Purpose: The aim of the present study was to evaluate the acute and sub-acute toxicity of the trunk bark hydro-ethanolic extract of Stereospermum Kunthianum Cham: in Wistar rats.
Methods: For acute toxicity assessment, three 12-week-old female rats were administered by gavage a single oral dose of 2000 mg/kg of the trunk bark hydro-ethanolic extract of Stereospermum kunthianum. LD50 was estimated after 14 days of observation. Subacute toxicity was assessed at three dose levels: 500, 1000, and 2000 mg/kg. The extract was administered by gavage to a batch of 6 male and female rats, daily for 28 days. Each batch consisted of 3 males and 3 males. A 4th batch that received only distilled water was used as a control. Monitoring body weight was carried out, and urinalysis, haematological, and biochemical analyses were carried out on urine, whole blood, and serum samples.
Results: The results showed that the hydroethanolic extract of trunk bark and hydro-ethanolic extract of Stereospermum kunthianum did not result in death among the animals; there were no obvious signs of toxicity. However, in animals that received the extract for 28 days, two deaths were noted in females, macroscopic examination also showed stomach distension compared to the controls. These stomach distension which was dose dependent without any disturbance of the determined biological parameters.
Conclusion: According to these results, LD50 was estimated to be greater than 2000 mg/kg. Female rats appear to be more sensitive than males in long-term. NOAEL was estimated below 500 mg/kg. The results showed that the trunk bark hydro-ethanolic extract of Stereospermum kunthianum Cham. Has low or no toxicity in Wistar rats at tested doses.
Stereospermum kunthianum, Trunk bark, Acute/sub-acute toxicity, LD50, Rat
ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; EDTA: Ethylene Diamine Tetra Acétique; HCT: Hematocrit level; LD: Lethal Dose; MCV: Corpuscular Volume; MCHC: Mean Corpuscular Hemoglobin Concentration; NOAEL: No Observable Adverse Effect Level; OECD: Economic Co-operation and Development; RBC: Red Blood Cells; WBC: White Blood Cells
In Africa, medicinal plants are often used in the treatment of various illnesses. Among these medicinal plants, S. kunthianumis widely used. This plant is also known as Bignonia lanata R. Br. ExnFrese or Dolichandrone smithii Baker is a species belonging to the Bignoniaceae family and native to tropical Africa. The bark extract of S. kunthianum is used in the treatment of syphilis, wounds, bronchitis, pneumonia and cough [1,2]. It's also indicated in cases of gastritis and erectile dysfunction [3]. Biological investigations have shown that S. kunthianum extracts possess antioxidant [4] antibacterial [5,6], anti-inflammatory [7], anti-convulsant [8], antihelmintic [9], antidiarrhoeal [10], analgesic [11] and wound healing [12] activities. The study is focused on the trunk bark, which is the most used part of the plant in Ferlo (Senegal).
These biological activities would be explained by the presence of many chemical components including polyphenols like tannins and flavonoids, saponins, terpenoids, higher fatty acids, coumarins, sterols [13-16].
Despite these proprieties and the multiple uses of the plant, few studies have focused on its toxicity. It is in this context that our study aimed to study the acute and sub-acute toxicity of the hydro-ethanolic extract of trunk bark in Wistar strain rats.
Trunk barks of S. kunthianum were harvested in Widou, in the municipality of Téssékéré (Louga region, Senegal). The plant has been identified at the Pharmacognosy and Botany Laboratory of the Faculty of Medicine, Pharmacy and Odontology (FMPO) of Cheikh Anta Diop University in Dakar and a voucher specimen (n° 1410) was deposited in the herbarium of the same institute (FMPO) for future reference. The bark (1 kg) was dried out in the sun during fifty days at room temperature, then pulverized using a Brabender-type electric grinder.
Wistar strain albino rats bred in the Toxicology and Hydrology Laboratory of the Cheikh Anta Diop University in Dakar (Senegal) were used. They were male and female rats aged 12 to 16 weeks. Females were nulliparous and non-pregnant. They were housed with 3 rats of the same sex in plastic cages with bedding made of wood shavings. They were fed with an appropriate commercial formula and given ad libitum filtered water. The room temperature varied between 20 and 25 °C.
The plant extract was prepared at the Pharmacognosy and Botany Laboratory of the Faculty of Medicine, Pharmacy and Odontology (FMPO) of Cheikh Anta Diop University in Dakar. The extraction was done by decoction under reflux of 50 g of S. kunthianum trunk bark powder in 500 ml of an ethanol/water mixture (80-20; v/v) for 30 minutes. Pumice stone was added to stabilize the boiling. After filtration, the solution was concentrated using a rotary evaporator at 70 °C. The result was a pasty extract that was dried in a dessiccator. The dry extract thus obtained constitutes the hydro-ethanolic extract (EHE).
The acute oral toxicity of the hydro-ethanolic extract of the trunk bark of S. kunthianum was assessed in rats by the toxicity class method according to the OECD Guideline 423 for the Testing of Chemicals adopted on December 17, 2001 [17].
The test substance was thus administered to 3 female rats weighing 170 ± 10g by gavage using a probe at a single dose of 2000 mg/kg bw. Mortality, general behavior, and any signs of toxicity were observed for 14 successive days.
Subacute oral toxicity of the hydroethanolic extract of S. kunthianum was assessed in rats in accordance with OECD guideline N°. 407 for the testing of chemicals adopted on October 3, 2008 [18].
For this test, 24 rats divided into 4 groups of 6 (3 males and 3 females) received the test substance by gavage for 28 consecutive days. Group 1 corresponds to the controls and are force-fed with distilled water with maximum volume, and groups 2, 3, and 4 received the test substance at doses of 500; 1000 and 2000 mg/kg. The rats are weighed the day before the test and once a week for the duration of the test. Urine was collected weekly for biochemical testing. Mortality, weight changes, and signs of toxicity are monitored during the test. At the end of the test, the rats are anaesthetized with a mixture of Xylazine/ketamine, blood is taken from the heart for biological examination, then the rats are sacrificed and a macroscopic examination is performed.
Urine samples are taken weekly to determine glycosuria and albuminuria using URS-3 (Urine Reagent Strips for Urinalysis) urine strips.
Complete blood count was performed on blood samples collected in an EDTA tube at the end of the test to determine the number of red blood cells, the number of white blood cells, the number of platelets, hemoglobin level, hematocrit level, corpuscular volume and mean corpuscular hemoglobin concentration.
Various parameters were determined with the Abbott Architect Ci 4100 Analyzer; these were assays of blood sugar, urea, creatinine, and transaminases (ASAT and ALAT), for renal and liver function.
The analyzes were performed with R software version 1.2.1335. Kendall's nonparametric test was used to see if the differences were significant. Values were presented as mean ± standard deviation. A difference between the values is considered significant for p (p-value) ˂ 0.05.
During the 14 days of observation, no death was observed after the administration of a single dose of 2000 mg/kg of hydro-ethanolic extract of S. kunthianum trunk bark. No other signs of toxicity (neurological, respiratory) were observed during these 14 days. LD50 was estimated to be greater than 2000 mg/kg.
Two (2) deaths among the 18 rats which received the extract were observed. These are two females, one having received a dose of 1000 mg/kg, the other 2000 mg/kg bw. These deaths occurred, respectively, on the 14th and 21st day. Throughout the duration of the test, no sign of respiratory or neurological suffering was observed.
The variations in body mass of the treated rats compared to the male and female controls are shown in Figure 1 and Figure 2. These results have shown a weight gain between D1 and D28 for all rats with non-significant differences in terms of gain weight between tested rats and control rats.
 Figure 1: Evolution of the body mass of male rats from D0 to D28. View Figure 1
Figure 1: Evolution of the body mass of male rats from D0 to D28. View Figure 1
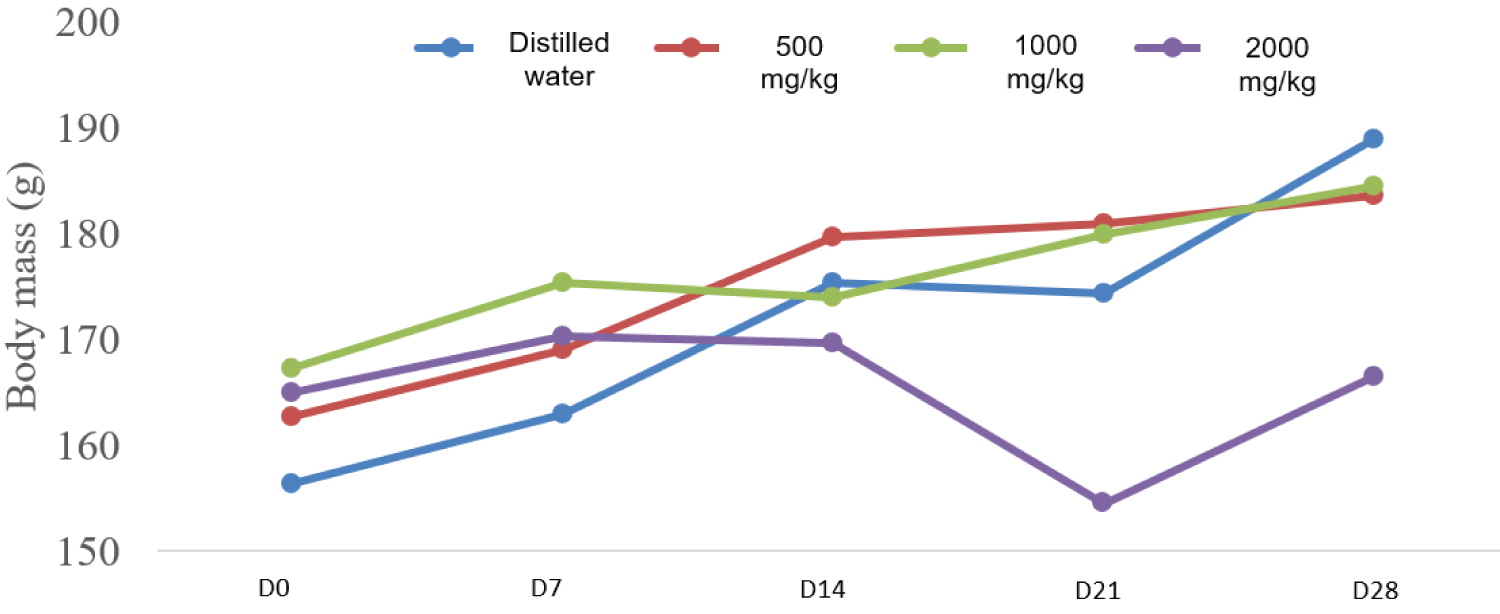 Figure 2: Evolution of the body mass of female rats from D0 to D28.
View Figure 2
Figure 2: Evolution of the body mass of female rats from D0 to D28.
View Figure 2
Table 1 and Table 2 summarize the effect of the aqueous extract of S. kunthianum on the haematological parameters. No significant difference was noted for these parameters in treated rats compared to the rats of the control group.
Table 1: Effects of hydro-ethanolic extract of Stereospermum kunthianum on hematological parameters of male rats. View Table 1
Table 2: Effects of hydro-ethanolic extract of Stereospermum kunthianum on hematological parameters of female rats. View Table 2
The results of the biochemical analyzes carried out are summarized in Table 3. For both hepatic and renal parameters, no statistically significant difference was noted in the treated rats compared to the controls.
Table 3: Effect of the hydro-ethanolic extract of Stereospermum kunthianum on some biochemical parameters of rats. View Table 3
Macroscopic examination showed a stomach distension compared to the control as illustrated in Figure 3. This hypertrophy increases with dose.
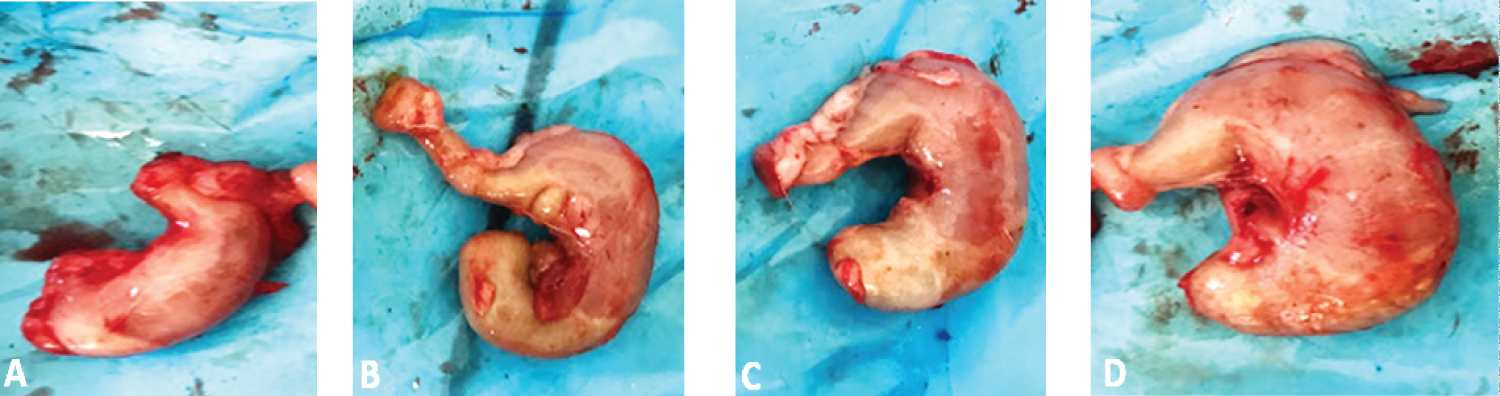 Figure 3: Macroscopic appearance of female rat stomach autopsy. (A) Control; (B) 500 mg/kg; (C) 1000 mg/kg; (D) 2000 mg/kg.
View Figure 3
Figure 3: Macroscopic appearance of female rat stomach autopsy. (A) Control; (B) 500 mg/kg; (C) 1000 mg/kg; (D) 2000 mg/kg.
View Figure 3
In this work, the solvents used for the extraction of active substances were ethanol and water. They made it possible to extract polar compounds such as polyphenols present in the bark of S. kunthianum but also compounds such as triterpenes, coumarins, which would also play a role in the activities noticed for this plant [3,19].
For the acute toxicity assessment, we used females because studies with conventional LD50 tests showed that there is usually no difference in susceptibility between the sexes, but when there are, in females are slightly more sensitive [18].
A limit test at the dose of 2000 mg/kg as recommended by the OECD to categorize a substance as non-toxic, was therefore performed and no deaths were observed during the first 48 hours 14 days after administration of the test substance. Indeed, the limit test is carried out when there are data relating to the absence or very low toxicity of a substance. Previous studies on the acute toxicity of the aqueous extract of the stem bark of S. kunthianum did not show toxicity even after administration of a single dose of 8000 mg/kg body weight [20,21]. This study clearly confirms our results which estimate that the LD50 would be greater than or equal to 2000 mg/kg. According to the Globally Harmonized System, the extract is unlikely to present an oral hazard under normal conditions of use.
For subacute toxicity, our results showed that the hydroethanol extract of the plant would not affect the growth of the animals at the doses tested compared to the controls.
Two deaths were noted in females at 1000 and 2000 mg/kg. These deaths could be related to the extract, but studies with a larger sample size are needed to be able to make a statement. Macroscopic examination showed distension of the stomach of the female rats which was dose dependent, suggesting that the extract may cause long-term damage to the stomach. Food was found in stomach in most of the treated rats, unlike the controls, which would suggest that the hydro-ethanolic extract of S. kunthianum could cause digestive function disorders after repeated intake because the rats had been fasted 24 h before sacrificing. The appearance and color of all other organs were normal.
Biologically, no disturbance of the measured parameters was observed. The results of the biochemical and hematological tests, more precisely the dosage of transaminases, glycemia, uremia, creatinemia, and blood count, showed no statistically significant difference in the control rats compared to the treated rats at different doses. Similarly, weekly urine tests did not reveal glycosuria or proteinuria in either control or treated rats. Our hydroethanolic extract would therefore not cause any impairment of hepatic, renal, or cardiac functions. Indeed, in the event of hepatic, cardiac, or renal lesions, an increase in serum urea-creatinine and transaminase activity is observed [21]. On the other hand, the study by Okpo and Ching found a rise in ALT after administration of an aqueous extract at a dose of 2000 mg/kg [22]. The extraction solvent could explain this difference.
This study provided insight into the safety of the hydro-ethanolic extract of S. kunthianum. A histopathological study would have provided more interesting results. Based on these results, we estimated a NOAEL for this study to be less than 500 mg/kg because even at this dose the stomach of some female rats was affected.
These results suggest that the hydro-ethanolic extract of the trunk bark of S. kunthianum does not cause acute toxicity after administration of a single dose of 2000 mg/kg of body weight. However, repeated administration for 28 days would be the cause of impairment of digestive function at the doses tested. Hence, it has become necessary to carry out a sub-acute toxicity study at doses lower than 500 mg/kg (b/w), to determine the NOAEL.
Authors have declared that no competing interests exist.