In this study we evaluated the urinary excretion of 1-OHPu in 31 healthy "street sweepers" and in 8 with a bronchitis process all smokers, exposed mainly to urban and industrial air pollution (they use semiautomatic systems to empty garbage road containers and/or manually collect bags with materials for recycling, but the exposure is also due to fumes from diesel engine combustion and to other environmental pollutants in areas where the production of PAHs is due to the operation of coal-fired power stations, lime furnaces, petrochemical candles, combustion of part of olive trees - Salento country), as an indicator of internal dose for exposure to fumes from the combustion of diesel and internal combustion engines, but also of domestic heating systems and industrial emissions of PAHs from petrochemical factories and coal-fuelled power stations) [18]. All the 39 "street sweepers", living in the same operational area (Brindisi-Italy), were male. Among the healthy (non bronchitic) "street sweepers", the smokers were found to be 12 (26%). For every worker the 1-OHPu urinary concentration was measured in samples collected at the beginning and at the end of their working periods for at least three consecutive days of work. The measurements were made by using the method of Jongeneelen (1987) [19], after enzymatic hydrolysis of β-glucuronides and/or sulphate conjugates, followed by analysis using high-performance liquid chromatography (HPLC), combined with a fluorimetric detector. The limit of detection of the method is 25 ng/l. The concentrations of urinary 1-OHPu were expressed in ng/g creatinine.
Environmental exposure values are reported in table 1. The geometric averages of the measured 1-OHPu concentration values are shown in table 2 & table 3, and histograms in figure 1, figure 2 and figure 3.The above values were compared with the ones of a group of 18 workers involved in indoor activities, in order to evaluate the usefulness of 1-OHPu assay as part of health monitoring processes. In figure 4 is reported ILCRinhalation in PAHs exposure and in figure 5 the same data on a logarithmic scale.
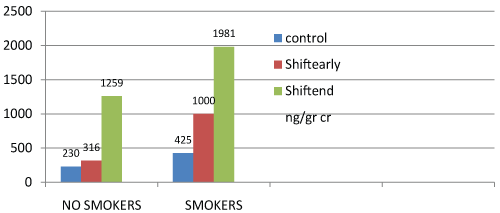 Figure 1: [1-OHPu concentration (ng/g creatinine)] at the beginning (shiftearly) and at the end (shiftend) of the working shift for no smokers and smokers.
View Figure 1
Figure 1: [1-OHPu concentration (ng/g creatinine)] at the beginning (shiftearly) and at the end (shiftend) of the working shift for no smokers and smokers.
View Figure 1
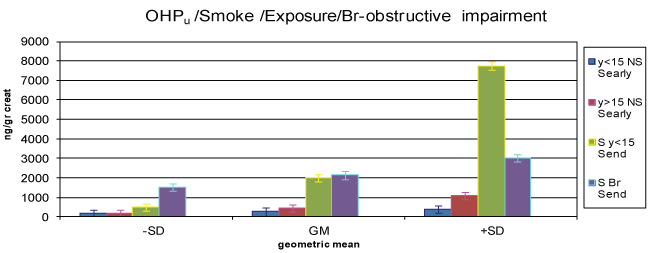 Figure 2: 1-OHPu concentration in never smokers early shift exposure < 15 years (y < 15 NS Searly) never smokers early shift exposure > 15 years (y >15 NS Searly), smokers < 15 y Send (S Send), bronchitis illness.
View Figure 2
Figure 2: 1-OHPu concentration in never smokers early shift exposure < 15 years (y < 15 NS Searly) never smokers early shift exposure > 15 years (y >15 NS Searly), smokers < 15 y Send (S Send), bronchitis illness.
View Figure 2
The concentration values associated to never smokers (NS) and forever smokers (S) and exposure (years) are shown in table 2 and figure 2, at the beginning and end of working shift.
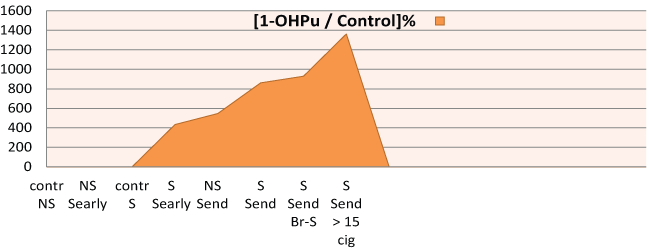 Figure 3: [1-OHPu ] in PAH exposure / [1-OHPu ] Controls % in workers groups: NS: Never smokers, S: Forever smokers, Shiftearly and Shiftend Br-S: Bronchitic smokers.
View Figure 3
Figure 3: [1-OHPu ] in PAH exposure / [1-OHPu ] Controls % in workers groups: NS: Never smokers, S: Forever smokers, Shiftearly and Shiftend Br-S: Bronchitic smokers.
View Figure 3
 Figure 4: ILCRinhalation in PAHs exposure
View Figure 4
Figure 4: ILCRinhalation in PAHs exposure
View Figure 4
ILCR calculated by PAHs exposure with benzo[a]pyrene concentration expressed in ng/m3- ILCR = {Ca [CSF (BW/70) 1/3]dayyear-1.year/BW.AT}.Cf [20,21] where:
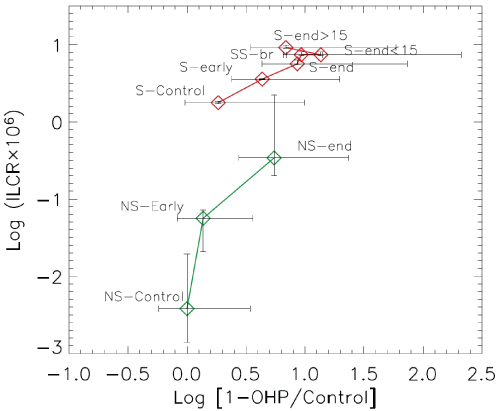 Figure 5: ILCRinhalation in PAHs exposure expressed on a logarithmic scale
View Figure 5
Figure 5: ILCRinhalation in PAHs exposure expressed on a logarithmic scale
View Figure 5
Table 1: Environmental exposure value. View Table 1
ILCR is incremental life-time cancer risk [22]
Table 2: 1OHPu shiftearly never smokers (Searly NS) and smokers (Searly S) and Shiftend never smokers (Send NS) and Shiftend Smokers (Send S) - t-test (*). View Table 2
Table 3: Concentration of 1-OHPu, associated to exposure (y) in never smokers (NS) and forever smokers (S), at the beginning (Searly) and end (Send) of working shift -t-test (*). View Table 3
Ca is PAH concentration as Bap ng/m3 in working shift
CSF (3,1) is cancer slope factor ng/m3 (WHO24); (mgKg-1day-1)-1 U.S.EPA, 2001
BW = body weight (75 Kg)
40 year of life in work
Dayyear-1 - days of work for year of work exposure (210)
AT = average time in work (40)
Cf = 10-6
For evaluating risks from inhalation exposure, MDH recommends a Health Risk Value (HRV) of 0.001 ng/m3 for B[a]P [23], which is based on a slope factor of 7.3 (mg/kg-day)-1. This slope factor is the geometric mean of the B[a]P slope factor range used by the U.S. Environmental Protection Agency (U.S. EPA, 2001).
The geometric mean concentration of 1-OHPu among the exposed workers at beginning of their working shift is greater in forever smokers (S) than for never smokers (NS) and for never smokers controls (control NS). But, the most significant data is that the exposed workers, both never smokers and forever smokers, at the end of their working shift (Send NS: Send S) show higher 1-OHPu concentration values than that of the same workers at the beginning of their working shift and of the control non-exposed workers. Tables and figures also demonstrate the smoke influence, associated with the exposure, i.e. the greater values of urinary excretion of 1-OHPu against the ones of never-smokers. More, the 1-OHPu concentration is significantly higher in forever smokers with more than 15 years of exposure, than in never smokers at the beginning of their working shift (Searly NS). Cigarette smoke factor is significant for smokers in conjunction with less than 15 years of exposure at the end of working shift (S end shift < 15 y exposure), as well as for the control group. The same happens for those with bronchial obstructive impairment.
In general 1-OHPu concentration behaves as an interesting variable, associated not only to recent exposure, but also to the effects of accumulation, since the increase in concentration is influenced by the years of exposure as well as by unhealthy breathing ways, likely to inflammation and edema accumulation.
As regards the application of preventive rules, the characteristics of smoking behavior are to be studied. Behaviors depend on the technology used by the worker. Sweepers attached to the moving medium with hook switch of dumpsters to the moving car, can hardly smoke during the service period, but they can smoke very much, assuming the related risk, at the beginning and end of their workday.
Our study showed an increase in the urinary excretion of 1-OHP at end of working shift with respect to the one at the beginning, in smokers and never smokers and in suffering from chronic bronchitis, exposed to pollution sources such as urban traffic, combustion fumes from diesel engines, home heating systems and emissions of PAHs from petrochemical factories and gas and coal combustion in power stations [18]. The smoking of tobacco is confirmed, among exposed to vehicular traffic, to be an important additional factor of increase of urinary excretion of 1-OHPu [1]. However, the contribution to the increase of global dose due to eight working hours is not negligible, due to the accumulation characteristics demonstrated by PAHs. Urinary excretion of 1-OHPu, as an indicator of internal dose of PAHs, proves to be not only an indicator of recent exposure, but also of accumulation in the past. It can be used as a signal to identify the groups at greatest risk, although its proved photochemical mutagenic effect is not considered to be specific for genotoxicity, as for 3-OHBaP [11]. However, it can be anyway considered as the best indicator of internal dose for exposure to PAHs for several categories of persons [12,13], taking also into account that Pyr is much more soluble in water and able to pass through the lung to blood and undergo biotransformation with respect to Bap [16,24]. Urinary excretion of 1-OHPu, as an indicator of the internal dose exposure to fumes from diesel engine combustion and to other environmental pollutants in areas where the production of PAHs is due to the operation of coal-fired power stations, lime furnaces, combustion of part of olive trees, petrochemical candles involves a careful evaluation of the risk factors in the context of the working procedure of street sweepers. The prevention of this kind of risk might be reduced by the use of electric and/or natural gas fuelled cars especially in waste sorting house by house.
The evidence of a greater concentration in bronchitic men ask for experimental studies to test the ability of Pyr to remain in the epithelium and in sub-bronchial epithelium, apparently to a greater extent, for greater dilution in the inflammatory process, and then to undergo a biotransformation activity slowdown in water-soluble metabolites. It establish a reserve as a pre-toxic pulmonary factor, similar to the genotoxic action of compounds like BaP [25], indicating accumulation phenomena well known for PAHs, since when attached to inhaled particles at the alveolar level are quickly released if placed at the surface of these particles, while associated to deeper penetration ones, like nanoparticles [26], are released more slowly [10] or involved in uptake endocytosis [27].
It could be useful to calculate the "cancer risk assessment" in our group and similar ones [28]. In this contest, the calculation of ILCR (incremental life-time cancer risk inhalation) in PAHs exposure as a factor of risk evaluation for lung cancer demonstrates a good association with 1-OHPu concentration, but only for smokers with exposure after finishing work and subjects with br-obstructive impairment (ILCRinhalation range 5, 6-9, 13 × 10-6), condition that contributes to the total risk with skin contact and ingestion [29]. We are in agreement with recent study [30] about the characteristics of variability of 1- OHPu excretion in different groups of workers exposed or in people with urban or industrial pollution, but in addition to environmental and behavioral factors are to be considered as important parameters as surface, size, clustering that affect in the process of uptake, intake and biotransformation in different types of atmospheric particulate in much part of nanoparticle size.
The possibility of implementing prevention is based on simple and effective principles and on the utilization of useful methods. In our case, the street sweepers in the performance of their work are exposed to a mixture of PAHs of some carcinogenic action. In the grading of risk, this exposure appears to be important, in addition to traditional influencing factors such as the time of exposure, cigarette smoking, food intake and the effects of accumulation within the working day, associated also to possible respiratory diseases. The 1-OHPu urinary concentration test appears useful to identify groups of persons exposed to greater risk of carcinogenicity. It is also a biological indicator of exposure to Benzo[a]pyrene, which among the PAHs is the most dangerous agent, according to ILCR values (ILCRinhalation range 5, 6-9, 13 × 10-6), and accumulates in the body. Jongeneelen (1992), proposed an occupancy limit of 1-OHPu urinary concentration of 2.3 μmol/mol creat for an environmental concentration of 2 μg/m3. Further studies on the concentrations in air show that Pyr values of 49 ng/m3 corresponding to 0.85 ng/ml, with considerable lowering of values [31].
The findings of the present study are linked to the beginnings of toxicology involving chimney sweeps, namely the early work by Sir Percival Potts in the late 1770s. His work contributed to the emerging science of epidemiology as well as toxicology and lead to one of the first public health laws in Europe, namely the Chimney Sweepers Act of 1788.
We believe that further results in the analysis of the permissible limits of carcinogenic risk, waiting for epidemiological projections, often late in terms of prevention, are key to the reduction of the negative effects, DNA damage and the development of cancer.