To examine the effects of a standardized neural mobilization treatment strategy in patients with Neurogenic Claudication (NC) related to degenerative Lumbar Spinal Stenosis (LSS).
Prospective case series. Seven older adults with NC related to LSS received a standardized treatment program. Outcome measures included the Swiss Spinal Stenosis questionnaire (SSS), Numeric Pain Rating Scale (NPRS), time to First Symptoms (TFS) and Total Ambulation Time (TAT). Outcomes were assessed at the 7th visit and 3-months.
Improvements were noted at the 7th visit in the NPRS (p = 0.027), SSS symptom (p = 0.018) and function (p = 0.017), TFS (p = 0.018) and TAT (p = 0.027). At 3-months, improvements were found in the SSS symptom (p = 0.022) and function (p = 0.016), but not NPRS (p = 0.115). At the 7th visit MCID was met in 5 of 7 participants for the NPRS and SSS symptoms and 7 of 7 for the SSS function. At 3-months the MCID was met in 3 of 5 participants in the NPRS, 4 of 5 in SSS symptoms, and 5 of 5 in SSS function.
Clinically meaningful improvements in pain and functional outcome measures were noted after the application of a standardized neural mobilization treatment strategy both immediately after the treatment period and at 3 month follow-up.
Neural mobilization, Neurogenic claudication, Walking tolerance, Hip mobilization, Lumbar spinal stenosis
HEP: Home Exercise Program; LSS: Lumbar Spinal Stenosis; MRI: Magnetic Resonance Imaging; MCID: Minimally Clinically Important Difference; NC: Neurogenic Claudication; NPRS: Numeric Pain Rating Scale; SSS: Swiss Spinal Stenosis Questionnaire; TFS: Time to First Symptoms; TAT: Total Ambulation Time
Degenerative Lumbar Spinal Stenosis (LSS) is a condition most often observed in people over 60-years-old [1,2]. It is the most common indication for spinal surgery in people older than 65 years [3] and is associated with three main areas of stenosis due to spinal degeneration: Central canal, lateral recess, and intervertebral foramen [4]. The degeneration leads to diminished space around the neural and vascular elements, which can eventually result in symptoms of lower limb Neurogenic Claudication (NC) [4,5]. Neurogenic claudication is a common complaint in people suffering from degenerative LSS that is characterized by lower limb symptoms during standing and walking activities [3]. Common symptoms include unilateral or bilateral leg pain, fatigue, paresthesia, and/or tightness ultimately resulting in impaired ability to stand or walk for prolonged periods of time [5,6]. These symptoms are relieved during sitting and/or in positions of lumbar flexion [4].
These common clinical symptoms are frequently related to spinal canal diameter changes with specific positions and movements. For example, spinal canal diameters increase with sitting and lumbar flexion, while they decrease with lumbar extension and in activities that load the spine such as standing and walking [7-9]. In addition, lumbar extension and walking increase epidural pressure which leads to increased compression of neural and vascular structures within the central spinal canal [10] and in the intervertebral foramen [9].
There is a growing body of evidence supporting conservative multi-modal treatment programs for managing degenerative LSS [11-15]. The design of these multi-modal treatment approaches vary but each often include stretching activities, lumbar strengthening, hip and spine mobilization, mechanical traction, neural mobilization, and aerobic exercise. Treatment duration in these studies also varies between 9-15 treatment sessions over a period of 3-6 weeks. These programs have demonstrated immediate and mid-term benefits, with one study reporting sustained benefit in a majority of subjects at one year [15]. Though the interventions in each study differed, each included an aerobic exercise component involving cycling or body-weight supported treadmill walking. Some authors incorporate aerobic exercises to improve the patients' overall fitness and for leg conditioning [11], while others emphasize that specific spinal positioning during aerobic exercise provides additional benefit by improving neural tissue hemodynamics [14-17].
Neural mobilization exercise is a treatment option for symptomatic LSS and may be a beneficial self-management option for patients with this condition. These exercises, in particular a supine tensioner technique, have been emphasized to varying degrees in studies examining multimodal conservative interventions for LSS [11,14,18]. Neural mobilization exercises are thought to facilitate neural gliding, improve tissue vascularity and disperse noxious fluids [19,20]. These effects may translate toward improved health and functioning of compressed neural tissue enabling the tissue to meet the metabolic and functional demands needed during walking activities. Neural mobilization exercises can be easily performed by patients at home, require no special equipment, and take little time to perform as a daily maintenance program.
Limitations in hip extension are commonly found in older adults [21], and patients may compensate for this loss of motion with anterior pelvic tilting and lumbar extension during gait [21,22] resulting in a dynamic narrowing of the lumbar spinal canals. Thus, using manual therapy to address hip motion limitations may prove beneficial in patients with LSS. Several studies have incorporated manual therapy to mobilize both the spine and hips to treat patients with symptomatic LSS [11,15,16,18].
The double knee to chest stretch is commonly used as a flexion-based exercise to promote opening of the spinal canals and to stretch tight lumbar paraspinal musculature [12,23,24]. This exercise is simple to perform and can be included in a Home Exercise Program (HEP).
The purpose of this study was to observe the effects of a neural mobilization treatment strategy on a group of patients with NC related to LSS.
This study is a prospective case series. Systematic consecutive sampling was used so that all patients screened that met the inclusion/exclusion criteria were asked to participate in the study and were included upon signing informed consent forms. Interventions were performed at a university-based outpatient clinic in Amarillo, Texas. The study protocol was approved by the Institutional Review Board at Texas Tech University Health Sciences Center.
Patients were recruited from local physician offices in the panhandle region of Texas from January 2018 - July 2019. The referral sources included neurosurgeons, an orthopedic spinal surgeon, and a neurologist. The principal investigator conducted a routine comprehensive physical therapy examination to determine the patient's clinical status. An additional investigator recorded the outcome measures. Seven participants with a mean age of 68.6 ± 8.8 years with reports of NC related to LSS were included in this study.
The inclusion criteria were participants aged 50-89 years-old, clinical symptoms of intermittent unilateral or bilateral leg pain occurring with walking and standing activities relieved only with sitting or by assuming flexed positions, leg symptoms rated as greater than 4/10 on the Numeric Pain Rating Scale (NPRS) and provoked within 15 minutes of walking, and Magnetic Resonance Imaging (MRI) confirmed lumbar spinal stenosis.
The exclusion criteria were previous lumbar surgery that included fusion, spinal injection in the last 6 weeks, impaired walking tolerance due to factors other than neurogenic claudication, inability to follow the rater's instructions, medical contraindication for hip mobilizations in extension or flexion, or any current medico-legal issues.
The participants attended 7 physical therapy visits (6 for treatment, 1 for outcome assessment), and were treated 2 times per week for 3 weeks. Participants were asked to not alter their medication intake during the entire study period. The intervention was performed by the primary investigator who is a licensed physical therapist with 11 years of clinical experience and is a board-certified clinical specialist in orthopaedic physical therapy.
The participant laid supine on a treatment table. The participant drew both knees as close to their chest as possible using their hands. This was repeated 3 times for 60 seconds (Figure 1a).
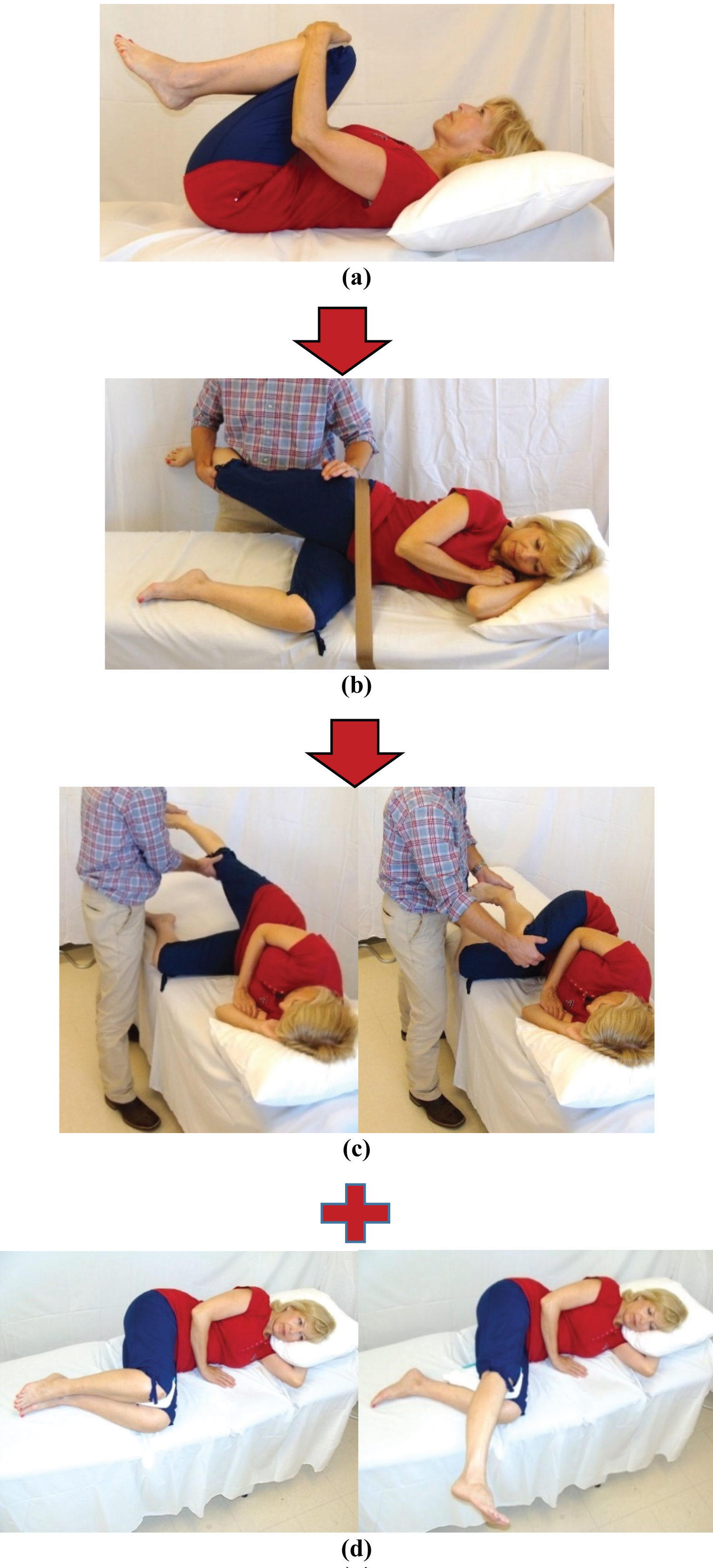 Figure 1: (a) Double knee to chest stretch; (b) Sidelying hip extension mobilization; (c) Passive neural mobilization; (d) Active neural mobilization.
View Figure 1
Figure 1: (a) Double knee to chest stretch; (b) Sidelying hip extension mobilization; (c) Passive neural mobilization; (d) Active neural mobilization.
View Figure 1
The participant was placed in sidelying. The table side hip and knee were flexed to approximately 90 degrees. A mobilization strap was placed around their pelvis and secured to the treatment table to stabilize the pelvis. The clinician grasped the participant's top leg placing the palm behind the greater trochanter and the other hand under the participant's knee keeping it in a slightly flexed position. The clinician moved the participant's hip into extension until a barrier was felt and then performed 2 sets of 30 grade III or IV oscillations in a dorsal-to-ventral direction. This was performed on each hip (Figure 1b).
The participant was placed in side lying. Their trunk was partially flexed forward to promote opening of the lumbar neural canals. The table side hip and knee were flexed to approximately 90 degrees. The clinician grasped the participant's top knee with one hand and their heel with the other. The clinician passively moved the participant's lower extremity into full hip and knee flexion then into neutral hip extension and full knee extension [25,26]. This process was repeated for a total of 5 sets of 30 repetitions at one cycle every 2 seconds on each lower extremity (Figure 1c).
This consisted of double knee to chest stretches (3 × 60 seconds) and active sidelying neural mobilization exercises. The participant flexed their trunk slightly forward, flexed their hips to approximately 80 degrees and their knees to approximately 90 degrees. The participant then actively moved their knee into extension then returned to the starting position. This was repeated for 30 repetitions on each side (Figure 1d). Participants were asked to perform the HEP twice daily during the treatment period and for at least 3 months after discharge.
Swiss Spinal Stenosis questionnaire (SSS): The SSS is a condition specific measure for patients with lumbar spinal stenosis. It consists of three separate subscales: Symptom severity, functional, and satisfaction. Lower scores represent fewer symptoms, greater function, and greater satisfaction with the results of treatment. The Minimal Clinically Important Difference (MCID) for the SSS symptom subscale is 0.36 and the functional subscale is 0.10 [27]. We substituted the word "surgery" in the satisfaction subscale and replaced it with "physical therapy treatment" in order to reflect the type of intervention the participant received.
Numerical Pain Rating Scale (NPRS): The NPRS has good test-retest reliability (ICC = 0.61) [28]. This measure uses an 11 point scale where "0" represents "no pain" and "10" represents "the worst imaginable pain". The MCID for NPRS in the LSS population is 1.5 [27]. Participants were asked to rate their average pain with walking activities during the past week.
Time to First Symptoms (TFS): Time to first symptoms assesses the amount of time it takes for leg symptom onset during ambulation. This measure has goodtest-retest reliability (CCC = 0.98) in subjects with lumbar spinal stenosis [29]. Participants walked on a treadmill in a fully erect posture at a self-selected comfortable pace and reported the first moment of leg symptom onset. Maximal walking time permitted was 15 minutes [29].
Total Ambulation Time (TAT): Total ambulation time is an objective test to measure walking tolerance and has good test-retest reliability (CCC = 0.96) in subjects with lumbar spinal stenosis [29]. Participants walked at a self-selected comfortable pace until they experienced symptom reproduction at a level that would cause them to stop walking in usual daily life situations [29]. Maximal walking time permitted was 15 minutes.
A secondary investigator, who did not provide patient treatment, assessed all outcome measures and was blinded to the baseline outcome measure scores during outcome reassessment. All outcome measures and HEP compliance were assessed at the initial examination and at the 7th visit. The NPRS, SSS subscales and HEP compliance were reassessed at 3-month follow-up using questionnaires mailed to the participants. Participants in this study received all physical therapy visits free of charge.
Descriptive statistics were used to assess the following outcome measures: (1) SSS symptom and functional subscales (baseline, 7th visit, 3-month follow-up) and SSS satisfaction subscale (7th visit, 3-month follow-up); (2) NPRS (baseline, 7th visit and 3-month follow-up); (3) TFS and TAT (baseline and 7th visit).
Statistical analyses were performed using SPSS version 25.0 (IBM Corp.). Data did not meet the assumptions of parametric data, therefore non-parametric tests were used. Statistical tests were performed as two-tailed tests with a significance level of 0.05.
Group differences between baseline and 7th visit for the NPRS, SSS symptom and functional subscales were assessed using Wilcoxon signed-ranks test. Effect size was determined using: r = Z/√N. Group differences between three time intervals (baseline, 7th visit, and 3-month follow-up) were assessed using the Friedman 2-way ANOVA by ranks. Effect size was determined using Kendall's W.
The Wilcoxon signed-ranks test was used to assess group differences for TFS and TAT (time: baseline vs. 7th visit). Effect size was determined using: r = Z/√N.
A total of 10 patients were recruited and screened for study participation; eight met the inclusion/exclusion criteria and consented to participate. One participant attended three treatment sessions, but withdrew from the study due to his inability to attend the planned follow-up visits. Data from seven participants were used for data analysis. Clinical examination and MRI findings for each participant are provided in (Table 1). All seven participants were reassessed at the 7th visit. Five of the seven participants were reassessed at the 3-month follow-up; two participants elected to have surgery.
Table 1: Clinical and MRI examination findings. View Table 1
Significant NPRS score improvements were found between baseline and the 7th visit (p = 0.027), but not at 3-months (p = 0.115). Five of seven participants met the MCID for the NPRS at the 7th visit and 3 of 5 participants met the MCID for the NPRS at 3 months (Table 2).
Table 2: Numerical pain rating scale. View Table 2
Significant improvements were found at both the 7th visit (p = 0.018) and at 3-months (p = 0.022). Five of seven participants met the MCID at the 7th visit and 4 of 5 met MCID at 3-months (Table 3).
Table 3: Swiss Spinal Stenosis Questionnaire (symptom and functional). View Table 3
Significant improvements were found at the 7th visit (p = 0.017) and at 3-months (p = 0.016). All participants met the MCID at the 7th visit and 5 of 5 reported the MCID at 3-months (Table 3).
Walking times improved in all participants (Table 4). Significant changes in walking times were found at the 7th visit in the TFS (p = 0.018) and TAT (p = 0.027). Additionally, we noted that self selected comfortable walking speeds increased in 6 of 7 participants and group median walking speeds increased from 1.2 mph (range 0.9-3.0) to 1.5 mph (range 1.0-3.2) at the 7th visit. Walking distances for TFS improved from a median of 88 ft to 578 ft and group distances for TAT improved from a median of 428 ft to 1579 ft.
Table 4: Walking measures. View Table 4
This is the first study to report outcomes of a standardized neural mobilization treatment program in people diagnosed with MRI confirmed degenerative LSS.
Clinically meaningful improvements in pain and functional outcome measures were noted immediately after the treatment period and at mid-term follow-up. The study participants reported high satisfaction rates with the treatment and benefits received; these high satisfaction rates were noted at the 7th visit and at 3-months (Table 5).
Table 5: Swiss Spinal Stenosis Questionnaire (satisfaction subscale). View Table 5
Participants who demonstrated the best response to this treatment regimen tended to display higher baseline NPRS, less lateral foraminal degeneration on MRI, less combined lateral foraminal and central canal stenosis on MRI, and longer symptom duration. Such observations could assist in identifying a subgroup of patients who are most likely to benefit from this treatment regimen; however, larger studies are necessary to confirm these findings.
There is ongoing debate regarding the exact cause of neurogenic claudication. Patients with LSS experience increased pressures in the epidural and intraforaminal spaces during standing and walking [9,10]. It is thought that increased epidural and intraforaminal pressures may result in neural ischemia, venous flow interruption with subsequent venous engorgement, or a combination of both [30,31]. The improvements in participant symptoms and walking tolerance in this study may have resulted from improvements in the overall health and fluid dynamics of the chronically compressed neural structures.
The purported benefits of neural mobilization are numerous and include facilitating nerve gliding, reducing nerve adherence, dispersion of noxious fluids, increasing neural vascularity, improving axoplasmic flow, and reducing dorsal horn and supraspinal sensitization [19,32]. Prior research demonstrated that both specific movements and general lower extremity range of motion resulted in sciatic nerve excursion [32,33].
Furthermore, neural mobilization exercises in the lower limb have been shown to alter neural fluid dynamics as far proximal as the L4 dural space [20]. Hip extension mobilizations are thought to improve hip extensibility during gait thereby reducing further increases in epidural and intraforaminal pressures that occur with compensatory lumbar extension and subsequent narrowing of the neural canals. Double knee to chest stretches may improve fluid dynamics in the affected neural structures by temporarily reducing neural compression in the central and lateral vertebral canals [9] and lengthening lumbar extensor musculature.
The positive results found in participants that completed the 3-month follow-up indicate that a HEP including neural mobilization and double knee to chest stretches may be beneficial for symptom maintenance. Study participants reported a high HEP compliance rate of 100% at the 7th visit and 75% at 3 month follow-up.
Two participants were unavailable at the 3-month follow-up due to electing for surgery. The outcome measures at the 7th visit showed that one of these participantsmet the MCID in the NPRS and SSS symptom subscale, but had minimal improvements in walking measures. The other participantmet the MCID in the SSS symptom and functional subscales, demonstrated a small increase in TFS, and demonstrated a dramatic improvement in TAT. Compared to other participants, these two participants expressed an interest in receiving surgery for their condition prior to inclusion in the study.
Study limitations include the small sample size and lack of a control group. These two factors limit the ability to assess whether a true treatment effect occurred in response to study interventions. Additionally, the long-term treatment effects were not assessed as the follow-up time was limited to three months.
This study describes a neural mobilization treatment strategy for patients with NC related to LSS. Neural mobilization, in particular the slider technique, is an often overlooked treatment approach for this patient population. Significant and clinically meaningful improvements were observed in pain symptoms, self-report functional mobility, and walking tolerance. Clinicians are encouraged to consider incorporating this neural mobilization strategy in their treatment approach for this patient population.
The authors report no conflicts of interest.
All authors provided a substantial contribution to the design and review of this study.