Pediatric Ulcerative Colitis (UC) is an Inflammatory Bowel Disease (IBD) that has potential morbidities, lifelong challenges and can leave both physical and psychosocial impacts. This study aimed to review the epidemiology, clinical presentations, and way of diagnosis, therapeutic approaches, complications and outcome of pediatric patients with UC.
A retrospective review of all patients diagnosed with UC in the pediatric department, Salmaniya medical complex, Bahrain, between 1984 and 2017 was conducted. Annual incidence and cumulative prevalence were calculated. Data about demographic and clinical characteristics; laboratory, radiological, endoscopic and histological investigations; medical therapy, number of exacerbations and hospital admissions, complications and outcomes were collected.
Out of 108 pediatric patients diagnosed with IBD, 58 (53.7%) patients were found to have UC. The estimated annual incidence was 1/105/year (range from 0 to 6 patients). The prevalence was 16.3 patients per 100,000 pediatric populations. Thirty-three (56.9%) were females and 25 (43.1%) were males. Median age at the time of study was 22.9 years (range 5.8 to 41.8). The most common clinical presentations were bloody diarrhea and recurrent abdominal pain. Family history of IBD was found in five patients. Twenty-four (41.4%) patients had associated diseases. Ten (71.4%) patients had positive Anti-Neutrophil Cytoplasmic Antibodies (ANCA) test. Forty (81.6%) patients had pancolitis. The most frequent medications were prednisolone, mesalazine and azathioprine. Biological therapies were used in eight. Surgical intervention was required in nine. Four patients had colectomy. Two patients died at the age of 17 and 26 years. Both had an associated liver diseases and one died because of colon cancer.
The study showed a significant increase in incidence and prevalence of UC in Bahrain. Female predominance and a high association with autoimmune diseases might support the autoimmune theory as a cause for the disease. Despite medical therapy, significant morbidities and mortalities were reported.
Children, Inflammatory bowel disease, Ulcerative colitis, Prevalence, Outcome
Ulcerative Colitis (UC) is a chronic Inflammatory Bowel Diseases (IBD) [1-6] that is characterized by relapses and remissions [2-4,6-8]. It mainly affects the mucosa and submucosa of the colon [8]. The aetiology of the disease is unclear and thought to be multifactorial [2,4,8]. Several factors may play a role in its development like genetic predisposition, environmental factors [2,4,8], immune dysregulation and bacterial flora [8].
UC can affect both adult and children [2]. Around one-quarter (25%) of patients presents in childhood [7,9]. Most children are diagnosed between 11 and 16 years of age and those who are diagnosed earlier were found to have a more severe disease [10]. Pediatric UC is a disorder that has potential morbidities and can leave both physical and psychosocial impact on children [2,5,9]. The diagnosis of pediatric UC is based on classical history, physical examinations, laboratory investigations, imaging, endoscopy and histopathology [11]. The incidence and prevalence of UC were more in Western countries but are increasing elsewhere in the world nowadays [8,9]. Studies about pediatric UC in the Middle East are scarce and up to date there were no published studies about pediatric UC coming from the Kingdom of Bahrain. The aim of this study is to review the prevalence, clinical presentations, and ways of diagnosis, different therapeutic approaches, complications and outcome of pediatric UC patients in Bahrain.
A retrospective review for all medical records of IBD patients diagnosed in the pediatric department, Salmaniya medical complex, Kingdom of Bahrain, between January 1984 and March 2017 was conducted. All pediatric (≤ 18 years) UC patients were included in the study and no patient was excluded. Annual incidence and cumulative prevalence of the disease were calculated. Data about gender, nationality, birth weight, admission weight and height, age at presentation, age at diagnosis, duration of the illness, family history of IBD, history of contact with a smoker, associated medical diseases and initial clinical presentations were collected. Investigations like full blood count, Erythrocytes Sedimentation Rate (ESR), C-Reactive Protein (CRP), liver function tests, Antineutrophil Cytoplasmic Antibodies (ANCA), Anti-Saccharomyces Cerevisiae Antibodies (ASCA), serum iron and ferritin levels, serum folate and vitamin B12 levels, serum 25-hydroxyvitamin D levels, helicobacter pylori serology and cultures, Purified Protein Derivatives (PPD) test, stool microscopy and cultures results were also collected. The results of radiological images including chest X-ray, abdominal ultrasound, small bowel series, abdominal Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) were gathered. Endoscopic findings using Olympus (PCF-230 and XQ-230) or Pentax endoscopes (EG-2901 and EC-380IF) and histopathological findings using hematoxylin and eosin stain to read the gastrointestinal biopsies were also collected. After excluding Crohns’ disease, infective and allergic causes of colitis, the diagnosis of UC was confirmed using clinical, biochemical, radiological, endoscopic and histopathologic findings. We adopted the criteria published by IBD working group of the European Society of Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN) to confirm the diagnosis [12,13]. Data about medical therapy received by each patient especially medications used to control UC whether at the time of diagnosis or during the follow up course, number of exacerbations, and number of hospital admissions, complications and outcomes were gathered.
The patients' data were initially entered into excel sheet then transferred to and analysed by Statistical Package for Social Sciences (SPSS) version 21. The frequencies and percentages were calculated for demographic data. Continuous variables were explored for normal distribution using Kolmogorov-Smirnov and Shapiro-Wilk tests. Grouped data are presented as mean and Standard Deviation (SD) for normally distributed variables or median and range for non-normally distributed variables. Kruskal Wallis test was used to compare the incidence between three time periods set every ten years. P value was considered to be statistically significant if less than 0.05.
This study was conducted in accordance with the principles of Helsinki Declaration and it was ethically approved by the secondary care medical research subcommittee, Salmaniya Medical Complex, Ministry of Health, Kingdom of Bahrain.
Out of 108 IBD patients diagnosed during the study period, 58 (53.7%) pediatric patients were found to have UC. The prevalence of UC in Bahrain was 16.3 patients per 100,000 people under 15 years of age. According to 2014 statistics, the total population of Bahrain was 1,314,564. Children less than 15 years of age accounted for 20.5% of the total population which was around 269,485 children. The cumulative prevalence of UC in Bahrain was shown in Figure 1. The estimated median annual incidence of UC was 1/105/year (range from 0 to 6 patients). The median incidence of UC patients between 1987 and 1996 was 1/105/year (range 0 to 3 patients). The median incidence of UC patients between 1997 and 2006 was 1.5/105/year (range 0 to 4 patients) and the median incidence between 2007 and 2016 was 2.5/105/year (range 0 to 6 patients). There was a significant statistical difference between the three time periods (P ≤ 0.0001). Demographic data and clinical characteristics of UC patients are presented in Table 1. Thirty-three (56.9%) were females and 25 patients (43.1%) were males. Fifty (86.2%) were Bahraini Arabs and eight (13.8%) patients were non-Bahraini; Asian and Arabs (two Indian, two Pakistani and one Saudi, one Qatari and one Syrian). All patients were born normally with no history of caesarean section deliveries. The median age at the time of study was 22.9 years (range 5.8 to 41.8 years). Family history of IBD was found in five (18.5%) patients, two were first degree relatives and three were second degree relatives.
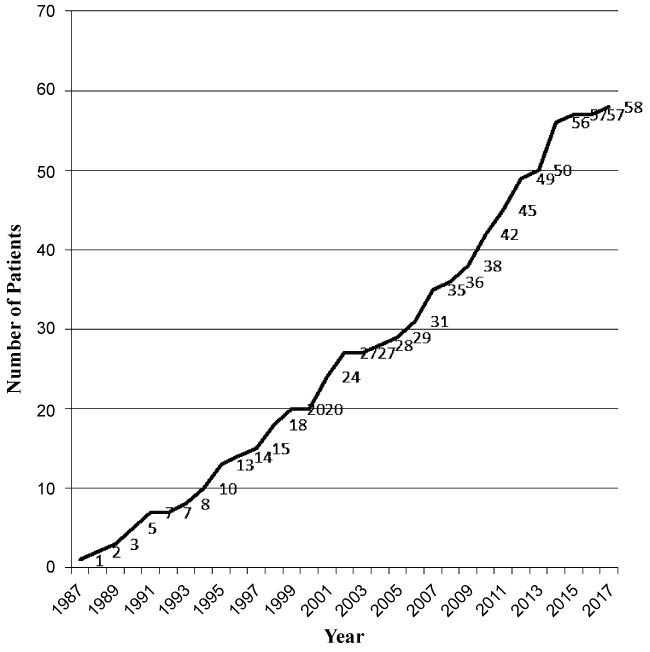 Figure 1: Cumulative prevalence of pediatric ulcerative colitis in Bahrain.
View Figure 1
Figure 1: Cumulative prevalence of pediatric ulcerative colitis in Bahrain.
View Figure 1
Table 1: Demographic data of 58 pediatric patients with ulcerative colitis. View Table 1
The most common initial clinical presentations were bloody diarrhea and recurrent abdominal pain (Table 2). Arthritis was found in two patients. Skin rash was noted in four patients, one of them had erythema nodosum but no patient had pyoderma gangrenosum. Eye involvement and clubbing were found in one patient.
Table 2: Clinical presentations of 58 pediatric patients with ulcerative colitis. View Table 2
Twenty-four (41.4%) patients had associated diseases. Eight (30.8%) patients had a significant anaemia, three of them were of autoimmune haemolytic anaemia type, four patients had glucose 6 phosphate deficiency and three were sickle cell trait. Overlap syndrome (Primary Sclerosing Cholangitis (PSC) and autoimmune hepatitis) was found in three patients while PSC alone and autoimmune hepatitis alone was found each in one patient. Insulin dependent diabetes mellitus was found in two patients. Growth hormone deficiency, vitiligo, vasculitis, nephritis, pericardial and pleural effusion, spondylopathies, bronchial asthma, food allergy, vitamin D deficiency, diaphragmatic hernia, seizure disorder, mental retardation, psychosis, developmental delay; each noted in one patient.
Laboratory features at the initial presentation of the disease are shown in Table 3. Low haemoglobin level was found in 40 (85%) patients with a mean of 10.5 grams per decilitre ± 2.5 SD (normal range 13 to 16.5 g/dl). Hypoproteinemia was found in six (13.6%) patients with a mean of 76.2 grams per litre ± 11.2 SD (normal range 64 to 82 g/l) and low vitamin D level in all tested seven patients with a mean of 39.1 nmol per litre ± 6.1 SD (normal range > 50 nmol/l). A PPD test and chest X-ray to exclude early tuberculosis exposure were done in all patients and all were negative. Three patients had liver biopsy performed to diagnose the associated liver diseases.
Table 3: Results of laboratory investigations at presentation of 58 pediatric patients with ulcerative colitis. View Table 3
Thirty-five (60%) patients had at least one radiological imaging whether abdominal ultrasound, barium study or abdominal computed tomography. Twenty (57%) of them had positive findings suggestive of UC. Radiological, endoscopic and histopathological data are shown in Table 4. Out of 26 (52%) patients who had upper gastrointestinal endoscopy, 17 (65.4%) patients had available reports of the endoscopy and 15 (88%) patients had positive findings. Oesophageal involvement was noted in nine patients (mild distal erythema in seven, small nodules in three and superficial small ulcers in two), gastric in 13 patients (mild erythema in nine, fine nodularity in seven, haemorrhagic spots in four and small tiny shallow ulcers in three) and duodenal in seven patients (erythema in three, fine nodularity in two and haemorrhagic spots in two). All the 58 UC patients underwent colonoscopy, but colonoscopy reports were available for 49 (84.5%) of them. Nine patients had no available reports. However, two of them had upper gastrointestinal endoscopy report available, two had positive barium studies suggestive of UC and one had positive ANCA and was on UC treatment. The nine patients were included only in the epidemiological data but not in the analysis of the rest of the data.
Table 4: Radiological, endoscopic and histopathological investigations of 58 pediatric patients with ulcerative colitis. View Table 4
Upper gastrointestinal histopathology reports were available for 25 (43%) patients, 23 (92%) had positive results. The results were positive in 12 (60%) out of 20 esophageal biopsies (reflux esophagitis in five, nonspecific chronic active esophagitis in four and mild congestion of squamous epithelium in three biopsies), 22 (88%) out of 25 gastric biopsies (nonspecific chronic active gastritis in 19, mild congestion in two and mild focal eosinophilic infiltrate in one) and 10 (50%) out of 20 duodenal biopsies (all had mild chronic active duodenitis). Colonic histopathology reports were available for 48 (82.7%) patients. Cryptitis, chronic active colitis, crypt abscess and crypt distortions were the main findings. Other nine findings were also founding in the tissue biopsies (crypt associated granulation tissue in eight biopsies, crypt branching in five, mucodepletion in four, focal Paneth cell proliferation in three, loss of crypts in two; and oxyphilic metaplasia, neuromatoid proliferation, ileitis and polypoid changes each in one biopsy).
The most frequently used medications were prednisolone, mesalazine and azathioprine (Table 5). Biological therapies were used in eight patients. No patient received cyclosporine or methotrexate.
Table 5: Medical therapy of 58 pediatric patients with ulcerative colitis at the time of diagnosis and during the follow up course. View Table 5
During the study period, 12 patients had at least one UC relapse over time. The median number of exacerbation episodes per patient was one episode ranging from zero to three episodes. Out of 61 admissions, 16 admissions were due to UC (26%). The median admission rate was one admission per patient ranging from one to 20 admissions. Three patients developed steroid dependence. Two patients required blood transfusion. Periorbital cellulitis, pneumonia, salmonella gastroenteritis, azathioprine induced pancreatitis and osteopenia each reported in one patient. Surgical intervention was required in nine patients. Four patients had colectomy (total colectomy in three and subtotal in one) due to severe disease and very poor response to medical treatment before the era of biologic therapy in two patients and with failure of biologic therapy in another two patients. Cholecystectomy, inguinal hernia repair, dermoid cyst removal, tonsillectomy and diaphragmatic hernia repair each in one patient.
The median follow-up period was 11 years (range 0.1 to 28.8 years). Two female patients with UC died at the age of 17 and 26 years of age respectively. Both of them had an associated liver disease, one had AIH and the other had PSC. The later patient refused colectomy and died because of colon cancer.
The incidence and prevalence of IBD is increasing world wide [8,9]. In the gulf countries, the incidence is showing an increasing tendency [1]. In this study, the incidence of UC had showed a rising trend mounting from 1/105/year between 1987 to 1996 to reach 2.5/105/year in the period between 2006-2016. This incidence is higher than Kuwait where Al-Qabandi, et al. [14] reported a total of 36 pediatric patients with UC which showed an estimated annual incidence of 0.6/105/year (CI 95%, 0.34-0.85). However, Al-Mofarreh and Al-Mofleh [11] studied 238 Saudi UC patients and found a steady stat in UC. The incidence of IBD usually peaks in late adolescence and early adulthood [5]. In this study, 41% of the patients presented between the age of 10 and 15 years. The prevalence of pediatric UC in our study was 16.3 per 100,000. It is lower than that reported by Ludvigsson, et al. [15] which was 25 per 100,000 on studying 585 UC pediatric patients from Sweden.
Like Al-Qabandi, et al. [14] study, this study showed female predominance. In contrast to Al-Mofarreh and Al-Mofleh [11] who found male predominance. The presence of family history was 18.5% in this study which is lower than the number reported in Kuwait 31% [14] but higher than the number reported by AlSaleem, et al. (9%) [16] who studied 188 patients in Saudi Arabia.
The clinical presentation of UC patients in this study was similar to other studies were bloody diarrhea and abdominal pain are the most common presentations [2,9,11,14,16,17]. For instance, Al-Qabandi, et al. [14] reported bloody diarrhea in 94% and abdominal pain in 78% of UC patients. The abdominal pains are attributed to intestinal distension secondary to the disease itself, to fibrotic stricture, adhesions or following surgical interventions [17]. Thirty-eight per cent of UC patient in this study had history of weight loss at presentation. Similarly, El Mouzan, et al. [18] reported a history of weight loss in 42% of 119 newly diagnosed pediatric patients with UC. This weight loss can be explained by poor dietary intake, increase gastrointestinal losses and increased energy expenditure due to chronic inflammatory activity [9,19]. Due to the malnutrition, the patients sometimes diagnosed as anorexia nervosa [9]. However, compared to Crohn’s disease, UC had shorter interval between onset of symptoms and signs and the diagnosis. It also had less systemic inflammation, therefore less effect on bone, appetite and nutritional status [14,20]. Children with IBD require detailed nutritional assessment on a regular basis [20].
Patient with UC are also at risk of having extra intestinal manifestations such as erythema nodosum and pyoderma gangrenosum that might be linked to genetics, inflammatory cytokines, bacteria/bacterial products or medications [2]. Liver also could be involved in patients with UC in the form of drug-induced hepatitis, autoimmune hepatitis or sclerosing cholangitis [2]. Musculoskeletal manifestation in the form of mild arthralgia or severe spondyloarthropathies especially in children positive for HLA-B27 had been also reported [2]. Sixteen (27.6%) patients in our study had an associated autoimmune disease including autoimmune hemolytic anemia, overlap syndrome, primary sclerosing cholangitis, autoimmune hepatitis, insulin dependent diabetes mellitus, vitiligo, nephritis and spondylopathies. This observation might support the immune theory as a cause for UC.
The etiology of UC is unclear. Significant factors play a role in its development such as the environmental factors, genetic variations, altered intestinal microbiota and the disturbances in the innate and adaptive immune response [3,4,21]. People living in the western countries are more susceptible to the disease and those living in underdeveloped countries might be protected from the disease due to the high exposure to the parasitic infestation [3]. The genetic overlap between UC and other autoimmune disease like psoriasis and ankylosing spondylitis is well established [22]. The interaction between the microbiome and the immune system seems to be important in the pathogenesis of IBD [23].
The standard laboratory tests used to diagnose UC in children are similar to those in adults [9]. Although some pediatric UC patients, especially those with mild disease, will have no laboratory abnormalities detected, the presence of iron deficiency anemia, elevated inflammatory markers e.g. ESR, CRP and fecal calprotectin, hypoalbuminemia and negative stool cultures are suggestive of IBD [2,9]. In this study, anemia was noted in 85% of patients. Similarly, AlSaleem, et al. [16] reported anemia in 75% but Al-Mofarreh and Al-Mofleh study [11] showed anemia in only 37%. In our study, high ESR and CRP was noted in 51% and 57% respectively. This is lower than that reported in Saudi patients (82% and 63% respectively) [16]. Hypoalbuminemia was seen in 47% of patients in this study compared to that reported by AlSaleem, et al. [16] which was 33%. Liver function tests can be elevated in UC patients either due to the disease activity or due to extra-intestinal complications like sclerosing cholangitis [2]. Even though serological test is available to screen for IBD, they fail to diagnose pediatric patients or give false positive results [2]. In this study, ANCA and ASCA tests were positive only in 71% and 66.7% of patients respectively. Patients with UC are more prone to infections either due to the disease itself or due to the use of immunosuppressive medications [3,24]. Bacterial infections should be excluded first in any pediatric patient presented with bloody diarrhea [25]. UC patients should be screened for Latent TB Infection (LTBI), hepatitis B, hepatitis C and HIV viral infections [20].
Radiological evaluation of children suspected to have UC should be individualized [2]. Abdominal ultrasound in UC can detect bowel wall thickening, peri-intestinal inflammation and ileus [26] and can be used to detect IBD in children with high success [25,26]. Although upper gastrointestinal study with Small Bowel Follow-Through (SBFT) is the main radiological modality in IBD patients, its information on the presence or the extent of disease are limited [2]. CT and MRI can provide better visualization of the inflamed mucosa and peri-intestinal involvement [2,21,25]. Ideally, all pediatric patients suspected to have UC should have endoscopic procedure (upper and lower gastrointestinal endoscopy and biopsies) to confirm the diagnosis [21,25]. Most of our patients had pancolitis (81.6%) on colonoscopy which is higher percentage than that reported by Al-Qabandi, et al. [14], Docktor, et al. [23] and AlSaleem, et al. [16] which was 64%, 55% and 46.1% respectively. This might be explained by either the late presentation or the severity of the disease in our population.
Treatment of UC includes both medical and surgical approaches [3]. Although advances in medicine have creates drugs to control UC and improve quality of life [1]. In this study, most of our patients received steroids (77.3%) followed by mesalazine (59.1%). This is unlike Virta and Kolho [20] and Ludvigsson, et al. [15] studies where most of their patients received aminosalicylates (94% and 81% respectively) more than steroids (72% and 44% respectively). Medical treatment of children with UC should consider both induction and maintenance phases [2,3]. Most clinicians rely on corticosteroids for induction of rapid remission [2,3,17,19,27,28]. However, one third of patients will fail to respond to corticosteroids [3]. The choices of maintenance medications will depend on the location of UC and the clinical severity [2]. Aminosalicylates (e.g. mesalazine), immunomodulators (e.g. azathioprine) and biologic agents (e.g. Infliximab (IFX) and adalimumab) are the main maintenance drugs used [2,3,17,27,28]. Mesalazine, at a dose of 2.4-4.8 g/day, is an effective therapy for patients with mild to moderate UC [3]. If no response to conventional therapy, Cyclosporine (CSs) has been used to treat severe flare-ups of UC [3]. In our study, azathioprine was used in 50% of patients higher than Virta and Kolho [27] where it was used in 30.4%. Azathioprine and 6-mercaptopurine are effective drugs for both induction of remission and maintenance [3]. Methotrexate (MTX) is another treatment option but it’s role in pediatric UC is not yet established [19]. When moderate to severe patient did not respond to aminosalicylates, CSs or immunosuppressive medications, biologic agents like anti-TNF-α monoclonal antibodies are recommended to be used [3]. Biological therapy use in the treatment of UC patients has significantly increased over the last decade [19]. It appears to be safe and well tolerated. It has improved disease control, patient growth and quality of life by pediatric patients with UC [3,7]. Infliximab (IFX), adalimumab, certolizumab pegol and golimumab are the current four anti-TNF-α agents used to treat UC patients [19,27,29]. IFX is an effective treatment for pediatric UC [3,7,19,23]. Anti-TNF-α drugs share the same pre-treatment protocol but they have different routes and times of administrations [27]. The decision to introduce anti-TNF-α agent is based on the disease severity, the presence of co-morbidities, direct and indirect costs of medical care, and possible benefits and risks [27]. Before starting anti-TNF-α therapy, a precise assessment of UC activity is crucial [29].
Compliance with medications is a major concern in children with UC [2]. Unlike adult UC, the relapses of pediatric UC have a damaging effect on children’s linear growth and pubertal development [2,19]. Steroid therapy can lead to osteopenia, osteoporosis and avascular necrosis [2,19]. Biological therapy has potential serious side effects like malignancy and infections and should be avoided in patients with acute infection [3,7]. The patients should be closely monitored for neutropenia and leucopenia to avoid life-threating infections especially if there were on biological therapy or other immunosuppressive medications [7].
Like any other retrospective studies, this study was limited by missing some data related to patients with UC like the history of consanguinity and the patients’ compliance with medications. In addition, the small number of the UC patients was also considered as limitation. However, this study is the first study from Bahrain to shade light on pediatric UC patients. This study can form a great source of information for any future studies tackling the same group of patients.
In conclusion, this study showed an increasing prevalence of pediatric UC in Bahrain. Female predominance and a high association with autoimmune diseases among patients with UC might support the autoimmune theory as a cause for this disease. Abdominal pain, bloody diarrhea and weight loss were the main presenting symptoms. Most of the patients had severe disease with pancolitis. Despite medical therapy, significant morbidities and mortalities were reported. Further multicenter studies focusing on the nutritional status, compliance with medications and the effect of the disease on the quality of life are needed for patients with a chronic disease like UC.
The authors gratefully acknowledge all those who provide care for patients with UC at the pediatric department, Salmaniya Medical Complex, kingdom of Bahrain.
None.
The authors declare no conflict of interest.