Graft-Versus-Host Disease (GvHD) remains a major complication of Allogeneic Stem Cell Transplantation (alloSCT). Anti-Thymocyte Globulin (ATG), however, reduces the incidence and severity of GvHD after alloSCT. A small number of studies suggest a role for ATG in Match-Related Donor (MRD) alloSCT. The aim of this study was to assess the risk of acute and chronic GvHD, engraftment, survival, and mortality in patients who received ATG prior to MRD alloSCT.
A matched-pair analysis was performed among patients treated with MRD alloSCT after GvHD prophylaxis with ATG (ATG+) and MRD alloSCT transplanted patients with cyclosporine/methotrexate prophylaxis (non-ATG). Altogether 11 pairs were identified that could be matched exactly for age, gender, diagnoses, and disease stage at the time of transplantation, cytogenetic and molecular genetic risk group, as well as age and gender of donors. This prospective single-center study includes patients after MRD alloSCT only.
All patients showed stable myeloid and platelet engraftment after alloSCT. On day +28, 91% of ATG+ and 73% of non-ATG patients showed complete donor chimerism (P < 0.03). The difference in acute GvHD was not significant. Chronic GvHD occurred in 60% of patients from ATG+ group and in all patients from control group (P < 0.0001). Extended chronic GvHD was observed in 22% and 50% of patients from ATG+ and non-ATG groups, respectively (P < 0.05). The differences in relapse, mortality, and overall survival in these groups of patients were not significant.
The addition of ATG to conventional GvHD prophylaxis was well tolerated and resulted in donor engraftment in this cohort of patients. Furthermore, conditioning with ATG led to a significant reduction in chronic GvHD without any increase in relapse.
Allogeneic stem cell transplantation, Graft-versus-host disease, Anti-thymocyte globulin, Hematopoietic chimerism
alloSCT: Allogeneic Stem Cell Transplantation; aGvHD: Acute Graft-Versus-Host Disease; AML: Acute Myeloid Leukemia; ATG: Anti-Thymocyte Globulin; BM: Bone Marrow; CDC: Complete Donor Chimerism; cGvHD: Chronic Graft-Versus-Host Disease; CLL: Chronic Lymphocytic Leukemia; CR: Complete Remission; CsA: Cyclosporin; DFS: Disease-Free Survival; DNA: Deoxyribonucleic Acid; EFS: Event-Free Survival; GvHD: Graft-Versus-Host Disease; GvL: Graft-Versus-Leukemia; i.v: Intravenous; MC: Mixed Chimerism; MPD: Myeloproliferative Disorders; MRD: Match-Related Donor; MTX: Methotrexate; MUD: Match-Unrelated Donor; OS: Overall Survival; PB: Peripheral Blood; PCR: Polymerase Chain Reaction; RIC: Reduced Intensity Conditioning; sAA: Severe Aplastic Anemia; STR: Short Tandem Repeat; TBI: Total Body Irradiation; TRM: Transplant-Related Mortality; WBC: White Blood Cells
Allogeneic Stem Cell Transplantation (alloSCT) from HLA-identical siblings or unrelated donors is a potentially curative treatment for several malignant and non-malignant hematological diseases. Graft-Versus-Host Disease (GvHD) remains a major complication of alloSCT that diminishes quality of life and decreases survival [1,2]. Transplant-Related Mortality (TRM) for patients with severe acute GvHD (aGvHD) grades 3-4 is extremely high [1]. Chronic GvHD (cGvHD) affects 30-70% of all alloSCT recipients with a median onset of 4-6 months after alloSCT [3]. Furthermore, cGvHD is associated with more frequent and severe infections. Several programs have been developed to prevent GvHD, including depletion of donor T-cells from the stem cell harvest, in-vivo T-cell depletion, the using of cyclophosphamide and combined immunosuppressive therapy post-transplant [4,5]. As to the latter, the preferred combinations are Cyclosporin (CsA) with Methotrexate (MTX) or tacrolimus and MTX, and these are now considered standard [4]. One way to intensify GvHD prophylaxis without hampering the beneficial Graft-Versus-Leukemia (GvL) is to use Anti-Thymocyte Globulin (ATG). ATG has been primarily used as part of the conditioning regimen for Severe Aplastic Anemia (sAA) patients [6,7]. Currently, ATG is applied for GvHD prophylaxis in patients at high risk of GvHD: this includes unrelated donor transplants, reduced-intensity transplants, and mismatched regimens [8-10]. There are numerous randomized trials and retrospective studies showing that GvHD is less frequent and less severe in patients receiving ATG in the conditioning regimen prior Match-Unrelated Donor (MUD) alloSCT [4,8,11-14]. ATG has been used for the dual purpose of host immunosuppression and of in vivo T-cell depletion. Its efficacy in preventing graft rejection is well established, and it is considered a crucial component in most mismatched transplantation regimens [11-14]. A small number of studies suggest the role of ATG in Match-Related Donor (MRD) alloSCT [10,15,16]. These studies have shown that adding ATG as part of the conditioning regimen before MRD alloSCT can reduce cGvHD without increasing the risk of relapse. The results of a prospective, multicenter, randomized study using ATG as part of a conditioning regimen were recently reported. The authors analyzed 155 acute leukemia patients and showed that using of ATG results in a significantly lower rate of chronic GvHD than the rate without ATG [17].
The aim of our prospective matched-pair analysis was to assess engraftment, the risk of aGvHD, cGvHD, relapse rate, survival, and mortality with or without ATG treatment prior to MRD alloSCT. A general procedure for the evaluation of engraftment after alloSCT is the analysis of hematopoietic chimerism [18,19]. Quantitative assessment of chimerism is particularly important after alloSCT with modified immunosuppression, since engraftment might be delayed and/or supportive measures required [18,20,21]. The conditioning regimens including ATG may initially result in Mixed Chimerism (MC), which gradually develops into Complete Donor Chimerism (CDC).
The reconstitution of immune cell occurs at different time points after alloSCT and can require up to 1 year [22]. Immune reconstitution after alloSCT with ATG as part of the conditioning regimen are rapid for innate immune cells, intermediate for B cells and CD8 T cells, and slow for CD4 T cells and natural killer T cells. In addition, some clinical and transplant factors, such as the number of cells in the transplant, the age of patients, the dose of ATG, are associated with a faster immune reconstitution [23].
The ultimate goal of the present study was to verify engraftment kinetics after conditioning regimens including ATG. This may allow a better understanding of the interplay of conditioning and immunological effects and could lead to improved ATG-based GvHD prophylaxis regimens in the future.
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All animal care was conducted in accordance with the Mayo Clinic Institutional Animal Care and Use Committee Standards. This specific study was approved under protocols A58912 and A49315. All animals were housed, fed, cared for, and euthanized by study personnel and animal husbandry staff of the Mayo Clinic Department of Comparative Medicine.
This prospective, single-center, matched-pair case-control study included patients who underwent MRD alloSCT and were receiving ATG in addition to conventional GvHD prophylaxis (ATG+). The matched-pair control group consisted of MRD alloSCT transplanted patients with cyclosporine/methotrexate prophylaxis (non-ATG).
Eleven pairs were matched for age, gender, diagnoses, and disease stage at the time of transplantation, cytogenetic and molecular genetic risk group, as well as age and gender of donors (Table 1). All patients underwent MRD alloSCT between September 1998 and January 2011 at the University Clinic Charité, Campus Benjamin Franklin, Berlin, Germany. Written, informed consent was obtained from all patients in accordance with the Declaration of Helsinki and the ethical guidelines of our institution.
Table 1: Clinical characteristics of patients according to ATG administration.View Table 1
The patients were given ATG (Thymoglobulin, Genzyme GmbH, Neu-Isenburg, Germany) 4.5 mg/kg Intravenous (i.v.) in divided doses over 3 days. The control group received CsA starting on day -1 at 5 mg/kg i.v. infusion and later switched to an equivalent oral dose. If no aGvHD occurred, CsA was tapered over 4-8 weeks with the intent to discontinue by 3-4 months from alloSCT. In control patients, MTX was given at 15 mg/m2 i.v. on day +1 and 10 mg/m2 on days +3, +6, and +11.
Chimerism analyses were based on the discrimination of donor and recipient alleles on Short Tandem Repeats (STRs) using PCR with fluorescence-labeled primers. Initial genotyping for the detection of informative STR loci was performed using Peripheral Blood (PB) from patient before alloSCT and donor or graft. After alloSCT, chimerism was analyzed on Bone Marrow (BM) and PB samples at day +28, +56, +100, +365, and at the end of examination.
DNA was extracted using a standard DNA extraction method (QIA-Amp; Hilden, Qiagen, Germany) as recommended by the manufacturer. For quantitative chimerism investigation we used AmpFℓSTR® Identifier® PCR Amplification KIT (Applied Biosystems by Life Technologies, Weiterstadt, Germany), which contains fluorescent-labeled primer pairs for simultaneous amplification of 16 different loci each. For quantification of chimerism, the areas under the curves were calculated using Genemapper Version 3.7 software (Applied Biosystems by Life Technologies, Weiterstadt, Germany). The sensitivity of the method is 1%.
The central tendency of the data was measured as the median and the dispersion of values around the median was expressed as the range. Median follow-up was calculated according to recommended criteria [24]. Comparisons between two groups were made using the two-sided exact Fisher test (dichotomous variables). P < 0.05 was considered significant. Overall Survival (OS) and Event-Free Survival (EFS) were estimated by the Kaplan-Meier method. Kaplan-Meier life tables were constructed for survival data and were compared by the log-rank test. Cox proportion hazard regressions were used for performing multivariate analysis for OS, Disease-Free Survival (DFS), and relapse. All calculations were performed using RASW Statistics Software Version (IBM, USA).
White Blood Cells (WBC) engraftment was defined as a count of more than 1.0 × 109/L for three consecutive days. The platelet count was required to be above 50 × 109/L without transfusion for 7 days for engraftment to be documented. Acute GvHD was assessed in patients surviving at least 25 days after alloSCT and graded according to standard criteria [25,26]. Chronic GvHD was assessed in patients who survived after day +100 and scored using the revised Seattle criteria [27,28]. Complete Remission (CR) was defined as a norm cellular BM containing less than 5% blasts and showing evidence of normal maturation of other BM elements. OS was defined as the time from diagnosis to the last contact or death. OS after alloSCT was estimated as the time from alloSCT. For patients achieving CR, DFS after alloSCT was the time from the date of alloSCT to a death in first CR or relapse.
Each group consists of 11 patients (Table 1). Three patients with sAA and three patients with myelodysplastic syndrome, as a matched-pair to sAA, were included in ATG+ and in the non-ATG group, respectively. The mean patient age was 49 (range: 18-73 years).
Five patients (45%) from both groups were transplanted in CR. Six patients from both groups underwent alloSCT in the advanced phases of disease. Cytogenetic and molecular genetic aberrations at the time of diagnosis corresponding to adverse risk group were found in four of 11 (36%) patients in both groups; these were complex cytogenetic aberration and FLT3-ITD mutation.
Conditioning regimens are shown in Table 1. Stem cell source was PB stem cells in all cases. CD34+ cells (x106/kg body weight) were administered in median amounts of 6.4 (range, 3.2-13.6). All patients received prophylaxis for infectious diseases according to the local guidelines. The median donor age was 45 and 49 years in the ATG+ and the non-ATG group, respectively.
Details of the presenting features of the ATG+ group as compared with non-ATG group of patients are given in Table 1. Following alloSCT, all patients showed stable myeloid and platelet engraftment. WBC engraftment was (at median) 16 days and 15 days, respectively. Platelet engraftment varied from 12 to 20 days in the ATG+ group and from 10 to 20 days in the control group. None of the patients suffered graft rejection.
Although analysis of immune recovery was not a primary goal of our study, we revised recovery of T-cells (level of CD3+/CD4+) and B-cell (Immunoglobulin G level). The differences in immune recovery were not significant. All patients with delayed immune recovery suffered bacterial, fungal, or viral infections. Two patients with delayed immune recovery (one from each group) died due to early relapse of disease (data not shown).
On day +28, 91% of ATG+ patients and 73% of non-ATG patients showed CDC (P < 0.03; Table 2). At the final follow-up, chimerism was complete in 82% of patients in both groups. Chimerism status between day +56 and the last follow-up did not reveal any switch of chimerism status (Table 2).
Table 2: Chimerism analysis in patients according to ATG administration. View Table 2
Univariate analysis for chimerism was tested using the variables listed in Table 3. Early MC (at day +28 and +56) was significantly associated with older patients and donor age, sex (males receiving female graft), advanced phase of disease, cytogenetic risk, RIC conditioning, and GvHD prophylaxis without ATG (Table 3). MC at day +100 was linked to small numbers of infused CD34+ cells, high incidence of aGvHD and cGvHD, high relapse rate, high overall mortality and TRM. MC that occurred during 1 year after alloSCT were significantly correlated with small number of infused CD34+ cells, early WBC and platelet engraftments, high relapse rate, high overall mortality, and TRM.
Table 3: Univariate analysis for mixed chimerism. View Table 3
The incidence of aGvHD and cGvHD are reported in Table 4. Grade 1 and 2 aGvHD was reported in three patients (27%) from each group. Severe aGvHD grade 3 and 4 was diagnosed in three (27%) and four (36%) patients from ATG+ and non-ATG groups, respectively. Chronic GvHD occurred in 6 of 10 (60%) patients from ATG+ and in all 10 patients from non-ATG groups, who were alive more 100 days after alloSCT (P < 0.0001). Extended cGvHD was present in two ATG+ patients and in five non-ATG patients group (P < 0.05). One patient from the ATG+ group died due to aGvHD. Two patients (one from each group) died due to cGvHD.
Table 4: GvHD and outcome in patients according to ATG administration. View Table 4
The variables tested in univariate analysis (Table 5) for aGvHD (grades 3-4) and for extensive cGvHD show that patient and donor age, female patient gender, and RIC were significantly associated with higher frequency of severe aGvHD. Higher incidence of severe cGvHD was correlated with patients and donor age, ATG receiving, and CDC at day +100.
Table 5: Univariate analysis for aGVHD and cGVHD. View Table 5
Median follow-up after alloSCT was 35 months. Relapse was diagnosed in 27% (3 of 11) of patients who received ATG and in 45% (5 of 11) of patients from the non-ATG group (P < 0.1; Table 4). Three relapsed patients from the ATG+ group died from relapse. The mortality rate was 54% and 45% at the last follow-up in the both groups, respectively. TRM accounted for 27% (3 of 11 patients) and this was identical in both studied groups.
The median OS after alloSCT was 826 days and 1243 days in patients with and without ATG prophylaxis, respectively. The differences in OS and DFS between patients groups were not significant (P < 0.6 and P < 0.3, respectively; Figure 1). We observed a tendency towards improved DFS in patients who received ATG compared with the control group.
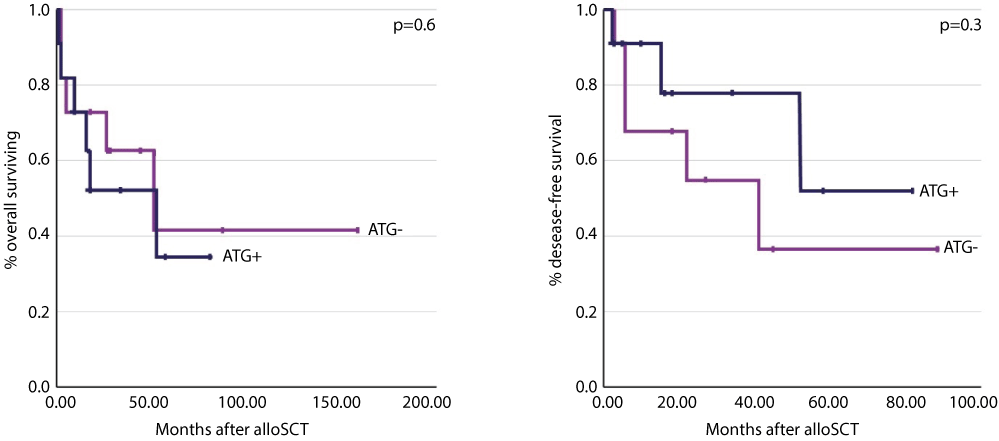 Figure 1: Kaplan-Meier analysis of the OS and DFS according to ATG administration. Comparison of A) OS and; B) DFS after MRD alloSCT in patients with and without conditioning included ATG. OS and DFS in two groups of patients were not significant.View Figure 1
Figure 1: Kaplan-Meier analysis of the OS and DFS according to ATG administration. Comparison of A) OS and; B) DFS after MRD alloSCT in patients with and without conditioning included ATG. OS and DFS in two groups of patients were not significant.View Figure 1
From the multivariate analysis (Table 6), aGvHD (none vs. grade 1-2 vs. grade 3-4), cGvHD (limited vs. extensive), and MC at 1 year after alloSCT were significant prognostic factors for OS. The relapse rate was significantly associated with DFS (P < 0.001) and was correlated with cytogenetic risk group of patients (P < 0.001).
Table 6: Multivariate analysis for OS, DFS, and relapse. View Table 6
In the past decades, the stem cell source for alloSCT has changed rapidly and completely from BM to PB [29,30]. One of the potential problems associated with PB stem cells is that cGvHD occurs with a higher frequency compared with BM [29,31-34]. This is a serious concern because severe aGvHD and cGvHD are the most common causes of death after alloSCT. A further concern for surviving patients with severe GvHD is an extreme reduction in quality of life [2]. ATG has been primarily used as part of the conditioning regimen for sAA patients [6,7] and has been used successfully in unrelated stem cell transplantation [8,9,11-14].
The efficacy of ATG treatment for GvHD prevention is not exactly understood. Despite its use over more than 30 years in alloSCT, the impact of ATG prevention on outcome after transplantation is still under debate. Moreover, there are a few data about efficacy of ATG before MRD alloSCT. In 2016, the results of prospective, multi-center, randomized study including significant cohort of acute leukemia patients were published [17]. The authors suggested that including ATG in the myeloablative conditioning regimen resulted in a significantly lower rate of chronic GvHD without decreasing of OS and DFS. However, the rate of a composite end point of survival free from chronic GvHD and survival free from relapse was significantly higher with ATG [17]. In contrast with this trial, our study is single-center matched-pair analysis. In our study, we did not observe any graft failures in the ATG+ and non-ATG groups and none of the patients suffered graft rejection.
Engraftment was confirmed using chimerism analysis. Appearance of CDC at day +28 after alloSCT was found in a significantly greater proportion of patients receiving ATG (P < 0.03; Table 2). In addition, chimerism analyses between day +56 and the last follow-up did not reveal any switch of chimerism status. Despite the small number of patients, we found a significant association between MC and some clinical figures (Table 3). Early MC (day +28 and +56 after alloSCT) was associated with older patients and donor age, gender (males receiving female graft), advanced phase of disease, high cytogenetic risk, RIC, and non-receipt of ATG in our study. MC at day +100 was associated with all data excepting GvHD prophylaxis, donor age and gender, WBC and platelet engraftment. Contrarily, mixed chimerism 1 year after alloSCT was correlated with the number of CD34+ infused cells, WBC, and platelet engraftment, relapse rate, overall mortality, and TRM. On multivariate analysis, MC at 1 year after alloSCT was associated with OS, DFS, and relapse rate (Table 6). Our data confirmed that chimerism analysis is a very important parameter in patients after alloSCT that not only helps to identify relapse, but is also correlated with engraftment and reflects the interaction of GvHD and GvL effects. The latter is substantial in patients after alloSCT with modified conditioning and GvHD prophylaxis, which relies on anti-leukemic effects to cure leukemia.
We did not found any differences in the time of immune recovery, as well as in incidence of infection complications in both groups. First, it may be due to a small number of patients. In addition, other factors, such as the number of CD34+ infused cells, conditioning and some others, could influence immune recovery [23]. Moreover, pronounced aGvHD are closely associated with delayed immune recovery. GvHD effects can contribute to impaired intrinsic B-cell functionality and Immunoglobulin production in late onset patients [35]. We have found delayed B cell recovery in all patients with aGvHD 3-4 grade. Moreover, we have found association between delayed B-cell recovery and early relapse. As expected, infectious complications were more often in patients with delayed immune recovery.
We investigated the frequency of both acute and chronic GvHD. The incidence of aGvHD was similar between the two patient groups. On univariate analysis, incidences of severe aGvHD were significantly associated with age of patients and donors, and RIC conditioning. We found that incidence of cGvHD was significantly lower in patients from the ATG+ group (P < 0.0001), and is consistent with previous randomized studies [13,17]. Moreover, extensive cGvHD (grades 3-4) was significantly less frequent in the ATG+ patients' group (P < 0.05). Statistical analysis shows that extensive GvHD was significantly higher in older patients and in patients receiving a transplant from an older donor. Although GvHD prophylaxes with ATG was not an independent prognostic factor for extensive cGvHD on multivariate analysis (data not shown), these parameters were significantly correlated in the univariate analysis (Tables 4 and Table 5).
None of the numerous retrospective studies has reported changes in survival in patients receiving ATG [10,15-17]. The findings of this prospective study were consistent with these studies in that TRM, relapse rate and OS were similar with or without ATG administration. We observed a tendency towards improved DFS in patients who received ATG compared with the control group (P < 0.3; Figure 1). Although some clinical features listed in Table 6 were significantly correlated with OS on univariate analysis, in the multivariate analysis, only incidence of aGvHD, appearance of extensive cGvHD, and MC 1 year after alloSCT were significantly linked with OS. The relapse rate was significantly associated with DFS (P < 0.001) and was correlated with the cytogenetic risk group of patients (P < 0.001).
This prospective matched-pair study demonstrated that the addition of ATG to conventional GvHD prophylaxis was well tolerated and resulted in donor engraftment in all evaluable patients. Furthermore, the administration of ATG significantly reduced the incidence of severe cGvHD without increasing relapse in MRD alloSCT. Although our study is limited to small number of patients, it does not contradict the data of multi-center study and confirms therapeutic effect of ATG in patients after match-related alloSCT. We conclude that using ATG may help to decrease the rate or severity of GvHD. This may not only allow a better understanding of the interplay of conditioning and immunological effects but may also lead to improved ATG-based GvHD prophylaxis regimens in the future.
Written informed consent was obtained from all patients in accordance with the Declaration of Helsinki and the ethical guidelines of our institution.