to no etoposide, the addition of etoposide to CHOP or a CHOP-like regimen did not improve OS or PFS, but was associated with longer hospitalisations and cytopenias requiring more transfusion [14].
Retrospective data from the Swedish Lymphoma Registry demonstrated that up-front auto-stem cell transplantation (SCT) might improve overall survival (OS) and PFS in patients with nodal PTCL and enteropathy-associated T-cell lymphoma (excluding anaplastic lymphoma kinase-positive ALCL) [12]. In the International T-cell project (a prospective cohort registry study) patients with PTCL who were refractory to treatment or who relapsed after initial therapy had an overall survival of 5.8 months, and 3-year survival rates were 21% and 28%, respectively [10]. However, 3-year survival rates were increased to 48% among patients who received and responded to salvage haematopoietic cell transplantation.
In the absence of sufficient randomized controlled studies to guide practice, observational studies and population-based registries with long follow-up can help to characterize patients with PTCL in different regions of the world and identify which treatment strategies might be more effective in specific patient populations. Data from several local and international registries have described the clinical presentation and outcomes of treatment in patients with PTCL in Europe, Asia and the USA [6,7,9,10,12-23].
However, to the best of our knowledge no data specific to Algeria has been published thus far. The purpose of this cohort PTCL registry study was to gain a better understanding of the clinical characteristics of PTCL patients in Algeria, current approaches and responses to first and second-line therapies, and to compare those with data from other international registries.
This was a multicenter, non-interventional, cohort registry study of all patients with a diagnosis of PTCL attending four specialist oncology clinics in Algeria (Etablissement Hopitalier Universitaire d'Oran, Centre Hopital Universite Annaba Ibn Rochd, Centre Hospitalo-Universitaire Benimessous Hospital, and Centre Hospitalo-Universitaire Frantz Fanon de Blida). The registry comprised retrospective and prospective data. For patients newly diagnosed after the study initiation date (June 2017), data were collected prospectively for 2 years, whereas for existing patients, retrospective data on diagnosis and treatment were obtained from medical records, and these patients were then followed prospectively for 2 years to obtain data on current disease status and treatments. Inclusion criteria were male or female ≥ 18 years of age and previous or new diagnosis of pathologically confirmed PTCL according to World Health Organization classification [2]. Patients with current psychiatric illness that would interfere with their ability to comply with protocol requirements or give informed consent to enroll in the prospective data collection portion of the study were excluded. Patients with confirmed PTCL were assessed as per local practice, and all treatment decisions, type, and timing of disease monitoring were at the discretion of the treating physician.
The protocol (ONC15-MEA-401) was reviewed and approved by institutional/independent ethics committees and complied with recommendations of the 18th World Health Congress (Helsinki, 1964) and all applicable amendments and with the laws and regulations, as well as any applicable guidelines of Algeria. All patients included in the study provided informed consent and were free to withdraw from the study at any time.
The primary objectives of the study were to describe the frequencies of different types of PTCL and to understand current treatment patterns (first and second line treatments) for PTCL patients in routine real-world clinical practice. Secondary objectives were to (1) Describe clinical characteristics of PTCL patients at the time of diagnosis and at the last visit; (2) Assess response rates to 1st line and 2nd line treatments; (3) Assess the tumor related characteristics on response to primary and subsequent treatment; and (4) Compare the clinical characteristics at diagnosis and relapse/progression and population-level mortality trends in Algeria with other registry data.
Continuous variables were summarized using means and standard deviations or medians and ranges or interquartile ranges, as appropriate. Categorical data were summarized as numbers and percentages. Percentages were calculated according to the number of patients for whom data are available. Where values are missing, the denominator, which will be less than the total number of participating patients, was reported in the summary table. Chi-square test or Fisher's exact test were used to compare categorical data and demonstrate differences between groups. Continuous data were compared using unpaired or paired Student's t-tests and one-way analysis of variance (ANOVA) or two way ANOVA for data measured at one point in time or repeated measurements, respectively. Non-parametric tests were applied where the assumptions underlying the Student's t-test or ANOVA were not satisfied. Pearson's and Spearman's correlation coefficients were used to study the strength of association between two continuous variables, depending on whether they were distributed normally or not. All statistical tests used a significance level of α = 0.05. Two-tailed tests were performed for all analyses that use statistical testing. Confidence intervals were presented with a 95% degree of confidence.
Time to progression was computed as the time from date of diagnosis to the date of progression for those who progressed, or date of death for those who did not have a record of progression, but had a record of death, or date of the last contact for those with no records of either progression or death. The latter group was considered censored. Kaplan Meier methods were used to obtain the median progression-free survival time, along with its 95% confidence interval (95%CI). Log-rank test, along with Cox regression, was used to compare progression-free survival time between the different PTCL types. The log-rank test was used to study differences in the estimates of endpoint events over time in relation to different treatment protocols and other prognostic factors that could affect survival. Cox proportional hazards regression model were applied to analyze the relationship between different treatment protocols and other prognostic factors and the estimates of the study endpoints. The Cox regression model was used to measure the effect of treatment protocols and each of the prognostic factors on the risk of failure after controlling for the effect of the all the other factors in the model. The assumption of proportional hazards was assessed by the visual judgment of the log-minus-log survival plots. Software Data management and analysis, and figures were performed using Statistical Package for Social Sciences (SPSS) vs. 23.
Between 4 June 2017 and 7 August 2018 a total of 60 patients were included in the registry. Fifty (83.3%) were existing patients for whom data on diagnosis and treatment were obtained from retrospective review of clinic records and prospective data were collected on current disease status and treatments. Ten patients (16.7%) were newly diagnosed and were followed prospectively for two years.
Demographics and disease characteristics at diagnosis are shown in Table 1. The majority of patients were male (56.7%) and mean age at diagnosis was 55.1 years (range 21.2 to 82.1). Almost two thirds were Caucasian, whereas slightly less than one third were Black. The most common PTCL type was nodal (45.0%), followed by leukemic (21.7%), cutaneous (15.0%) and extranodal (13.6%). Overall, at diagnosis, the majority of patients already had advanced disease, with 16.9% in stage 3 and 64.4% in stage 4. Approximately 90% had Eastern Cooperative Oncology Group (ECOG) performance status ≤ 2 and the median ECOG performance status was 1. The most common PTCL subtypes were PTCL-NOS (20.3%), ALCL (ALK+/-) (16.9%) and adult T-cell leukemia/lymphoma (16.9%) (Table 2). There were no statistically significant differences in sex, race or age at diagnosis between the different subtypes of PTCL. In each subtype, the majority of patients were in stage 4 at diagnosis, except for those with NK/TCL nasal type or primary cutaneous CD30 positive T-cell lymphoproliferative PTCL, of whom 20% and 0%, respectively, were in stage 4 (p for NK/TCL nasal type vs. other subtypes = 0.01). All 4 of the patients with primary cutaneous CD30 positive T-cell lymphoproliferative PTCL had an ECOG performance status of 0 (p = 0.006).
Table 1: Demographics and disease characteristics at diagnosis (N = 60). View Table 1
Table 2: PTCL subtypes disease stage. View Table 2
Data relating to comorbidities were obtained at the first prospective visit. Thirty six of the 60 patients (60%) had one or more comorbidity, of which the most frequent were diabetes mellitus (11/60; 18.3%), hypertension (9/60; 15.0%), arterial hypertension (4/60; 6.6%), asthma, herniated disc and neutropenia (2/60; 3.3% each). Other comorbidities were limited to 1 patient each.
At diagnosis, mean (SD) systolic blood pressure (BP) was 119.2 (22.5) mmHg and mean diastolic BP was 73.9 (29.4) mmHg; mean body weight was 70.9 kg and mean body mass index (BMI) was 24.4 kg/m2. These parameters did not change significantly between baseline and the final visit. General examination was unremarkable in the majority of patients both at baseline and final visit, and there were no significant differences in any of the variables between these two assessments. At baseline, the most common documented abnormalities were enlarged lymph nodes in 11 out of 36 patients and dermatological signs or symptoms in 9 out of 37 patients.
Results for hematological, hepatic and renal laboratory investigations are shown in Table 3.
Table 3: Haematological and laboratory data at baseline and last observation. View Table 3
In comparison with baseline investigations, haemoglobin, haematocrit, and red blood cell, lymphocyte, monocyte, basophil and eosinophil counts were not significantly different at the final visit. However, white blood cell, neutrophil and platelet counts were significantly lower at the final visit (p = 0.001, 0.043 and 0.017, respectively).
In comparison with baseline, at the final visit there were significant increases in aspartate aminotransferase (AST) and total bilirubin (p = 0.018 and 0.001, respectively), whereas changes in alanine aminotransferase (ALT) and alkaline phosphatase (ALP) did not reach statistical significance. Serum creatinine level remained stable.
In the majority of patients serology was not done or not reported either at the baseline retrospective visit or first prospective visit. Two patients had results for human T-lymphotropic virus (HTLV)-1, viral capsid antigen antibody (VCA) immunoglobulin G (IgG) and Epstein Barr nuclear antigen (EBNA). Both were negative for HTLV-1 and VCA IgG. One patient tested positive for EBNA, whereas the other's results were negative.
Data about first line treatment at baseline visit were available for 44 patients. At this visit, the most commonly used first-line treatments were CHOP-21 (18/44; 40.9%), CHOEP-21 (11/44; 25.0%) and COP (5/44; 11.4%). Linker regimens and mini CHOP were first line therapies in 2 (4.5%) patients each and 1 patient (2.3%) each received bendamustine, VCAP, cyclophosphamide or methotrexate. One patient did not receive treatment. Although there were some observed differences between the different PTCL types in selected baseline regimen, the only one that was statistically significant was more frequent use of Linker regimens for lymphoblastic T-lymphoma as compared to other types (p = 0.048). Other differences did not reach statistical significance.
Documented lines of treatment (from first-recorded treatment) and outcomes for all 60 patients are shown in Table 4 and a full description of each chemotherapy regimen is listed beneath the table.
Table 4: Lines of treatment and outcomes. View Table 4
Median progression-free survival (PFS) was estimated at 10.8 months (95%CI 5.3-16.3) (Figure 1). Although some differences were observed between types of PTCL (Figure 2), they did not achieve statistical significance, and the small sample sizes in some of the groups precluded estimation of median survival times.
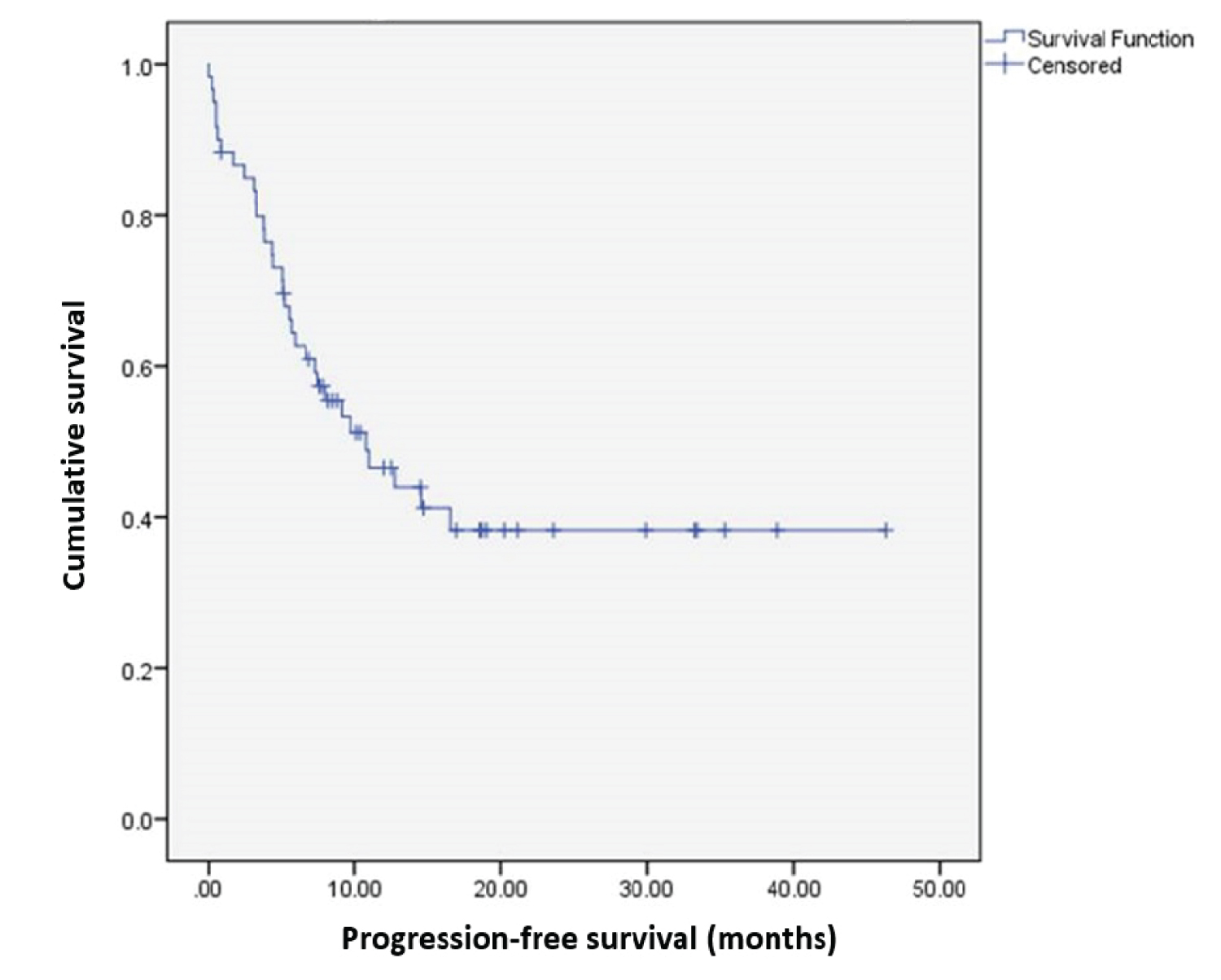 Figure 1: Progression-free survival in the overall population. View Figure 1
Figure 1: Progression-free survival in the overall population. View Figure 1
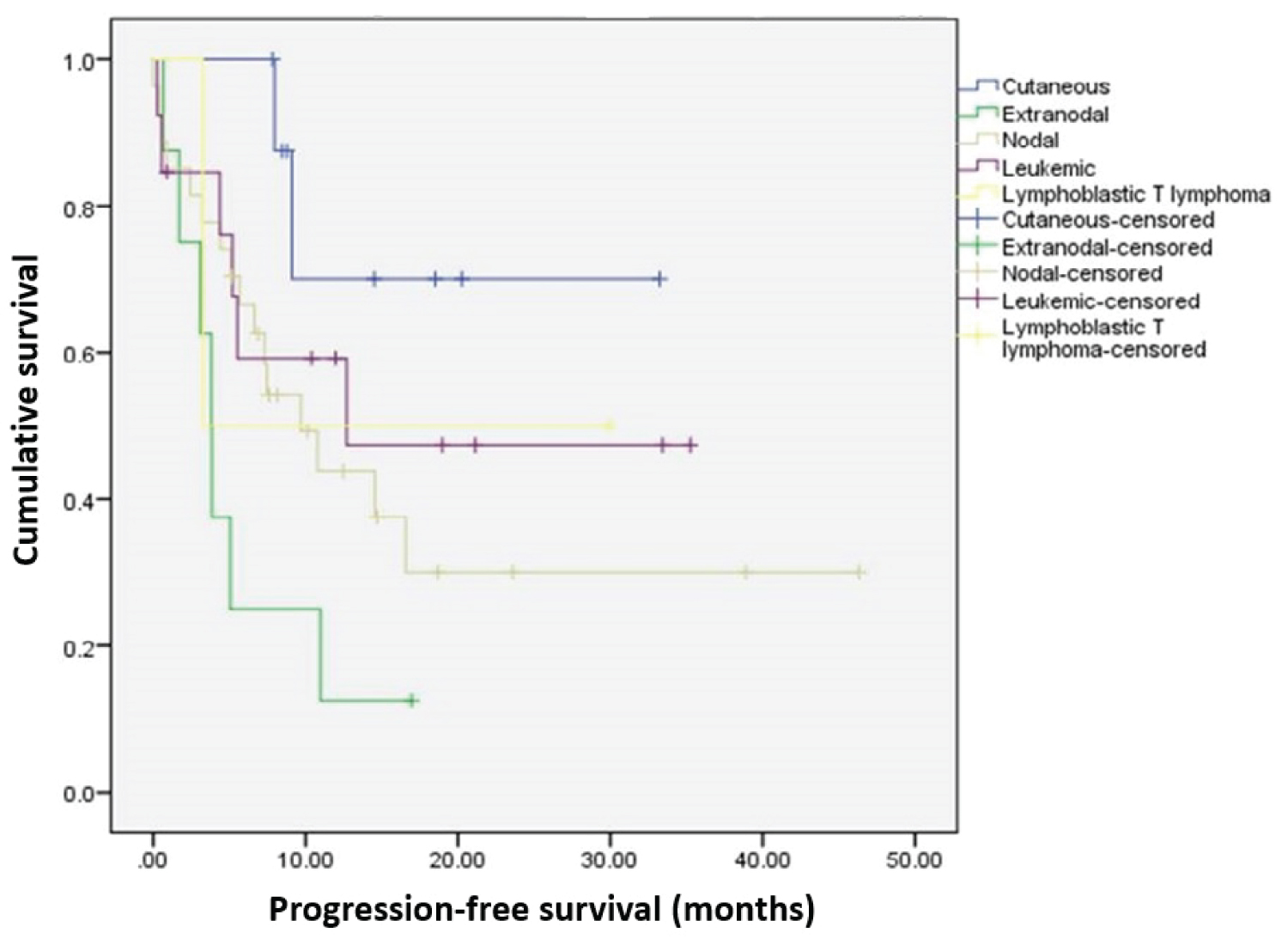 Figure 2: Progression-free survival by PTCL subtype. View Figure 2
Figure 2: Progression-free survival by PTCL subtype. View Figure 2
At last observation, 45% of the patients had died and 1.7% were lost to follow up. Although there were differences between subtypes in terms of mortality, they did not reach statistical significance due to the small sample size in each subtype (p = 0.891). Median survival was 14.5 months. Due to the large amount of censoring (55%), it was not possible to estimate confidence intervals. Median survival did not differ between PTCL types (p = 0.167 for log-rank test and p = 0.168 for Cox proportional hazard mode). Although due to the small sample sizes, median survival for individual subtypes could not be estimated, the log-rank test showed that there was no significant difference in the survival distribution between subtypes (p = 0.263 for log-rank test and p = 0.266 using Cox regression).
One-year and 3-year survival were 57.3% (95%CI: 43.7%-70.9%) and 49.3% (95% CI: 34.9%-63.7%), respectively.
In this Algerian cohort of patients with PTCL, slightly more than half were male and the mean and median ages at diagnosis were 55.1 and 54.9 years, respectively. The most common PTCL types were nodal (45%) and leukemic (22%) and the majority of patients were in stages 3 or 4 at diagnosis. Although there was a wide spread of PTCL subtypes, the most common were PTCL-NOS (20%), anaplastic large cell lymphoma (ALCL) (17%), adult T-cell leukemia/lymphoma (ATLL) (17%), NKTCL nasal type (9%) and AITL (9%). Serology data were unavailable for all but two of the patients and no conclusions can be made about possible oncogenic co-factors that might influence the subtype distribution.
Baseline clinical examination was unremarkable except for documentation of enlarged lymph nodes in 11/60 patients (18%) and dermatological signs or symptoms in 9/60 (15%). Median values for hemoglobin and red blood cells indicated that anemia was common at baseline and also at the last observation. Of the 59 patients who had documented treatment, approximately two thirds received either CHOP or CHOEP as the documented first-line treatment regimen, and overall, there were no statistically significant differences between the different PTCL subtypes in selected baseline regimen. Approximately half of the patients (29/60; 49%) received a second-line treatment and 8 (14%) received third-line treatment. CHOEP-21 (6/29; 21%) GEMOX (14%), DHAP and ESHAP (3/29; 10% each) were the most commonly employed second-line regimens. A positive outcome (SD, CR, PR, ongoing treatment) was recorded for only 12/31 (39%) of the patients who did not progress to a second-line regimen, suggesting that a first-line regimen was unsuccessful in 47/59 (80%) of patients. Median survival was 14.5 months and 1-year and 3-year survival were 57% and 49%, respectively. Because of the small numbers of patients in the different subtype groups, we were unable to determine whether survival rates might be different between subgroups.
Overall, the phenotype of our Algerian cohort is not markedly different from that observed in a large international cohort of patients with PTCL and NKTCL. Among 1314 patients from 22 countries, the most common subtypes were PTCL-NOS (25.9%), angioimmunoblastic type (18.5%), NKTCL (10.4%), and ATLL (9.6%) [6]. PTCL-NOS was the most common subtype in both North America and Europe (34%), whereas NKTCL and ATLL were common in Asia. In this respect, the frequencies of both NKTCL and ATLL in our cohort were somewhat in between those reported in Asia (approximately 22% and 25%, respectively) and North America (approximately 5% and 2%) and Europe (approximately 4% and 1%). In a multinational, multi-center, prospective registry of 490 patients with PTCL enrolled from 9 Asian countries, of whom 59% were male and the median age was 59 years, NKTCL (28%) and AITL (24%) accounted for more than half of the patients, and PTCL-NOS for only 20% [7]. ALCL (ALK +/-) comprised 16% of the cohort.
The predominant treatments for PTCL-NOS, AITL and ALCL were CHOP-like and CHOEP-like, and no significant difference was observed in survival between these regimens. Although previous registry data has suggested that adding etoposide to CHOP might improve outcomes, data are conflicting and we did not observe any apparent benefit with CHOEP versus CHOP. A recent meta-analysis suggested that there were no differences in therapeutic effect for patients with PTCL between CHOP and CHOEP with regards to CR, PR, and overall response rate [24].
Although because of heterogeneity of data in different registries it is difficult to make direct comparisons, survival rates in our cohort appear to be similar or slightly lower to those observed in other registries. In a prospective cohort study of 256 patients with nodal PTCL in America, of whom 63% received a doxorubicin or doxorubicin plus etoposide-containing regimen as initial therapy and approximately 76% were stage III or IV, estimated median survival was 43 months and 12- and 24-month survival rates were 71.7% and 58.7%, respectively [9]. In the Swedish registry, 499 patients out of 594 treated with chemotherapy received CHOP or CHOEP as first-line treatment and the overall response rate was 65-75% [12]. In the overall cohort of 755 patients, 65% were stage III or IV and 5-year overall survival was 34%. Among 198 patients in a Korean nationwide prospective registry, 61% were male and the majority presented as advanced stage disease [15]. CHOP or CHOP-like therapy was the first-line regimen in 75% and the 2-year overall survival rate was 64.4%.
In this cohort of Algerian patients with PTCL, the majority of patients were men presenting at a young age with advanced disease. The most common subtypes of PTCL included PTCL-NOS, ALCL, ATLL, NKTL and AITL. This spectrum of subtypes resembles more closely that observed in Asian countries than in European or North American non-Hispanic White patients. Most patients are treated with an initial regimen of CHOP or CHOEP-based chemotherapy. The prognosis for these patients remains poor, with less than one out of five responding satisfactorily to first-line therapy, and fewer than half surviving for more than 3 years.
The study and writing the report were supported by a non-restrictive grant from Mundipharma Middle East FZ-LLC. The study was conducted independently of the sponsor with assistance from a third party contract research organization.
NBB, MAB, MTA and FG were jointly responsible for conceiving the study, all decisions relating to the conduct of the study, interpretation of the results and decision to submit the article for publication. DW wrote the first draft of the study. All of the authors reviewed, contributed to, and approved drafts and the final manuscript and agree to be responsible for all aspects of this work. DW reports personal fees from Mundipharma Middle East FZ-LLC. The authors report no other conflicts of interests relating to this study.