Necrotising enterocolitis is an intestinal inflammatory disease, with the highest rate of incidence occurring in premature infants born before 36-weeks, due to infants missing out on the in-utero ingestion of vernix caseosa. The in-utero consumption of vernix caseosa is a key biological process, necessary for the effective and healthy modulation of the infant's intestinal microbiome, as the branched chain fatty acid content of vernix caseosa allows many branched chain fatty acid commensal bacteria to colonise the infant's gastrointestinal tract. The in-utero ingestion of vernix caseosa during the last month of pregnancy creates a specific niche within the infant's intestines for branched chain fatty acid containing bacteria, such as Bifidobacterium sp., Bacteroides sp. and Propionibacterium sp. The importance of Bifidobacterium sp., Bacteroides sp. and Propionibacterium sp. to colonise the infant's intestines, is due to the capacity of these branched chain fatty acid commensal bacteria to produce large amounts of short-chain fatty acids (SCFAs), that modulate and lower levels of intestinal inflammation. Without a full-term pregnancy, the infant's intestines are depleted of branched chain fatty acids and in-turn branched chain fatty acid containing bacteria, providing the opportunity for abnormal microbes to colonise the infant's intestines. The microbial imbalance seen in the intestines of premature infants can propagate further health issues encountered by a premature gestation, such as intestinal ischemia or other intestinal epithelial damage. In order to combat the pathogenesis of necrotising enterocolitis, the promotion of a healthy intestinal microbiome in premature infants via the enumeration of the intestines with branched chain fatty acids must occur. Based upon the infant's requirement of large amounts of branched chain fatty acids to effectively modulate the intestinal microbiome, a Bacillus subtilis isolate has been selected due to a high branched chain fatty acid content. The ability to enumerate the premature infant's intestines with branched chain fatty acids, will allow commensal branched chain fatty acid containing bacteria to colonise, effectively decreasing the number of abnormal bacteria present in the intestines as well as assisting in modulating the intestinal inflammatory response.
Necrotising enterocolitis, Inflammation, Vernix caseosa, Premature, Branched chain fatty acid, Probiotic, Bacillus subtilis
2Dc: Hydrophobic Thickness; ApL: Average Lateral Area Occupied by Lipids; BCAA: Branched Chain Amino Acids; BCFA: Branched Chain Fatty Acid; CFU: Colony Forming Unit; EFSA: European Food Safety Association; FDA: Food and Drug Administration; GRAS: Generally Recognized as Safe; NEC: Necrotising Enterocolitis; SCD: Order Parameter; QPS: Qualified Presumption of Safety
Necrotising enterocolitis (NEC) is an intestinal inflammatory disease described as a degradation and, in some cases, the total mortality of a portion of the bowel [1]. Although cases of NEC have been recorded in adults, the greatest risk is posed to premature infants with a gestation period lower than 36-weeks and can result in death in 20-30% of cases [2-4]. The progression of NEC begins when the infant's intestines are colonised by abnormal bacteria, leading to bacterial invasion of the intestinal epithelium and the host producing a local inflammatory response [5]. An infant's intestines express much greater levels of pro-inflammatory cytokines than adult intestines, subjecting infants to a far greater risk of bacterially induced inflammation [6]. Factors which increase a premature infant's susceptibility to bacterial proliferation through the intestinal epithelia are the underdevelopment of a premature infant's lungs and intestines [7]. The underdeveloped lungs and intestines in premature infants stipulates a bacterial invasion as due to inadequate oxygen flow, intestinal walls are damaged allowing bacteria passage into the epithelial layer, resulting in inflammation and eventual ruptures in the bowel walls [7]. Although the progression of NEC requires bacterial proliferation through the intestinal lumen, the lack of microbial diversity present in the intestines of premature infants allows for this invasion to occur by opportunistic or pathogenetic microbes [4]. In premature infant's intestines, approximately 64 genera of bacteria have been identified, whilst over 200 genera have been identified in full-term infants [4,8,9]. The high degree of disparity observed between microbial colonisation in premature and full-term infants has been directly linked to the infant's premature gestation period (< 36-weeks'), and more specifically that the premature infant does not ingest vernix caseosa in-utero [4].
Vernix caseosa is a naturally occurring biofilm on the foetus' skin in-utero, which starts being produced at the gestational age of 24-weeks' [10]. Once believed to be a foetal epidermal protective barrier, the implications of vernix caseosa's lipid content upon infant gastrointestinal health has defined vernix caseosa ingestion in-utero as an essential process in infants developing a healthy intestinal microbiome [4,11]. The in-utero consumption of vernix caseosa by the foetus occurs during the last month of pregnancy ensures that a full-term infant's intestines are enumerated with branched-chain fatty acids (BCFAs) [12,13]. The inadequate supply of BCFAs to premature infants is shown as the BCFA content of vernix caseosa differed significantly between premature and full-term infants, at 25.29% and 43.02% of total fatty acids, respectively [11]. The foetus' ingestion of BCFA in vernix caseosa directly affects the microbial composition and diversity in the infant's intestines by allowing commensal bacteria that utilise BCFAs to colonise the intestines, such as Bacteroidetes, Propionibacterium and Bifidobacterium [4,14]. The importance of BCFAs and Bifidobacterium for an infant's intestinal health is also demonstrated through the relative concentrations of BCFAs and viable load of Bifidobacterium (102-104 CFU/ml) present in breast milk [15,16]. The constant supply of BCFAs during the last month of pregnancy via vernix caseosa and on-going after birth via breast milk, suggests that an infant's BCFA requirements may extend beyond birth and until a consistent proteinaceous diet is commenced [4,16-18]. The continuous source of BCFAs is eventually replaced through naturally acquired branched-chain amino acids (BCAAs) present in the child's diet, found in meats, dairy products, and legumes [19-21]. Once a child's solid food regime has begun, the enumeration of the intestines will shift from BCFAs to the BCAAS, valine, leucine, and isoleucine. BCAAs are the essential building blocks of BCFAs, with valine and leucine being used to produce iso-BCFAs and isoleucine being required to synthesise anteiso-BCFAs [22]. Many BCFA containing bacteria also produce a combination of end products from amino acids and carbohydrates which are beneficial to gastrointestinal health, including the short-chain fatty acids (SCFAs); acetate, propionate, and butyrate [23-25].
BCFAs are present in the membranes of many prokaryotes and are categorised by either a single (monomethyl) or multiple methyl-branches (dimethyl or multi-methyl), located on the penultimate 2nd last carbon (iso-BCFA), antepenultimate 3rd last carbon (anteiso-BCFA) or a mid-chain carbon [26-28]. Like unsaturated fatty acids (UFA) in higher organisms, BCFAs in bacteria affect the cell membrane's fluidity, with increasing concentrations forcing the membrane into a disordered state (Figure 1.1) [29,30]. This disordered state allows for better transport through the membrane, membrane protein structure and functionality, and cellular signal transduction and trafficking (Figure 1.1) [31]. The contrast of this disordered state is seen through the increased concentration of straight-chain fatty acids (SCFA) (C16:0) to greater than 20% (< 20%), which results in an ordered state in the membrane (Figure 1.2) [30]. It is during this transition, that the membrane increases in hydrophobic thickness (2Dc) and decreases in average lateral area occupied by lipids (ApL) (Figure 1.2) [30,32,33]. The membrane enters the ordered state as fatty acyl chains pack tightly together, the membrane thickens and loses fluidity, greatly impeding regular cellular processes, such as active and passive transport, membrane protein structure and signal transduction (Figure 1.2) [30].
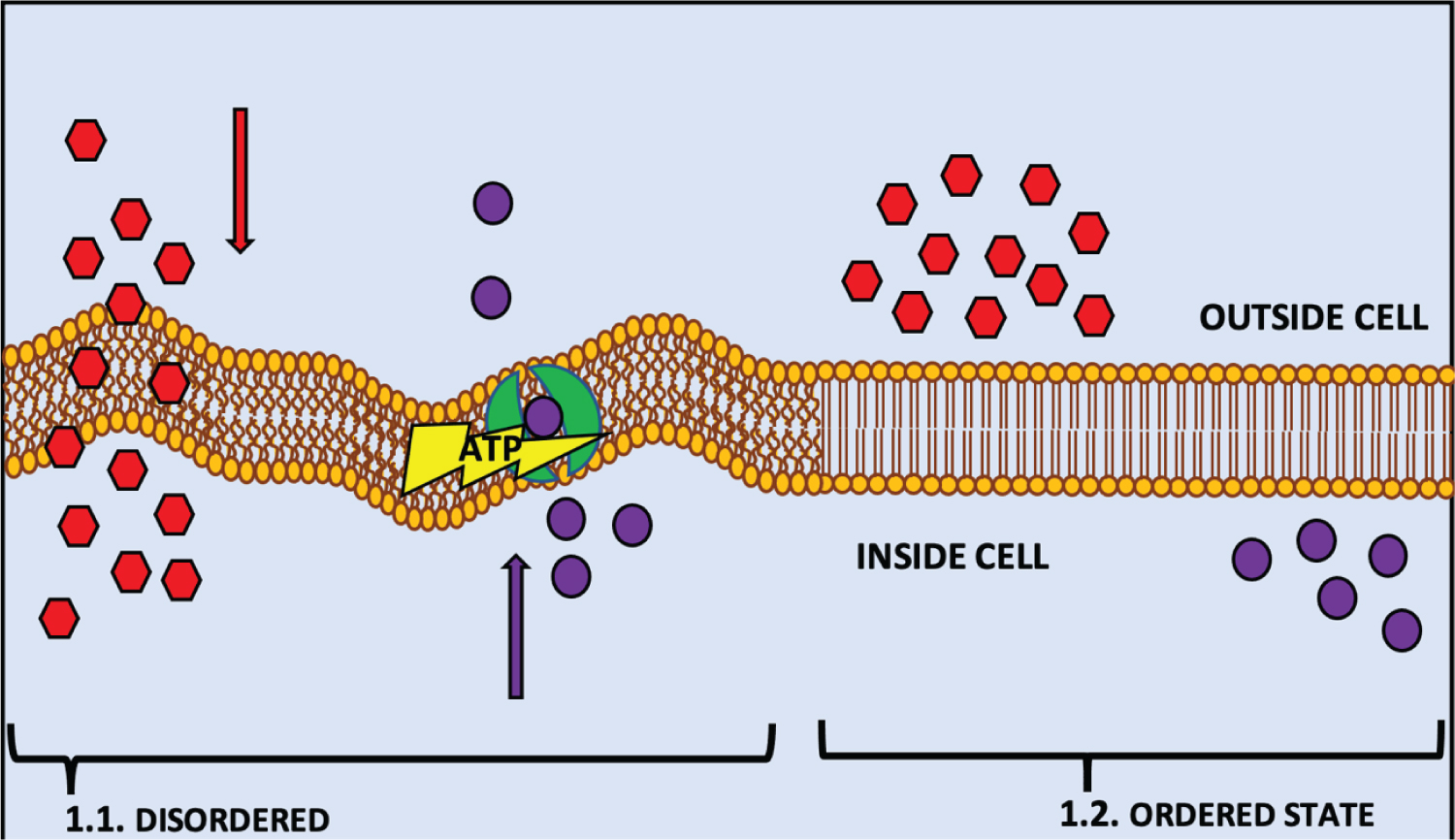 Figure 1: Is a representation of a phospholipid bilayer cell membrane in a disordered state; (2a) and an ordered state (2b) as a result of their fatty acid concentration. (1.1.) demonstrates the fluidity of the membrane in a disordered state that is induced by the increased concentration of BCFAs in many bacteria (Unsaturated fatty acids (UFA) in higher organisms), which positively the rigidity of the membrane in an ordered state that is caused by an increase of SCFAs (n16:0) to < 20% in the membrane, which inhibit or decrease the action of multiple metabolic routes in and out of the cell.
View Figure 1
Figure 1: Is a representation of a phospholipid bilayer cell membrane in a disordered state; (2a) and an ordered state (2b) as a result of their fatty acid concentration. (1.1.) demonstrates the fluidity of the membrane in a disordered state that is induced by the increased concentration of BCFAs in many bacteria (Unsaturated fatty acids (UFA) in higher organisms), which positively the rigidity of the membrane in an ordered state that is caused by an increase of SCFAs (n16:0) to < 20% in the membrane, which inhibit or decrease the action of multiple metabolic routes in and out of the cell.
View Figure 1
A study by [34] investigated the effectiveness of the number of methyl branches (mono-, di- or poly-methyl) and the chain position of the methyl branch/es on a fatty acid (C16:0) to maintain the thickness and fluidity of the cellular membrane [34]. The study revealed no trend in bilayer thickness in relation to methyl branches at the anteiso-, iso- or mid chain position, with all membranes containing BCFAs maintaining a bilayer < 4 nm, with 3-4 nm being the average bilayer thickness of functional lipid bilayer membranes [34,35]. However, [34] found that multiple methyl branched BCFAs can have a combinational effect of all branches on a membrane's fluidity, as their corresponding monomethyl BCFAs [34]. The order of membranes was defined by the order parameter SCD, which measures the mobility of carbon-deuterium bonds along the fatty acid acyl-chains [34,36,37]. Where a perfectly ordered state of the membrane is indicated by a SCD of 1, and a membrane in a disordered state is represented by a SCD between ~0.2-0 [34]. A decreasing SCD was correlated with the carbon position of methyl-branches for mid-chain, anteiso- and iso-BCFAs [34]. Membranes containing BCFAs with a mid-chain branch experienced a decline in SCD to below 0.2 at the carbon position of the branch, with the SCD continuing to decrease after the branch until the terminal-end of the chain (carbon 16). A SCD of below 0.2 was achieved by membranes containing anteiso- and iso- methyl-branches at the middle of the chain (carbon 8), with the SCD decreasing to below 0.1 at the carbon position of the methyl-branches; anteiso-14 and iso-15 [34]. BCFAs with multiple-methyl branches displayed the same sharp decline in SCD at the location of the branch, however the distance between the branches on the chain was seen to affect the SCD [34]. Multiple-methyl-BCFAs with branches at 4 carbon positions apart experienced a decline in SCD at the location of branches, whilst SCD continued to steadily decrease until another branch or the terminal-end of the chain [34]. For multiple-methyl-BCFAs with branches at 2 carbon positions apart showed the same decline at the location of branches, however, saw an increase in SCD from branch-to-branch position, with SCD only decreasing terminally at the carbon position after all the branches [34]. This indicates that the methyl-branches of BCFAs positively affect a membrane's order and fluidity, yet for multiple-methyl- branches located within 2 carbon positions increased the membrane's order throughout the area of the chain where the branches were located [34].
The impact of BCFAs on the colonisation of the infant's intestines by bacteria is shown by the comparison of which bacteria dominate a premature infant's intestines, opposed to a full-term infant. Bacteria that do not utilise BCFAs, such as Proteobacteria and non-BCFA Firmicutes are consistently identified as the main coloniser of a premature infant's intestines (< 36-weeks'), whereas BCFA containing Bacteroidetes, Actinobacteria and BCFA Firmicutes contribute considerably to the intestinal microbiome of a full-term (40-weeks') infant (Table 1) [38-45]. The colonisation of the infant's intestines is seen to be greatly affected by a gestation period of over 36-weeks and BCFA consumption, as approximately 65% of the families which colonise a healthy full-term infant's intestine contain and utilise BCFAs (Table 1) [38-45]. The ability of these BCFA-bacteria to colonise a newborn infant's intestines is due to the enumeration of the infant's intestines with BCFAs via vernix caseosa during the last month of pregnancy (after 36 weeks') [4,14,17,44]. In accordance with BCFA supply to infant's intestines occurring during the last month of gestation, premature infants born before 36 weeks' do not have sufficient BCFAs present throughout intestines, and as a result these BCFA-bacteria found in full-term infants are unable to colonise premature infants at the same rate [4,11,17,44,46].
Table 1: This table contains, by Phyla, Order & Family, bacteria that are often present in a healthy human full-term infant (< 36 weeks') gastrointestinal microbiome [38-42]. In red are the families of bacteria, which contain branched-chain fatty acids (BCFAs) [43-45]. View Table 1
The inability for BCFA-bacteria to colonise the premature (< 36 weeks') infant intestines can lead to an imbalance and a decrease in species richness in the infant's intestinal microbiome, where it is observed that only one or two phyla of bacteria (Proteobacteria and Firmicutes) dominate the intestines (Figure 2.1 and Figure 2.2) [47-52]. Inversely, in full-term (40 weeks') infants, as a result of in-utero vernix caseosa ingestion, the intestinal environment has received a constant supply of BCFAs during the final month of gestation [4,14,17,44]. Subsequently from the ingestion of BCFAs by full-term infants, the full-term intestinal microbiome contains significantly more BCFA-bacteria, specifically Bacteroidetes, and decreased levels of Proteobacteria and Firmicutes, than seen in both premature infant groups (Figure 2.3) [47-52]. The increased numbers of Bacteroidetes and decline in Proteobacteria and Firmicutes from premature to full-term infants, establishes a balance within the intestinal microbiome of full-term infants, where now three phyla of bacteria are relatively prevalent (Figure 2.1, Figure 2.2, Figure 2.3) [47-52]. The phylum Actinobacteria, which encompasses Bifidobacterium sp., was seen to increase in the intestines of infants without NEC when compared with infants with NEC, with highest levels being observed in healthy premature infants, although still being recorded in relative amounts in full-term infants (Figure 2.1, Figure 2.2 and Figure 2.3) [47-52]. Reasons that suggests this result, are that many of the healthy premature infants were breast-fed, and breastmilk contains BCFAs and a viable load of Bifidobacterium sp. [4,16-18,47-52]. Also, the lack of Bacteroidetes in healthy premature infants may have provided the opportunity for further growth by Actinobacteria [47-52]. Despite the higher rate of colonisation by Actinobacteria in healthy premature infants, the levels of Bifidobacterium sp. present in the intestines are consistently greater in full-term infants, with breastfed full-term infants recording the highest rates of Bifidobacterium sp. colonisation (Figure 2) [47-52]. Although NEC cases in premature infants have not been directly linked to specific pathogens, the increased numbers of Clostridium sp. and Klebsiella sp. are consistently recorded in infants with NEC [53-55]. Whilst having increased numbers of pathogens, premature infant intestines are also observed with low levels of BCFA commensal bacteria, such as Bacteroidetes sp., Bifidobacterium sp. and Propionibacterium sp., compared to full-term infants (Table 1 and Figure 2) [38-42,47-52].
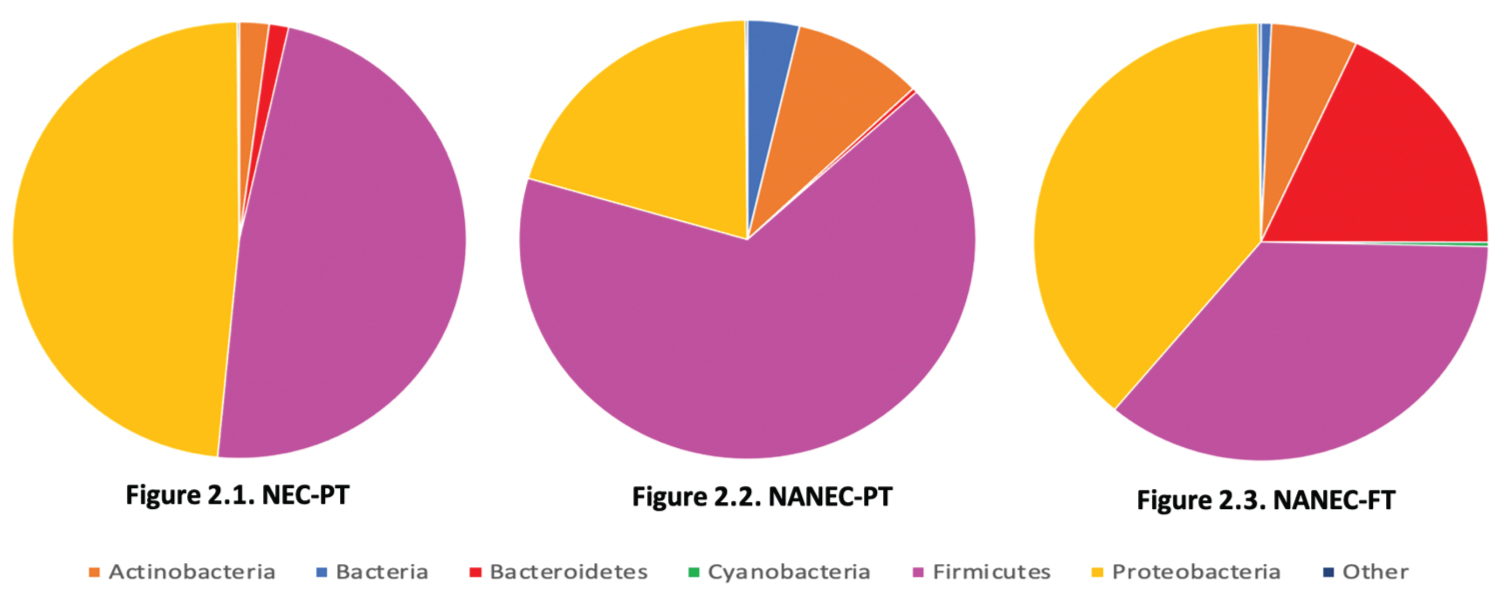 Figure 2: Pie graphs developed from available data (as per Figure Methods) illustrating the composition of the intestinal microbiomes of premature infants born before 36 weeks with necrotizing enterocolitis (Figure 2.1. NEC-PT), premature infants born before 36 weeks that are healthy (Figure 2.2. NANEC-PT) and full-term infants born after 40 weeks that are healthy (Figure 2.3.NANEC-FT).
View Figure 2
Figure 2: Pie graphs developed from available data (as per Figure Methods) illustrating the composition of the intestinal microbiomes of premature infants born before 36 weeks with necrotizing enterocolitis (Figure 2.1. NEC-PT), premature infants born before 36 weeks that are healthy (Figure 2.2. NANEC-PT) and full-term infants born after 40 weeks that are healthy (Figure 2.3.NANEC-FT).
View Figure 2
Many commensal bacteria within the human gastrointestinal tract utilise BCFAs to survive the intestines varying environmental conditions, such as Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp., and therefore are recorded significantly more in the intestines of full-term infants than pre-term infants [47-52]. The link between the increased colonisation of Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. and full-term gestation (40 weeks') can be seen from the comparison of the BCFA content of vernix caseosa and the BCFA requirements of Bacteroides fragilis, Bifidobacterium bifidum, Bifidobacterium breve, Propionibacterium freudenreichii, Propionibacterium shermanii and Propionibacterium jensenii (Figure 3 and Figure 4) [11,17,56-58]. Bacteroides fragilis utilises three BCFAs; anteiso-C15:0 (32.2%), iso-C15:0 (17.3%) and anteiso-C17:0 (1.7%); Bifidobacterium bifidum utilises five BCFAs; iso-C16:1 (5.3%), iso-C18:0 (2.6%), iso-C14:0 (1.1%) and iso-C16:0 (0.5%); Bifidobacterium breve utilises three BCFAs; iso-C14:0 (24.6%), iso-C16:0 (0.5%) and iso-C18:0 (0.5%) Propionibacterium freudenreichii utilises six BCFAs; anteiso-C15:0 (68.1%), anteiso-C17:0 (11.5%), iso-C15:0 (4.5%), iso-C17:0 (1.5%), anteiso-C13:0 (1%) and iso-C14:0 (1%); Propionibacterium shermanii utilises four BCFAs; anteiso-C15:0 (42.6%), anteiso-C17:0 (9%), iso-C15:0 (4.5%) and iso-C17:0 (1%); and Propionibacterium jensenii utilises four BCFAs; iso-C15:0 (31.6%), anteiso-C15:0 (16.8%), anteiso-C17:0 (7.8%) and iso-C17:0 (0.6%) (Figure 4) [56-58].
 Figures 3: Branched-chain fatty acids of vernix caseosa from studies; 1. Ran-Ressler, et al. (2008) (blue), 2. Li, et al. (2021)(orange). BCFAs in Ran-Ressler, et al. (2008) were in mono & dimethyl-form, with anteiso-, iso& mid-chain configurations. BCFAs in Li, et al. (2021) were in monomethyl form, with anteiso and iso-configuration. (A = anteiso, I = Iso, diMe = dimethyl, me = monomethyl, C = carbon [11,17].
View Figure 3
Figures 3: Branched-chain fatty acids of vernix caseosa from studies; 1. Ran-Ressler, et al. (2008) (blue), 2. Li, et al. (2021)(orange). BCFAs in Ran-Ressler, et al. (2008) were in mono & dimethyl-form, with anteiso-, iso& mid-chain configurations. BCFAs in Li, et al. (2021) were in monomethyl form, with anteiso and iso-configuration. (A = anteiso, I = Iso, diMe = dimethyl, me = monomethyl, C = carbon [11,17].
View Figure 3
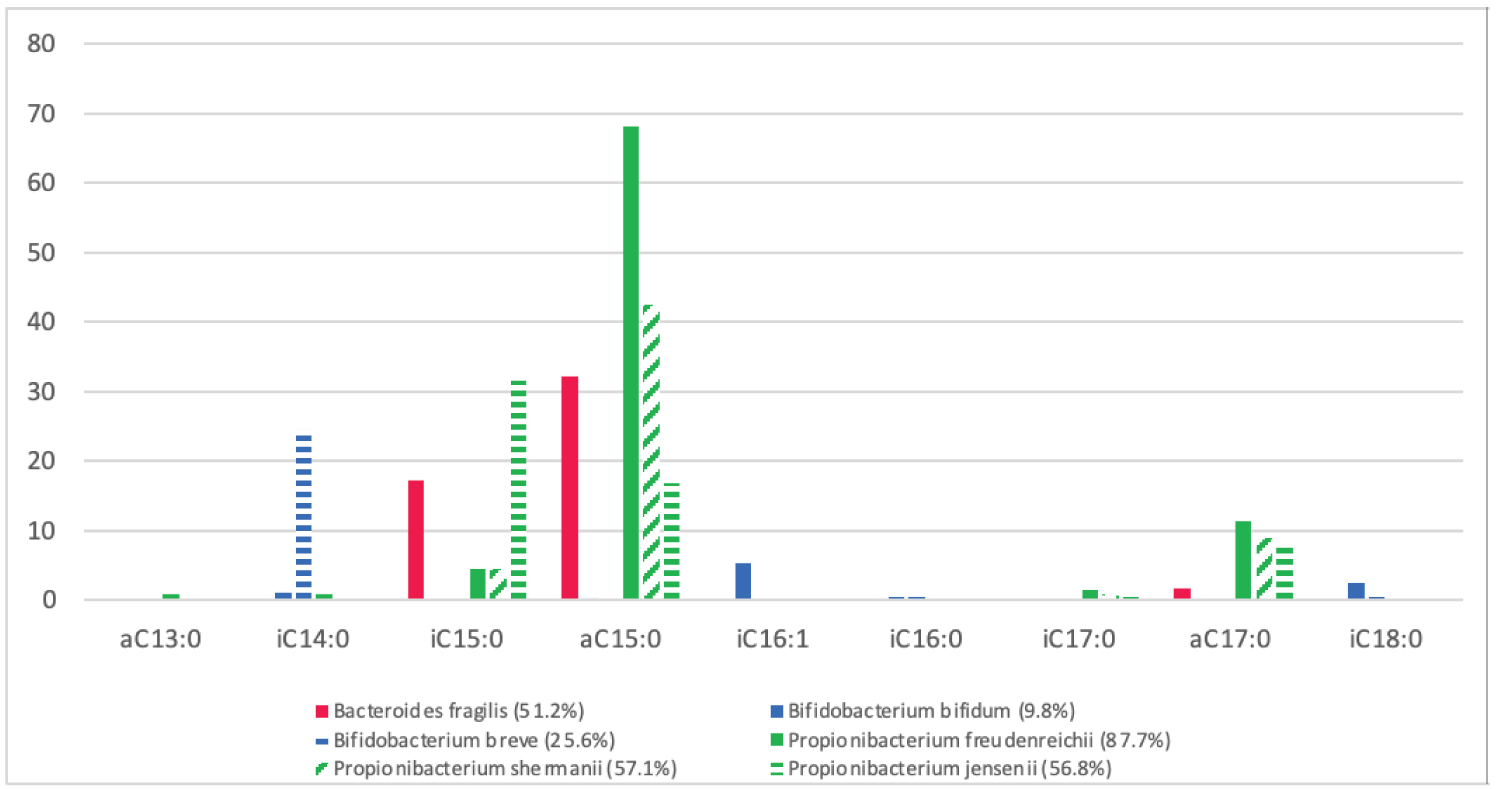 Figure 4: The BCFA membrane content of commensal BCFA-bacteria; Bifidobacterium bifidum and Bifidobacterium berve, Bacteroides fragilis, Propionibacterium freudenreichii, Propionibacterium shermanii and Propionibacterium jensenii, in relation to total FA. ( I = iso-branch, a = anteiso-branch, C = carbon chain) [56-58].
View Figure 4
Figure 4: The BCFA membrane content of commensal BCFA-bacteria; Bifidobacterium bifidum and Bifidobacterium berve, Bacteroides fragilis, Propionibacterium freudenreichii, Propionibacterium shermanii and Propionibacterium jensenii, in relation to total FA. ( I = iso-branch, a = anteiso-branch, C = carbon chain) [56-58].
View Figure 4
From Figure 3 and Figure 4 it is seen that the majority of BCFAs contained by Bacteroides fragilis, Bifidobacterium bifidum, Bifidobacterium breve, Propionibacterium freudenreichii, Propionibacterium shermanii and Propionibacterium jensenii are present in relative amounts in vernix caseosa (Figure 3 and Figure 4) [11,17,56-58]. With exception of iso-C17:0 which was not isolated from vernix caseosa and was found in the screened Propionibacterium sp. (Figure 3 and Figure 4) [11,17,56-58]. Due to iso-C17:0 only attributing to approximately 1.15% of the total BCFA of the Propionibacterium sp. could suggest that Propionibacterium sp. would be able to maintain membrane fluidity in the intestines of a newborn infant, despite iso-C17:0 being absent in vernix caseosa [11,17,57]. The ability of Propionibacterium sp. to colonise a full-term infant's intestine despite vernix caseosa not containing iso-C17:0 is demonstrated as Propionibacterium sp. are consistently recorded as one of the first colonisers of the intestines of full-term infants [59-61]. The abiliedty of Bacteroides sp. and Bifidobacterium sp. to colonise the full-term infant's intestines after birth is also seen as together, Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. represent the majority of first intestinal colonisers in full-term infants [62,63]. Propionibacterium sp. colonisation in early infancy occurs from mother-infant transfer, as Propionibacterium sp. are one of the most prevalent bacteria isolated from amniotic fluid and are also present in human breastmilk [61,64]. The transmission of Bacteroides fragilis and Bifidobacterium sp. from mother-infant has also been established by past studies and believed to be at a result of vertical transmission [65-68]. Little is known about the mechanics of Bacteroides fragilis transfer from mother-infant, although has been confirmed in multiple studies through strain-level metagenomic profiling of mother and infant faecal samples [65,66,68]. The colonisation of Bifidobacterium sp. in new born infants occurs through various mother-infant channels, as Bifidobacterium sp. are naturally present in the breast milk and vaginal canal of mothers [67]. Subsequently, due to the prevalence of Bifidobacterium sp. in breastmilk and the vaginal cavity, infants which are breastfed and delivered vaginally are observed with increased numbers of Bifidobacterium sp. [69].
Through the comparison of the BCFA content of vernix caseosa and Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. it is seen how in-utero ingestion of vernix caseosa would positively influence Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. colonisation in the intestines of full-term infants (Figure 3 and Figure 4) [11,17,56-58]. Vernix caseosa is seen to play an essential role in supplying BCFAs to the infant's intestines, which in turn allows the successful mother-infant transfer of Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. (Figure 3 and Figure 4) [11,17,56-58]. The processes of in-utero vernix caseosa ingestion and vertical transmission from mother-infant of commensal bacteria appear to work in partisan to allow the infant to develop a healthy intestinal microbiome [4,14,62,63,65,66,68,69]. The potential for in-utero ingestion of vernix caseosa to lower the mortality rates of NEC can be inferred due to the positive effect of vernix caseosa BCFAs on Bifidobacterium sp. intestinal colonisation [4,11,17]. A study by [4] demonstrated the importance of Bifidobacterium sp. to the development of a healthy intestinal microbiome, as increased rates of Bifidobacterium sp. in the intestines of rat-pups was found to be correlated with lower incidences of NEC in supplemented rat-pups [4]. The requirement of a healthy balanced intestinal microbiome is increasingly important for infants, as with immature intestines and immune system, infants are far more susceptible to pathogen-mediated inflammation and disease [70-72].
For the treatment of inflammatory intestinal diseases, such as NEC, during the early stages of disease development, the administration of anti-inflammatory drugs and antibiotic is often the first course of action [73]. If the affected area of the intestines is considerable and the intestines are perforated, medical treatment alone will not suffice and surgery to remove the disease affected section of the intestines may be necessary [74]. Although the prescription of premature infants with anti-inflammatory drugs will provide temporary mediation of inflammatory responses in the intestines, the enumeration of the intestines with antibiotics may only further the dysbiosis in the infant's intestinal microbiome [75]. However, novel new therapies have arisen for the treatment of intestinal diseases, that utilise probiotic bacteria to stabilise the intestinal microbiome and restore a healthy intestinal environment [76].
For a bacterium to be considered as a probiotic it must benefit the host by improving the individual's intestinal health through modulating a stable intestinal microbiome [77]. Probiotics can work through the supplementation of viable and non-viable bacterium, and in some cases only components of the microbial cells, known as prebiotics [78]. Probiotics are present naturally in many food-products and the environment as commensal non-pathogenic bacteria, however, are also acquired and produced commercially in formulas and capsules for human consumption [79]. When developing an intestinal probiotic various protocols and requirements are present to ensure the safety and effectiveness of the final product [80]. A probiotic bacterium must be able to survive the biochemical barriers present throughout the gastrointestinal tract such as pH and bile acids, to ensure that a substantial amount of the probiotic reaches the intestines [81]. In the case of viable bacteria, once in the intestines, the bacteria must be capable of adhering to the intestinal epithelia and colonising the intestines [82]. The adhesion of the bacteria to the intestinal lining allows for the probiotic to increase in concentration within the intestinal environment and positively affect the host's intestinal microbiome [83]. In many cases probiotics are used to address and lower the incidence of intestinal diseases by effectively balancing the host's intestinal microbiota [84].
With the inflammatory intestinal disease, NEC, branched-chain fatty acids (BCFAs) are the prebiotic that can effectively modulate a healthy intestinal microbiome within premature infants, by increasing the colonisation rates of commensal BCFA bacteria [4,17,85]. Through the on-going supplement for a month after birth with BCFAs, a premature infant will be provided with a substitute of BCFAs, in lieu of in-utero vernix caseosa consumption [4,11-13,17]. The use of post-natal BCFAs as a prebiotic for premature infants will allow the host's intestines to be enumerated with BCFAs and positively influence the colonisation of BCFA-containing bacteria in the intestines [4,8,9,11-13,17,56-58]. With an increased concentration of BCFAs within the infant intestinal environment, an increase in BCFA bacteria, and in particular BCFA bacteria that produce SCFAs would be expected, such as Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. [4,8,9,11-13,17,56-58]. From the increase of BCFA bacteria within the premature infant intestinal microbiome, progression to a more balanced intestinal microbiome may be seen in premature infants, and the premature infant intestinal microbiome will potentially become more reminiscent of a full-term infant's intestinal microbiome (Figure 2) [4,8,9,11-13,17,47-52,56-58]. Through the analysis of the BCFA components of a full-term infant's vernix caseosa and the BCFA requirements of integral infant intestinal BCFA bacteria, such as Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp., the positive effects of BCFA consumption from vernix caseosa upon the intestinal colonisation of Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. can be perceived (Figure 2 and Figure 4) [4,8,9,11-13,17,47-52,56-58]. In accordance with Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. all been known to play a role in intestinal homeostasis and modulation of intestinal inflammation through the production of SCFAs [23-25,86-88]. It can be theorised that the supplement of the appropriate BCFAs that ensures the increased intestinal colonisation of Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. in preterm infants, may in turn develop a healthier intestinal microbiome in infants capable of modulating intestinal inflammation [11,17,23-25,56-58,86-88]. The importance of intestinal bacteria such as, Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. in the infant intestinal microbiome is paramount, due to the increased pro-inflammatory factors present throughout a newborn infant's intestines [72,89,90]. Due to a lack of BCFAs, premature infants with and without NEC are consistently recorded to contain low levels to none of Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. within their intestinal microbiome, which is a contributing factor to the low levels of SCFAs seen in premature infant's intestines when compared with full-term infants [47-52]. A study by [89] has recently implicated the role SCFAs may have in lowering the incidence of NEC and potentially treating NEC in premature infants, by examining the anti-inflammatory effects of SCFAs within the intestinal environment of humans [89]. This study validates the idea for the potential to treat NEC via increasing the levels of SCFA producing bacteria within the infant's intestines, however, does not mention the role BCFAs would play in allowing BCFA bacteria which produce SCFAs, such as Bacteroides sp., Bifidobacterium sp., and Propionibacterium sp. to colonise the infant's intestines [25,56-58,89,91,92].
When selecting a species of bacteria that would be an effective probiotic against the microbial imbalance which precedes NEC, a commensal bacterium with a high BCFA membrane content must be chosen [4,44,93,94]. The necessity for a benign commensal bacterial strain is paramount when dealing with immunocompromised patients, such as premature infants and premature infants with NEC [95]. A key requirement of any ingestible product commercially available is a quality control and risk assessment set forth by governing bodies. The measures put in place for the assessment of probiotics are, by the Food and Drug Administration (FDA); the Generally Recognised as Safe (GRAS) model, and by the European Food Safety Authority (EFSA); the Qualified Presumptions of Safety (QPS) model [96,97]. Both these organisations aim to review the safety and potential hazards of a food additive and establish a new product as either safe for consumers or not [96,97]. In regard to live bacteria in probiotics, the investigation into the risks must delve past the expression of toxins and look into the presence of potentially dangerous genetic material, such as genes conferring to antibiotic resistance [96-98]. The first issue of a potential probiotic bacterium containing antibiotic resistant genes is the fact that if the bacteria opportunely caused an infection in an individual, the mode of antibiotic treatment will be limited [99,100]. However, the problem of more concern which arises when introducing a bacterium with antibiotic resistance genes into an intestinal environment, is the chance for horizontal gene transfer to result in a pathogen or opportunistic pathogen acquiring genes for antibiotic resistance [101,102].
Once a probiotic has passed the quality and risk assessments, it must also be proven to be commercially reproduceable. The factors affecting a products commercial potential are the costs to produce the probiotic on a large-scale, proven viability of probiotic after manufacturing and the demonstration of an appropriate shelf-life [103,104].
13 probiotic bacteria, by genus, species, and strain have been selected to be screened as a potential probiotic for the treatment of NEC (Table 2, Table 3, Table 4, Table 5 and Table 6). From the fore mentioned requirements, the bacterium's probiotic potential is examined in regard to their efficacy to work within the gastrointestinal environment; whether they are capable of exerting health benefits against NEC; if the bacterium has been assessed as safe for human consumption; and whether reproducing this probiotic on a large scale is commercially viable.
Table 2: Lists 13 potential probiotic bacteria, at the genus, species, and strain level (when applicable). Table 2 also covers key factors in the determination of a bacterium's probiotic potential, such as whether the bacterium is commensal, is resistant to SIF, can adhere to intestinal epithelia and will stabilise intestinal microflora. (SIF = Simulated Intestinal Fluids) [105,107-134]. View Table 2
Table 3: Lists 13 potential probiotic bacteria, at the genus, species, and strain level (when applicable). Table 3 expresses whether or not, each of the 13 potential probiotic bacteria are recognised as safe for human consumption by the FDA; GRAS, and EFSA; QPS. The potential for side-effects is listed and if the bacteria is already currently used in probiotics. GRAS = Generally Recognised as Safe, QPS = Qualified Presumption of Safety [105,126-135]. View Table 3
Table 4: Lists 13 potential probiotic bacteria, at the genus, species, and strain level (when applicable). Table 4 examines the 13 potential probiotic bacteria specifically for their applicability against the development of the disease Necrotising Enterocolitis (NEC), by analysing BCFA membrane content, both anteiso- and iso-, and whether the bacterium produces short-chain fatty acids (SCFA) [43,56-58,86,87,137-142]. View Table 4
Table 5: Table 5 displays the whether the listed strains of probiotic bacteria are resistant or susceptible to the following cell wall inhibiting antibiotics; Penicillin (PEN), Ampicillin (AMP), Dicloxacillin (DIC), Vancomycin (VAN), Bacitracin (BAC). (S = Sensitive |R = Resistance | N/A = Information not available |R/S = Bacteria has demonstrated resistance and susceptibility in studies) [130,131,144,146-148]. View Table 5
Table 6: Table 6 displays the whether the listed strains of probiotic bacteria are resistant or susceptible to the following protein synthesis inhibiting antibiotics; Tetracycline (TET), Gentamycin (GEN), Kanamycin (KAN), Neomycin (NEO), Clindamycin (CLI), Chloramphenicol (CHL), Erythromycin (ERY), Mupirocin (MUP). (S = Sensitive |R = Resistance | N/A = Information not available | R/S = Bacteria has demonstrated resistance and susceptibility in studies) [130,131,144,146-148]. View Table 6
In Table 2, all 13 potential probiotic bacteria are screened for their efficacy to work within the intestinal environment, and positively affect the intestinal microbiome (Table 2). All bacteria, except Propionibacterium sp., were able to be defined down to the strain level, which is important when defining a bacterium's genetic capabilities, with genes often differing between strains of the same species (Table 2) [80]. Propionibacterium sp. were unable to be defined by strain, as probiotic Propionibacterium sp. are mainly represented naturally in dairy-products and have been minimally used in commercial probiotics [105]. Knowledge of the origin source of a bacterium is also necessary, as when an origin source is similar to or the same as the intended inoculation site, the potential efficiency of that bacterium to succeed in that environment is increased [106]. All Bifidobacterium sp. and, Lactobacillus rhamnosus PA-LR12 and Lactobacillus casei Lc-11, are all naturally found within the human gastrointestinal tract, which highlights the expected success of these bacteria to adhere to intestinal epithelia and survive in the intestinal environment (Table 2) [106-110]. All the other bacteria listed are known to originate from non-human gastrointestinal sources, with all the Propionibacterium sp. being identified in dairy-products, both Bacillus sp. being identified in plant or soil matter, and Lactobacillus plantarum LA-PL10 also being identified in plant matter (Table 2) [111,112]. However, despite not having a human gastrointestinal origin, all these bacteria have been confirmed to be resistant to intestinal fluids, demonstrating the capability to survive varying pH levels and bile acids, as well as adherence to the intestinal epithelia (Table 2) [109-125]. Moreover, all 13 bacteria are known commensal non-pathogenic bacteria and have all been observed to positively affect the intestinal microflora and stabilise intestinal microbiomes [105,126-134] (Table 2).
In Table 3 it is shown that all 13 bacteria have been previously assessed for safety and quality control by the governing bodies: FDA and EFSA (Table 3) [105,126-135]. All strains of bacteria, with the exception of Propionibacterium acidipropionici and Propionibacterium jensenii have been recorded as GRAS by the FDA, and all 13 strains were classified as QPS by the EFSA (Table 3) [105,126-135]. All bacteria having certification by either or both the FDA and EFSA, and are defined as safe for human consumption, and have the potential to be used as food-additives. Moreover, the success of probiotic bacteria being utilised commercially as a probiotic requires the in-depth knowledge of potential and adverse side-effects which may occur once a regime of the probiotic has been commenced by a consumer [96,97]. The side-effects encountered by individuals after the enumeration of their intestines by each of the 13 listed probiotic strains were minimal, ranging from increased gas production and bloating to acute diarrhea in rare occasions [105,126-135]. The increased production of gas and subsequent bloating of individuals, although considered a side-effect of probiotic consumption, can actually pertain to the course of action of the probiotic. The higher levels of gas being produced is not unordinary, as when the number of good microbiota in the intestinal environment increases, in particular those which break down carbohydrates and produce SCFAs, gas is often the accumulative end-product [136].
Table 4 moves past a general assessment of the bacteria for probiotic qualities and delves into the applicability of each bacterium in regard to their ability to stabilise the intestinal microbiota in individuals with NEC. Although NEC is a disease which results from a myriad of factors, the lack of BCFAs throughout the gastrointestinal tract of an infant due to premature birth, ensures the imbalanced and unhealthy intestinal microbiome that precedes NEC [4,11,17]. As premature infants have missed out on the consumption of BCFAs via vernix caseosa in-utero, their intestinal environment in turn lack the specific niche that is created by the presence of BCFAs, and consequently the BCFA-containing bacteria that would colonise the intestines [4,11,17]. Therefore, when selecting a bacterium with the potential to create this BCFA-niche within the intestinal environment of premature infants, bacteria with a high BCFA content must be selected for. The production of SCFAs by bacteria is also of interest when investigating the potential of a probiotic bacterium to affect a positive influence against NEC, due to SCFAs role in modulating intestinal health and inflammatory responses [23-25,89,91,92]. However, the production of SCFAs by the probiotic bacterium is not necessary, as long as the selected bacterium has the ability to increase the colonisation rates within the intestines of bacteria which do produce SCFAs [76,78].
Immediately from Table 4, it is concluded that all Lactobacillus sp.; L. rhamnosus PA-LR12, L. casei Lc-11 and L. plantarum LA-PL10, only have trace-levels of anteiso-BCFAs and no iso-BCFAs (Table 4) [58]. From the low levels of BCFAs seen in Lactobacillus sp. they are not expected to be capable of increasing levels of BCFAs in the intestinal environment, and therefore are deemed as not appropriate candidates for the use as a probiotic against the development of NEC (Table 4) [58]. Some of the listed Bifidobacterium sp. were also observed to contain low levels of BCFAs; Bifidobacterium lactis AD011 (1.5-2.5%), Bifidobacterium longum BB536 (0.6-1.0%) and Bifidobacterium infantis M-63 (0.6-1.0%) and were consequently taken out of contention for the use a probiotic to lower the incidence of NEC (Table 4) [58].
Based upon BCFA-content, 6 of the 13 listed bacteria in Table 4 were identified to contain > 20% BCFAs and are consequently accepted as bacteria with the probiotic potential to lower the incidence of NEC [4,17]. Species of bacteria belonging to the Bacillus genera were seen to have the highest amount of BCFA-content; Bacillus coagulans SZN1969 (89.0%) and Bacillus subtilis DE111 (95.0%), containing both anteiso-BCFAs and iso-BCFAs in relative amounts [137]. The Propionibacterium sp. were identified to contain the next largest quantities of both anteiso-BCFAs and iso-BCFAs; P. acidipropionici (42.0-50.0%), P. freudenreichii (40.0-52.0%) and P. jensenii (38.0-57.0%) [57] and only one Bifidobacterium sp. was observed to have a BCFA membrane content >20%; B. breve M-16V (24.6-25.0%) [58]. From the analysis of BCFA membrane content of these 13 probiotic bacteria, the Bacillus species; B. coagulans SZN1969 and B. subtilis DE111, have been observed to have considerably larger amounts of BCFAs in their membranes than the other 11 probiotic bacteria [57,58,137-139]. Although, both B. coagulans SZN1969 and B. subtilis DE111 do not produce SCFAs, whilst the other 11 probiotic bacteria have been observed to produce SCFAs from the fermentation of dietary carbohydrates [86,87,140-142]. However due to the high BCFA content observed in both Bacillus sp., a considerable rise in intestinal BCFAs could be expected with the consistent ingestion of a Bacillus sp. based probiotic, as many of the vegetative cells of Bacillus sp. would perish in the stomach and subsequently release BCFAs into the gastrointestinal environment [143]. As a result of the influx of BCFAs from a Bacillus sp. based probiotic, BCFA containing bacteria that do produce large quantities of SCFAs will be able to colonise, such as; Bifidobacterium sp., Propionibacterium sp. and Bacteroides sp [58,86,87,137-142]. From the prebiotic action of Bacillus sp. BCFAs, Bacillus sp. are implicated as the best candidates for a probiotic capable of dramatically increasing the levels of both anteiso-BCFAs and iso-BCFAs within the gastrointestinal tract of premature infants and creating a stable intestinal microbiome able to modulate intestinal inflammation via SCFAs [57,58,86,87,137-143].
In Table 5, Table 6 and Table 7, the remaining 6 potential probiotic bacteria are screened for sensitivity and resistance to some of the most commonly used antibiotics, to establish what bacterium may harbour antibiotic resistant genes (Table 5, Table 6 and Table 7). B. breve M-16V expresses resistance to 5 of the 18 listed antibiotics, which presents B. breve M-16V as the greatest hazard for potential horizontal gene transfer of antibiotic resistant genes, out of the 6 listed bacteria (Table 5, Table 6 and Table 7) [144]. As result of the great degree of antibiotic resistance demonstrated by B. breve M-16V, B. breve M-16V is consequently eliminated from contention for probiotic selection [145]. All Propionibacterium sp., P. acidipropionici, P. freudenreichii and P. jensenii, are rejected from being selected as potential probiotics due to consistent reports of antibiotic resistance and also the lack of available information on their antibiotic susceptibility and resistance [146-148]. Both Bacillus sp. are presented as ideal candidates for probiotic use, with B. coagulans SZN1969 exhibiting sensitivity to 16 of the 18 listed antibiotics (the effects of dicloxacillin and rifampicin on B. coagulans SZN1969 was undetermined) and B. subtilis DE111 being sensitive to all 18 antibiotics [131,132]. Due to the susceptibility of B. subtilis DE111 to all 18 listed antibiotics, B. subtilis DE111 presents the lowest probability for the introduction of antibiotic resistant genes into the intestinal microbiome via horizontal gene transfer [101,132].
Table 7: Table 7 displays the whether the listed strains of probiotic bacteria are resistant or susceptible to the following DNA and RNA synthesis inhibiting antibiotics; Metronidazole (MET), Moxifloxacin (MOX), Ciprofloxacin (CIP), Rifampicin (RIF), Trimethoprim (TRI). (S = Sensitive |R = Resistance | N/A = Information not available| R/S = Bacteria has demonstrated resistance and susceptibility in studies) [130,131,144,146-148]. View Table 7
From the comparative analysis of the 13 bacteria in regard to effectiveness and safety for the use in the treatment of premature infants with NEC, both Bacillus sp., B. coagulans SZN1969 and B. subtilis DE111, proved to be highly viable options. B. coagulans SZN1969 and B. subtilis DE111, although not being of human gastrointestinal origin, demonstrated the appropriate qualities required of a potential intestinal probiotic bacterium, such as being a commensal bacterium, resistance to intestinal fluids, ability to adhere to intestinal epithelia and efficacy in modulating the intestinal microbiome (Table 2). Both B. coagulans SZN1969 and B. subtilis DE111 have also been identified as safe for human consumption, having passed FDA and EFSA approvals, being awarded GRAS and QPS status, respectively (Table 3). The safeness of introducing B. coagulans SZN1969 and B. subtilis DE111 into the intestinal microbiomes of infants was also determined by investigating the potential anti-microbial genetic material harboured by each bacterium (Table 5, Table 6 and Table 7). Both B. coagulans SZN1969 and B. subtilis DE111 exhibited the highest rates of susceptibility to the three classes of commonly used antibiotics, with only B. subtilis DE111 being susceptible to all the 18 listed antibiotics (Table 5, Table 6 and Table 7). Due to the lowest rate of potential antibiotic resistance genes seen in B. subtilis DE111, B. subtilis DE111 becomes the preferred option in regard to use as an infant intestinal probiotic over B. coagulans SZN1969 (Table 5, Table 6 and Table 7). Although B. coagulans SZN1969 demonstrates a wide range of susceptibility to antibiotics (17/18), the susceptibility or resistance of B. coagulans SZN1969 to dicloxacillin and rifampicin is unknown (Table 5, Table 6 and Table 7). From the lack of information relating to B. coagulans SZN1969 susceptibility/resistance to dicloxacillin and rifampicin, there is the potential for B. coagulans SZN1969 to introduce genes pertaining to either dicloxacillin or rifampicin resistance into the infant intestinal microbiome via horizontal gene transfer (Table 5, Table 6 and Table 7). Whereas B. subtilis DE111 was susceptible to all 18 antibiotics and posed the lowest risk of introducing genes conferring to antibiotic resistance into the infant's intestinal microbiome (Table 5, Table 6 and Table 7).
Due to the necessity of a probiotic bacterium for the treatment of NEC to contain and have the ability to produce large quantities of membrane BCFAs, both Bacillus sp. were set apart from the other potential probiotic candidates, with B. coagulans SZN1969 and B. subtilis DE111 containing BCFAs in the membrane at 90-95% (Table 4) [137]. The natural production of high volumes of BCFAs by B. coagulans SZN1969 (90-95%) and B. subtilis DE111 (90-95%) suggests that if either were consumed by a premature infant in relative quantities, upon the bacteria's death and subsequent lysing, the BCFAs present in the premature infant's intestines would increase drastically [144]. From the post-natal consumption of BCFAs, the necessary BCFA niche required by commensal BCFA bacteria may be created and a premature infant's intestines will have the opportunity to develop a stable and healthy intestinal microbiome, which would be incited by in-utero vernix caseosa consumption in full-term infants [4,11,17]. To establish whether a Bacillus sp. based BCFA probiotic has the ability to modulate the intestinal microbiome of premature infants post-natal, the fatty acid profiles of B. coagulans, B. subtilis and vernix caseosa must be compared. As a result of this comparison, the efficacy of a Bacillus sp. BCFA probiotic to enumerate a premature infant's intestines with the appropriate BCFAs and stimulate the development of a healthy intestinal microbiome in premature infants in the leu of in-utero vernix caseosa consumption can be hypothesised.
A review of the available literature allowed for a comparison of the BCFA content of vernix caseosa, B. coagulans and B. subtilis (Figure 3 and Figure 5). Vernix caseosa is seen to contain a considerably lower amount of BCFAs than B. coagulans (90-95%) and B. subtilis (90-95%), with 43-48% of the fatty acids in vernix caseosa having methyl-branches [11,17,137]. Although the content of BCFAs in vernix caseosa is almost half that of both Bacillus sp., vernix caseosa contained BCFAs with chain lengths ranging from C11-C26, whilst B. coagulans and B. subtilis BCFAs chain lengths ranged from C14-C17 and C12-C17, respectively (Figure 3 and Figure 5) [11,17,137]. The increased variation in BCFA chain lengths results in 30 types of BCFAs being identified in vernix caseosa, whilst a lower BCFA variation was observed in both B. coagulans, with 9 BCFAs, and B. subtilis, with 12 BCFAs (Figure 3 and Figure 5) [11,17,137].
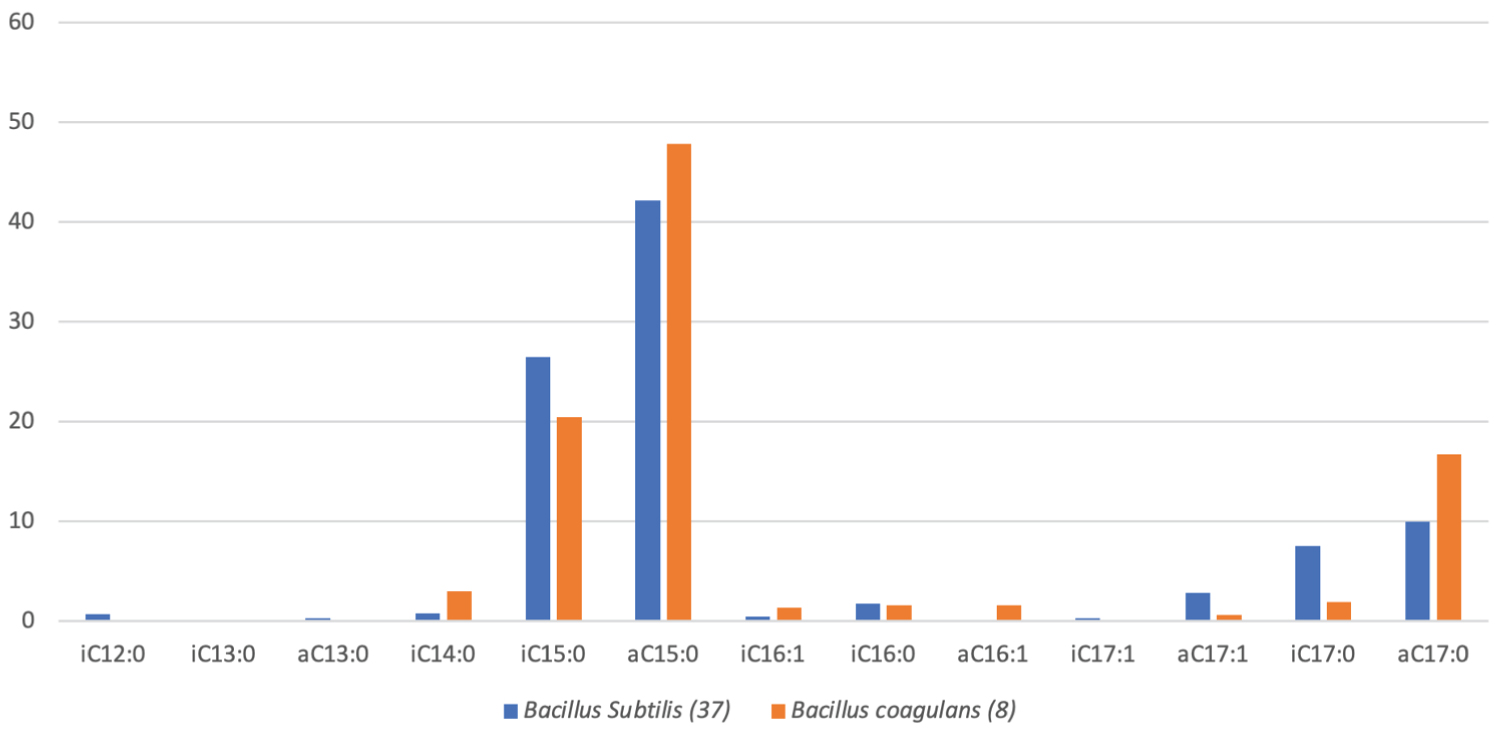 Figure 5: A box graph of the average percentage (%) of branched chain fatty acids (BCFAs) present in the phospholipid bilayer membranes of 37 Bacillus subtilis and 8 Bacillus coagulans. (A = anteiso, I = Iso, C = Carbon chain) [137].
View Figure 5
Figure 5: A box graph of the average percentage (%) of branched chain fatty acids (BCFAs) present in the phospholipid bilayer membranes of 37 Bacillus subtilis and 8 Bacillus coagulans. (A = anteiso, I = Iso, C = Carbon chain) [137].
View Figure 5
The higher volume of BCFAs seen in vernix caseosa is also attested to some BCFAs in vernix caseosa having di-methyl (diMe) branches and methyl (Me) branches that are not located on mid-chain positions, with dimethyl- and mid-chain methyl- BCFAs making up respectively, 10.9% and 1.9% of total fatty acids in vernix caseosa (Figure 3) [17]. This is a stark contrast of the fatty acid profiles of both B. coagulans and B. subtilis, that are only comprised of monomethyl BCFAs with the methyl-branches being located either on the anteiso- or iso-carbon chain positions [137]. The ratio of anteiso- and iso-methyl-branches also differed between vernix caseosa and Bacillus sp., with iso-BCFAs being the most prominent at 55.2% of total BCFA in vernix caseosa, whilst both B. coagulans and B. subtilis are alternatively dominated by anteiso-BCFAs, at 70.2% and 59.3% of total BCFAs, respectively [11,17,137]. Although, anteiso-BCFAs only comprised 17.2% of BCFAs in vernix caseosa, dimethyl-BCFAs that contained a methyl-branch located on the anteiso-carbon chain position made up 18.1% of total BCFAs present in vernix caseosa [17]. As past literature has indicated of the additive qualities of multiple methyl branches upon a membrane's fluidity and ordered state, it could be possible to suggest that di-methyl BCFAs with anteiso-branch locations are being utilised by intestinal bacteria as anteiso-BCFAs, however the role of di-methyl BCFAs within the intestinal microbiome is unknown [17,34].
Overall, the BCFAs predominantly utilised from vernix caseosa, as seen from the comparison of BCFAs identified in the vernix caseosa and the meconium of full-term infants in two separate studies, demonstrated a preferential uptake of anteiso- and iso- BCFAs with short carbon chain lengths (C11-C17) by the infant's intestinal microbiome [11,17]. The favoured acquirement of BCFAs with chain C11-C17 in length by the infant intestinal microbiome from vernix caseosa, with the shorter BCFAs (C11-C15) being elongated by the intestinal microbiome to C16-C17 BCFAs, is in line with other work which exposed anteiso-C17:0 and iso-C16:0 as the most preferentially utilised BCFAs by the human small intestines [11,17,149]. This preferred absorption of anteiso-C17:0 (3rd last) and iso-C16:0 (2nd) BCFAs may be due to the increased effectiveness of BCFAs which have a methyl-branch located at carbon position 15 on maintaining membrane structure and fluidity, as both anteiso-C17:0 and iso-C16:0 have methyl-branches located on the 15th carbon [34,149]. This increased utilisation of BCFAs with carbon chain lengths ranging from C11-C17 from vernix caseosa implicates the greatest potential success of B. subtilis to provide the BCFAs required by an infant's intestinal microbiome, as BCFAs contained by B. subtilis are seen to range from C12-C17, opposed to C14-C17 in B. coagulans (Figure 5) [137]. The greater range of BCFA chain lengths in B. subtilis will provide an increased availability of BCFAs to the infant intestinal microbiome, which will allow a wider diversity of BCFA containing bacteria to colonise the premature infant's intestines (Figure 5) [137]. It is also of importance for the bacteria used for the BCFA probiotic to contain relative levels of BCFAs with shorter carbon chains as seen in vernix caseosa (C11-C14), due to BCFAs with C11-C14 long chains not being isolated in the meconium (Figure 3 and Figure 5) [11,17,137]. It has been theorised that the predominant utilisation of the shorter chained BCFAs from the vernix caseosa by the infant intestinal microbiome is due to the elongation of shorter chain BCFAs to create anteiso-C17:0 and iso-C16:0 BCFAs, which is in line with the preferred absorption of anteiso-C17:0 and iso-C16:0 [149]. However, shorter chain BCFAs, such as iso-C14:0 are a major membrane component of the common infant intestinal bacteria, Bifidobacterium breve, which suggests that the importance of shorter chain BCFAs extends beyond being elongated to mid-chain length BCFAs (C16-C17) (Figure 4) [58].
B. subtilis was established to have a BCFA content most similar to the BCFAs observed in vernix caseosa, with a larger range of BCFA chain lengths than B. coagulans (C12-C17 and C14-C17, respectively) (Figure 3 and Figure 5). The importance of the elected probiotic strain for the treatment of NEC to have a wide range of BCFA chain lengths is indicative of vernix caseosa's BCFA profile, with chains ranging from C11-C26 (Figure 3). From meconium analysis of BCFAs, the favoured acquirement of BCFAs with chain C11-C17 in length by the infant intestinal microbiome from vernix caseosa was observed and suggests that the large BCFA chain length range seen in B. subtilis may be more effective in mimicking vernix caseosa (Figure 3 and Figure 5).
In order to establish whether a B. subtilis based BCFA probiotic would be capable of positively influencing the intestinal colonisation of major infant intestinal SCFA producers in the absence of in-utero vernix caseosa ingestion, in-vivo testing of the B. subtilis. based BCFA probiotic is required. Although, based on the similarities of BCFA present in the SCFA producers, Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp., and B. subtilis, the potential for a B. subtilis BCFA probiotic to positively influence the colonisation of Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp. can be perceived, as a similar range of BCFA chain lengths is observed (C12-C17 and C13-C18, respectively) (Figure 4 and Figure 5) [56-58,137].
Moreover, for a B. subtilis BCFA to effectively modulate the infant intestinal microbiome like vernix caseosa, the B. subtilis BCFA probiotic must not only be similar regarding BCFA content but must also be administered for at least one month post-natal to mimic vernix caseosa ingestion in-utero [150]. In partisan with the enumeration of the intestines with BCFAs, the transmission of commensal BCFA bacteria, Bacteroides sp., Bifidobacterium sp. and Propionibacterium sp., is also pivotal, and is seen to occur naturally during birth, breastfeeding and physical touch [65-68].
I would like to extend thanks to my collaborator Dr Anya Shindler, my La Trobe University supervisors Dr Anya Shindler, Dr Jennifer Wood, Prof Ashley Franks, and my Vernx supervisor Prof Craig Patch.