We reported two pediatric rare cases with massive splenic infarction that in both of them the interspersed hyperechoic linear striation of the splenic parenchyma was first suggestive sign of infarction, and diagnosis was confirmed by color Doppler and surgery.
Pediatric, Splenic Infarction, Ultrasound
Massive splenic infarction (MSI) is characterized by vessel occlusion, parenchymal ischemia and subsequent tissue necrosis involving more than half of the spleen. Etiologies of MSI include Sickle cell disease and Sickle cell variants, coagulopathies, therapeutic embolization of the spleen, sarcoidosis, hematological malignancies and organ transplant. Infection has also been linked to MSI [1]. Other unique causes of splenic infarction include Gaucher disease, pancreatitis, splenic artery aneurysm, septic emboli in endocarditis and collagen vascular diseases [2].
In this report, we present two cases of massive splenic infarction with specific sonographic pattern include two rare etiologies: 1) Fungal endocarditic with predisposing chronic granulomatous disease. 2) Spontaneous and associated with congenital diaphragmatic hernia.
A 9-year-old girl presented at our institution with diffuse abdominal pain, low grade fever, vomiting and failure to pass the stool. Her past medical history was normal with no disease. On admission, the patient was alert but looking ill and mildly agitated. She had tachycardia and fever. In physical examination, diffuse abdominal tenderness was detected with no rebound tenderness and guarding. Laboratory findings showed leukocytosis (WBC = 13800) with PMN = 86% and mild anemia (Hb = 11.7), thrombocytosis (PLT = 704000), increased ESR (ESR = 28) and LDH = 600. After resuscitation, the patient underwent urgent laparotomy with the diagnosis of peritonitis. In the abdominal exploration, 200 cc serous secretions including a lot of fibrin was drained and sent for pathology. The entire of GI tract had serositis and generalized inflammation of intestinal loops and peritoneum was seen. The omentum had edematous and hemorrhagic appearance. A lot of fibrin was seen in peritoneal cavity without known origin for rupture. Appendectomy and omental resection was done, and second look was recommended after 48 h.
In post-AP chest X-ray and upper GI series revealed hiatal hernia in the association with gastric volvulus and Morgagni hernia (Figure 1). The ultrasound was showed the mild enlargement of spleen with the longitudinal diameter of 126 mm that had heterogeneous echo pattern. The high frequency scanning with superficial probe was show diffuse hypoechogenicity of the splenic parenchyma with interspersed hyperechoic linear striation (lacy appearance). Splenic capsule was thick, and an irregular subscapular homogenous hyperechoic area is observed at upper pole. No vascular flow detected in spleen capsule and parenchyma on color Doppler assessment (Figure 2). Also, diaphragmatic defects and associated hernia was obvious on the ultrasound.
 Figure 1: AP Upper GI view shows hiatal hernia with intra-thoracic stomach. There is a tubular gas pattern in right cardio-phrenic angle that in lateral view revealed associated Morgagni hernia.
View Figure 1
Figure 1: AP Upper GI view shows hiatal hernia with intra-thoracic stomach. There is a tubular gas pattern in right cardio-phrenic angle that in lateral view revealed associated Morgagni hernia.
View Figure 1
 Figure 2: Sonographic pattern of spontaneous hemorrhagic splenic infarction in associated with diaphragmatic hernia: A) Mild splenomegaly with heterogeneous echo pattern; B) Diffuse hypoechogenicity of the splenic parenchyma with interspersed hyperechoic linear striation and irregular subscapular homogenous hyperechoic area; C) No vascular blood flow detected in splenic parenchyma. Capsule is thick.
View Figure 2
Figure 2: Sonographic pattern of spontaneous hemorrhagic splenic infarction in associated with diaphragmatic hernia: A) Mild splenomegaly with heterogeneous echo pattern; B) Diffuse hypoechogenicity of the splenic parenchyma with interspersed hyperechoic linear striation and irregular subscapular homogenous hyperechoic area; C) No vascular blood flow detected in splenic parenchyma. Capsule is thick.
View Figure 2
The patient went to the operation room for the second time. The spleen was in normal position and non-wandering. It had an infarcted appearance and splenectomy was done. The intra-thoracically herniated gastric volvulus and some intestinal loops were seen and returned to abdominal cavity to the normal position. Then, the diaphragmatic defects were repaired. Macroscopic and histopathologic findings was confirmed the diagnosis of diffuse hemorrhagic necrosis of spleen.
The second case was a 3.5-years-old male child a known case of chronic granulomatous disease that was admitted with restlessness, anorexia, high grade fever and multiple necrotic lesions on hands and feet (ecthyma gangrenosum). Liver was palpable 2 cm below costal margin and spleen was palpable in deep inspiration. His family history wasn't significant. He was received broad spectrum antibiotics without response. Echocardiogram showed large vegetation on mitral valve and blood PCR for fungal infection was positive. Other laboratory tests were within normal limits.
Abdominal sonography showed normal position and size spleen (longitudinal diameter: 65 mm) with subcapsular hypoechoic shadow with 7 mm diameter. Intersperse linear echogenicity was seen in parenchyma. Two wedge shape hypoechoic foci and intersperse linear striation was seen in parenchyma. Despite hypervascularity of surrounding soft tissue, no vascular blood flow was seen in splenic parenchyma (Figure 3A and Figure 3B). Except mild hepatomegaly, otherwise ultrasound finding was normal.
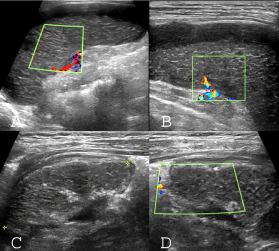 Figure 3: Sonographic pattern of splenic infarction in CGD case: A,B) Normal size spleen with peripheral and subscapular hypoechoic area; the splenic parenchyma has an interspersed hyperechoic linear striation and no vascular blood flow detected in splenic parenchyma. C,D) Follow up ultrasound images; decrease of spleen size and shrinkage border with capsular and parenchymal calcification without significant revascularization.
View Figure 3
Figure 3: Sonographic pattern of splenic infarction in CGD case: A,B) Normal size spleen with peripheral and subscapular hypoechoic area; the splenic parenchyma has an interspersed hyperechoic linear striation and no vascular blood flow detected in splenic parenchyma. C,D) Follow up ultrasound images; decrease of spleen size and shrinkage border with capsular and parenchymal calcification without significant revascularization.
View Figure 3
Interestingly, severe abdominal pain or guarding was not a significant symptom and it was responded to conservative management. Amphotericin B liposomal was added to therapeutic regime. He discharged with good general status.
In second visit on 3.5 month next, the child was well being and without any abdominal discomfort. The follow up ultrasound shows spleen size to decrease (42 mm) with shrinkage border, capsular and paranchymal calcification, and without significant revascularization (Figure 3C and Figure 3D).
Few reports of splenic infarction of various etiologies including hematological and non-hematological causes have been published. Massive splenic infarction (MSI) is an extremely rare condition [3]. Focal splenic infarcts are common in patients with Sickle cell anemia (SCA) but these are usually small and repetitive leading ultimately to autosplenectomy. The few case reports published in literature that MSI can also be associated with Sickle cell anemia [3-7]. Splenic infarction secondary to acute pancreatitis was believed to be uncommon, but the number of reported cases is greater than previously thought. The incidence of splenic infarction in acute pancreatitis is about 7%; The most common cause of it is splenic vein thrombosis [2] that occurring in about 1-2% of pancreatitis [8]. Another cases report study showed MSI in septic shock [2], polycythemia vera [8], splenic arterial dissection and factor V leiden mutation [9] and sarcoidosis [10]; The mechanism of MSI in these patients was unknown.
Spontaneous MSI is even rarer occurrence [3]. Reports of spontaneous MSI suggest underlying comorbidities as possible causes but do not point toward a definitive source [1]. The first case presented in this paper is probably the rare occurrence of a spontaneous MSI for which the definitive cause of infarction was unclear. We noticed to an association between MSI and hiatal hernia in this case but there weren't any proven mechanisms to explain this association; the spleen was in normal position and non-wandering. However, hypoperfusion due to hiatal herniation of gastric and splenic vascular pedicle can be a probable hypothesis for cause of MSI. We found no documented evidences in the previous studies to show this relationship. As in a 9-years-old child with sickle cell anemia (SCA), clinical findings of SCA and especially small spleen due to autosplenectomy process were observed, Hemoglobin electrophoresis for detection of SCA was not performed in this case; because, the mentioned findings were not present and SCA is very rare in our geographic region.
The septic emboli from endocarditis may be etiology of massive splenic infarction in the second our case. This cause is mentioned in other report, through fungal embolus as an etiology is rare [2].
The presentation of MSI is variable, but the majority present with sudden onset of severe pain in the left upper quadrant of abdomen. Among the reported cases in the literature, the majority was presented with LUQ pain [1]. That may be associated with nausea, vomiting, fever and chills. These symptoms are non-specific and may be confused with other causes of acute abdominal pain [3]. In first our case the patient didn't have any localized left upper quadrant pain. She was admitted with an unusual presentation of diffuse abdominal pain and tenderness and was operated with the diagnosis of peritonitis. The presentation of diffuse abdominal pain in the case of an MSI in unusual and typically occurs if infarction has led to rupture [11]. In our second case, abdominal symptoms were no significant, probably due to other clinical picture of background disease.
It mentioned that ultrasound is less sensitive for acute infarcts because there is no differential between the echogenicity of infracted and normal tissue and CT may be the most valuable imaging modality for making the diagnosis [2]. However, sonography is a first useful imaging technique for the initial assessment and subsequent follow-up of suspected splenic infarcts [12]. Focal splenic infarcts are seen as multiple triangular or wedge-shaped hypoechoic areas that were identified at the periphery of the splenic parenchyma in initial phase and echogenic as fibrosis occurs. Color Doppler imaging may show a lack of perfusion [13-17].
For massive splenic infarcts, in a retrospective Llewellyn, et al study, they noticed highly reflective bright linear bands within lesions (bright band sign) that had the classic appearance of splenic infarcts [18,19]. In our cases, the sonographic appearances of massive splenic infarction were: 1) Normal size or mild enlargement of the spleen; 2) Diffuse hypoechogenic of the splenic parenchyma with interspersed hyperechoic linear striation; 3) Subcapsular of irregular homogenous hyperechoic or hypoechoic area; 4) Absence of vascular flow is observed in color Doppler.
In overall, an interspersed hyperechoic linear striation of the splenic parenchyma and subcapsular abnormal shadow are the valuable signs of massive splenic infarct on ultrasound, and color Doppler and CT scan can be used for diagnosis confirmation.