Acute cholecystitis is a common surgical emergency, for which ultrasound (US) is the most common first-line diagnostic imaging test. Computed tomography (CT) is an alternative or complementary test for the assessment of acute cholecystitis. Acute cholecystitis is diagnosed based on clinical history and assessment, biochemical examination, and US findings. CT is considered only in case of unclear clinical symptoms or discrepant findings. We aimed to retrospectively compare the efficiency of CT and sonography for the diagnosis of acute cholecystitis in 103 patients who underwent laparoscopic cholecystectomy for acute cholecystitis during 2009-2018. The sensitivity and specificity of US and CT were comparatively analyzed on the basis of the intraoperative findings and histological outcomes. All patients underwent US and CT, of which 60 were diagnosed with acute cholecystitis based on histopathology. US and CT demonstrated 100% and 100% specificity, 77% and 77% negative predictive value (NPV), and 72% and 85% sensitivity for the diagnosis of acute cholecystitis, respectively. Thus, CT revealed a higher degree of sensitivity for acute cholecystitis diagnosis. However, US remain the first choice of test for diagnosis.
Acute cholecystitis, Diagnosis, Ultrasound, Computed Tomography, Gallbladder, Comparative Study
Acute cholecystitis is a common emergency surgical condition that is characterized by inflammation of the gallbladder secondary to an obstruction in the gallbladder neck, often resulting from cholelithiasis [1]. Gallstones are a common finding, accounting for 10-15% of all cases occurring in the general population. Of all the patients with cholelithiasis, 1-4% present with symptoms every year, while some of these (30%) develop acute cholecystitis [1-4].
The diagnosis of acute cholecystitis is confirmed on the basis of typical anamnesis, including recurrent or unrelenting right-upper quadrant pain, fever, nausea, and clinical examination findings of right-upper quadrant tenderness, positive Murphy sign, elevated laboratory findings for acute inflammation, and ultrasound (US) [5,6]. Only in the case of unclear clinical symptoms or discrepant findings, a further radiological examination is conducted by computed tomography (CT). In the current literature, the accuracy of US diagnosis in case of acute cholecystitis has been reported at 81% [7]. In the literature, however, we noted only a few studies that analyzed the sensitivity and specificity of CT in the diagnosis of acute cholecystitis. The aim of this retrospective study was to determine the accuracy of CT and US for the diagnosis of acute cholecystitis.
This was a retrospective single-center cohort study that aimed to search the radiological database of patients who underwent emergency laparoscopic cholecystectomy (LCHE) at our institution from 2009 to 2018. Suspected acute cholecystitis was diagnosed in 103 patient encounters, and both US and CT were conducted for all patients. CT was performed without contrast material, secondary to poor renal function, in 10 patients (10%), while CT was performed with a contrast material in 93 patients (90%). Approval from the regional ethic committee was obtained for this retrospective study. Patient data were de-identified and stored securely. The demographic as well as the clinical and pathological data were retrieved from an electronic database. The diagnosis of acute cholecystitis was considered to be probable in patients with at least 2-3 diagnostic criteria described in the Tokyo guidelines, as follows: Clinical parameter (fever), findings from blood chemistry (elevated white blood count), and positive US [8]. Only in the case of unclear clinical symptoms or discrepant findings, a further radiological examination was performed through CT. The findings of US and CT were compared with the histopathological outcomes. The sensitivity and specificity of US and CT for the diagnoses of acute cholecystitis were then analyzed.
US examinations were performed by 1 of the 4 US technologists of 5-25 years of experience using a US unit (Logiq 9; GE Healthcare) with 5-MHz curved array transducers. Color flow doppler was performed to identify the increased flow rate in cases of gallbladder wall thickening of > 5 mm at the technologist's discretion. CT was performed using 64-MDCT scanners using a 3-5-mm collimation. The patients who received IV contrast material were administered with 100 mL iopromide 300 (Iopamiron300; Bayer Healthcare) at the rate of 2-3 mL/s. The diagnosis of cholecystitis on an US was made if 2 major criteria or 1 major and 2 minor criteria were met. The major criteria included sonographic Murphy sign, gallbladder wall thickening > 3 mm, and pericholecystic fluid. The minor criteria included intra- or extrahepatic biliary dilatation and gallbladder hydrops (transverse diameter > 5 cm) [8]. The diagnosis of cholecystitis on CT scan was made in case of ≥ 2 of the following criteria: Gallbladder distention, wall thickening > 4 mm, mucosal hyper enhancement, or pericholecystic fat stranding or fluid [9] (Figure 1).
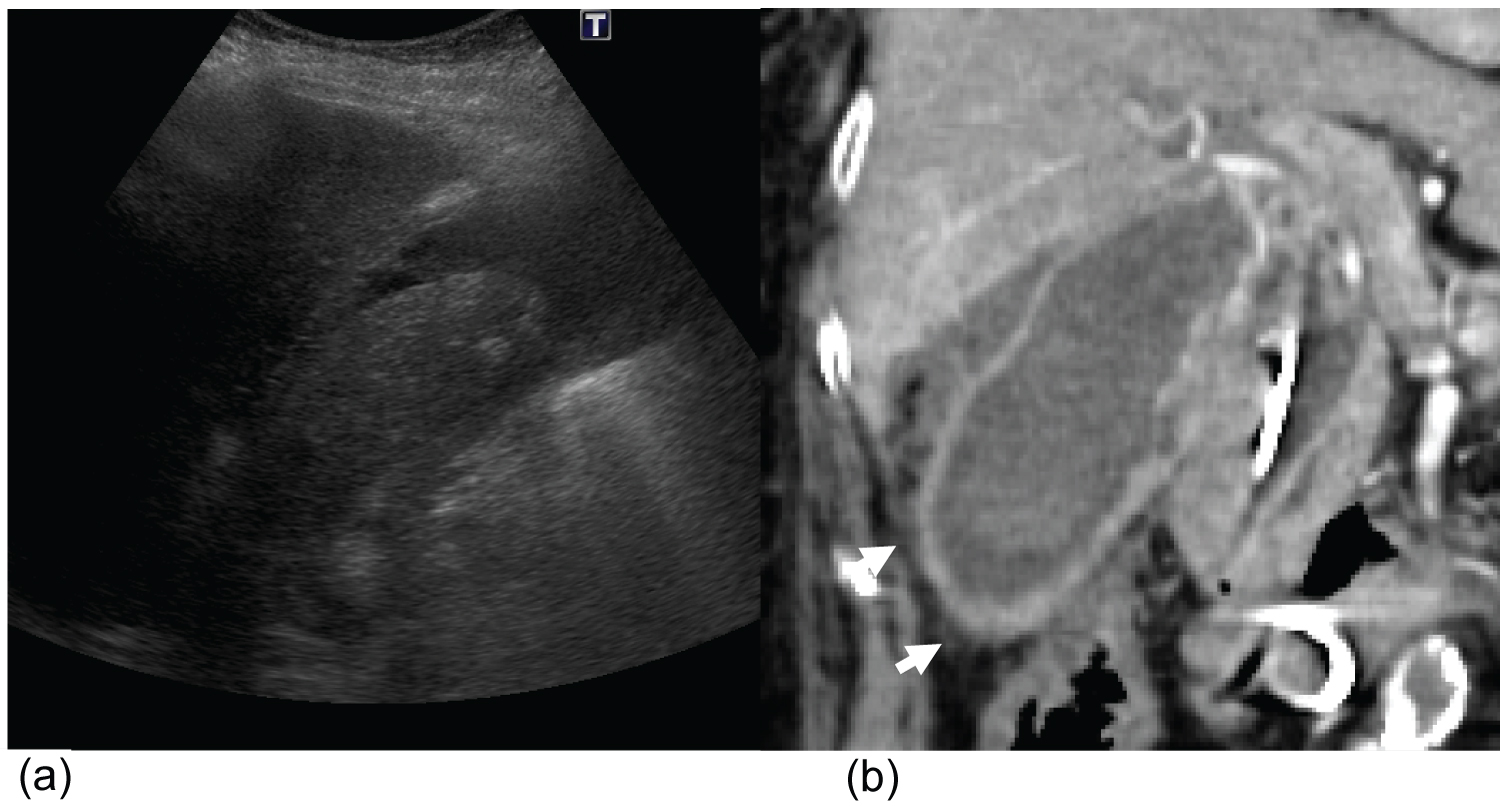 Figure 1: A 60-year-old man with acute calculous cholecystitis (histologically proven).
Figure 1: A 60-year-old man with acute calculous cholecystitis (histologically proven).
Longitudinal (a) and transverse (b) ultrasound imaging of the gallbladder. A single 3-cm mobile gallstone can be seen. No appreciable gallbladder wall thickening or probe tenderness was noted. Portal venous phase CT abdomen and pelvis (performed approximately 48-h later) demonstrated pericholecystic fat stranding (arrows) and gallbladder wall hyperenhancement.
View Figure 1
LCHE was performed in a standardized procedure commonly used in our institution. The criteria for inclusion were LCHE in case of acute cholecystitis. Patients with an open-performed cholecystectomy were not excluded from the analyses. LCHE was performed in a standardized procedure commonly used in our institution. The pathologists at our institution reviewed all surgical specimens. All histopathological specimens demonstrated inflammatory changes in sync with chronic cholecystitis. No patients presented with a "normal" gallbladder based on histology results. A total of 60 cases indicated acute cholecystitis.
We evaluated the preoperative characteristics, including demographics (sex, mean age, and laboratory values [i.e., white blood cell count, C-reactive protein, alkaline phosphatase, and bilirubin]). Histopathological diagnosis was considered as the reference standard for the diagnosis of acute cholecystitis. Positive results were recorded if the reporting pathologists had noted acute changes in the tissue specimen analysis. This report often consisted of neutrophil infiltration or the presence of abscess formation. Serum neutrophil count prior to surgical intervention was documented for each patient as well. To determine the relative accuracy of US and CT for the diagnosis of acute cholecystitis, patient images were reviewed by abdominal radiologist (with 24-year experienced) and compared with the histopathological results. For both US and CT, the results were characterized as either false-positive, true-positive, false-negative, or false-positive. Using this data, sensitivities, specificities, and odds ratios were calculated. McNemar's test was used to analyze the differences between the 2 imaging modalities. The data were analyzed using the IBM SPSS 21.0 software package (SPSS Inc., Chicago, IL, USA). Statistical significance was set at p < 0.05.
There were 103 patients included in the present study (58 men, 45 women (mean age: 63 years; age range: 34-90 years). There were 60 cases diagnosed with acute cholecystitis by histopahologically. The remaining 43 patients were not diagnosed with acute cholecystitis (including diverticulitis, gastroenteritis, common bile duct stones, duodenal ulcer, no abnormality, and other unknown causes). The US showed a sensitivity of 72% (43/60) and a negative predictive value (NPV) of 72% (43/60). CT showed a sensitivity of 85% (51/60) and an NPV of 83% (43/52). No false-positives were noted in either group, yielding specificity and positive predictive values (PPVs) of 100%. CT and US both correctly made the diagnosis of acute cholecystitis in 38 of the 60 patients (63%) who underwent both US and CT (Table 1).
Table 1: Diagnostic sensitivity, specificity, PPV, and NPV for imaging studies. View Table 1
The diagnosis of acute cystitis was made on CT and not on US in 12 patients (20%). The opposite was true for 3 patients (5%) with acute cholecystitis diagnosed on US, but missed on CT. Several statistically significant figures were obtained when each of the imaging parameter were analyzed. Even when the sensitivity of both US and CT were relatively low in the detection of acute cholecystitis, the odds ratio of CT and US in detecting acute cholecystitis was 3 (p < 0.01) and 4.6 (p < 0.01), respectively. Gallbladder wall thickening and sonographic probe tenderness were found to act as reliable predictors of acute cholecystitis as the odds ratio for gallbladder wall thickening (≥ 3 mm) was 6.27 (95% CI 2.72-14.45), while that for sonographic probe tenderness was an odds ratio of 4.53 (95% CI 1.94-10.57). CT findings of pericholecystic fluid were a good indicator for acute cholecystitis with an odds ratio of 3.86 (95% CI 1.55-9.62) (Figure 2).
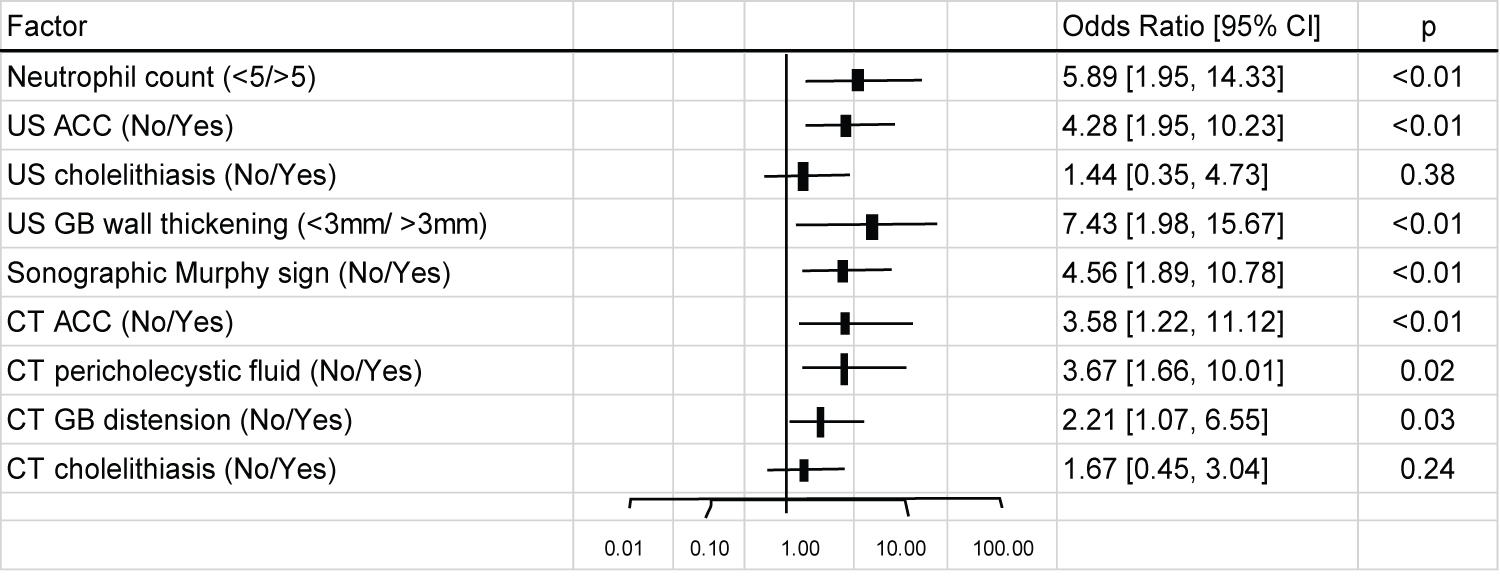 Figure 2: Odds ratio for each imaging parameter for the detection of acute cholecystitis.
View Figure 2
Figure 2: Odds ratio for each imaging parameter for the detection of acute cholecystitis.
View Figure 2
US were considered as the first-line of imaging technique for the evaluation of patients who were clinically suspected for acute cholecystitis [10]. At our institution, CT was performed when the US findings were equivocal or when the clinical findings were nonspecific. There have been only a few comparative studies between CT and US with respect to the diagnosis of acute cholecystitis [11]. Our results demonstrated that CT yielded a specificity of 100% and a sensitivity of 85% with an NPV of 77%. Sonography demonstrated a specificity of 100% and a sensitivity of 72% with an NPV of 77% in our retrospective analyses. In our series, the sensitivities for US and CT were relatively low (47% and 45%, respectively) when compared to the recent literature reporting the sensitivity for the detection of cholecystitis by US of 73-100% and by CT of 73% [7,12].
However, a past study did report a sensitivity of 54% and a specificity of 81% for acute cholecystitis on US [7,12], while another reported a higher sensitivity of 92% on CT for the detection of acute cholecystitis relative to that by US (79%) [13]. Notably, there are other reliable modalities for the detection of acute cholecystitis, such as radionuclide scanning and magnetic resonance imaging, but their utilization is limited in real-time setting [14]. In addition, our study revealed that CT possesses a high sensitivity and specificity in case of acute cholecystitis. Nevertheless, US offer the following major advantages: high sensitivity for cholelithiasis, lack of ionizing radiation or contrast injection, fast availability, and relatively low cost [15].
LCHE is a standard surgical procedure offered to patients with acute calculous cholecystitis and biliary colic. Nevertheless, surgical intervention is sometimes required for acalculous cholecystitis [15]. Acalculous cholecystitis can develop in the setting of severe systemic disease, diabetes mellites, malignant disease, vasculitis, congestive cardiac disease, or shock [16]. In our case series, 17 patients with acute acalculous cholecystitis were recorded, and the sensitivity of US and CT in detecting the disease was moderately low, but specificity was high. We acknowledge the inherent bias of our case series, such that the patients were drawn from a cohort of all patients undergoing emergent cholecystectomy, rather than from a specific cohort presenting with acute right-upper quadrant pain or those with cholecystitis that was managed through a non-operative intervention. This is a limitation of the study design. Other limitations of this study include the fact that diagnosis made by CT and US involved subjective assessment. Wall thickening was subject to observer variability. The presence or absence of pericholecystic inflammatory change was also a subjective assessment. The assessment of gallbladder probe tenderness on US was subjective, and could have been influenced by the administration of opioid analgesia prior to performing US. Moreover, there is a possibility that a proportion of the acalculous cholecystitis group had gallstones that were missed on both imaging and during surgery. For instance, small cystic duct calculi may have possibly gone undetected.
As expected in our case series, US is a superior tool to CT for the detection of cholelithiasis, which is consistent with the relevant literature reports [17]. CT detection of cholelithiasis relies heavily on the presence of calcification and, occasionally, on fat or gas attenuation with several gallstones being occult on CT. The specificity of CT in detecting cholelithiasis in this study was 55%, which is comparable to the literature report [13].
Our study suggests that certain parameters are highly associated with acute cholecystitis, such as gallbladder wall thickening (≥ 3 mm) and sonographic probe tenderness, but careful consideration should be given to such findings considering the possible confounding factors. Sonographic probe tenderness is a subjective finding that can be masked by the presence of analgesia prior to scanning. Gallbladder wall thickening can herald other medical conditions, such as cardiac failure, liver failure, hypoalbuminemia, or hepatitis [18].
We noted a significant false-negative rate for both US and CT, and a potential reason for this finding is that the pathological appearance on imaging may have been influenced by the time duration since the onset of symptoms and the time of scanning.
There was another limitation in our study that we analyzed only those patients who were treated with LCHE. Patients treated with antibiotics were excluded.
We thus conclude that CT is comparable to US for the diagnosis of acute cholecystitis. We also recommend US as the first choice for acute right-upper abdominal pain with a suspected history of gall stone disease. If these findings are unclear, CT can provide a useful supplement radiological examination with a high overall accuracy for the diagnosis of acute cholecystitis.
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used "Conceptualization, M.T. and T.O.; methodology, M.T..; software, M.T..; validation, M.T.; T.O.; investigation, M.T.; resources, M.T.; data curation, I.N.; Y.Y.; writing-original draft preparation, T.O.; writing-review and editing, M.T.; supervision, K.I.; funding acquisition, M.T. All authors have read and agreed to the published version of the manuscript." Please turn to the CRediT taxonomy for the term explanation. Authorship must be limited to those who have contributed substantially to the work reported.
This research received no external funding.
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by our local institutional review board (Ethical Committee of Kindai University No. 23-101).
Informed consent for data collection was obtained from all subjects involved in the study.
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy reasons.
The authors declare no conflict of interest.