Leptospirosis is one of the most common zoonoses in the world. Human infection occurs after exposure to contaminated water or soil; hence infection is more common in rural settings. It is an uncommon cause of acute liver injury, and usually causes an anicteric illness. Here we describe a rare case of a 50-year-old man who presented with jaundice, transaminitis, hyperbilirubinemia, and acute kidney injury. He was diagnosed with Leptospirosis with reported history of exposure to rodents in an urban setting. He was treated with doxycycline with subsequent resolution of his liver injury.
Given the low incidence in non-tropical urbanized areas and the ease of treatment if started promptly, our case highlights the importance of identifying leptospirosis as a potential cause for acute liver injury in the appropriate clinical scenario.
Leptospirosis, Weil's Syndrome, Hepatitis, Icteric Leptospirosis
Leptospirosis is a wide-spread and prevalent zoonotic disease caused by pathogenic spirochetes of the genus Leptospira. It is thought to be an underreported infection, with an estimated incidence of more than one million cases annually worldwide. In the United States, however, the incidence is low [1].
This organism infects both wild and domestic mammals, like cattle, rodents, swine, dogs, horses, and sheep. Rodents are the most important reservoirs, and once infected, they harbor and shed the pathogen through their urine. Human infection occurs when animal urine or contaminated water or soil comes in contact with mucous membranes, conjunctivae, or skin lacerations [2].
On average, clinical manifestations of leptospirosis start after a 10-day incubation period. The infection varies from mild and self-limited illness in most cases, to a severe and potentially fatal disease. Symptoms are non-specific and include a wide range of different system involvement including gastrointestinal, respiratory, musculoskeletal, hepatic, and genitourinary systems.
A 50-year-old African American man presented to the emergency department of a New York City hospital with a chief compliant of bilateral leg weakness. Patient said that one week prior to presentation, he had sudden onset of abdominal cramps, nausea, multiple episodes of non-bloody, non-bilious vomiting, and watery diarrhea, which all lasted for a few days. Patient attributed his gastrointestinal symptoms to street food he ate prior to the onset of his illness. His condition deteriorated, and he developed subjective fevers, chills, and severe muscle aches, mainly in his legs. Patient's muscle pain progressively worsened, to the point where he felt his legs became weak, causing difficulty walking, so he decided to seek medical attention. He denied any recent travel, sick contacts, alcohol use, use of over the counter or herbal supplements, similar episodes in the past, or prior history of liver disease. The patient had a past medical history significant for syphilis, treated with penicillin as a teenager, hemorrhoids, and a “kidney problem”, which he could not provide further details about. He had no significant past surgical history. He reported smoking marijuana, denied cigarette smoking or other illicit drug use. He had no family history of liver or gastrointestinal diseases or malignancy. He had no allergies. He worked at a restaurant, and was sexually active with one long-term female partner, he did not use protection. Review of systems revealed perianal discomfort which he attributed to hemorrhoids.
Vital signs on admission were blood pressure of 174/97 mmHg, T 97.9 °F, heart rate 103 beats per minute, respiratory rate 18 breaths per minute, and oxygen saturation of 98% on room air. Physical exam was remarkable for scleral icterus and jaundiced skin. Abdominal examination showed epigastric tenderness without guarding or rigidity, the abdomen was otherwise soft, non-distended, without palpable masses or organomegaly. Musculoskeletal exam revealed bilateral lower extremity tenderness, muscle power and sensation were preserved.
Laboratory data on presentation were significant for a hemoglobin of 8.8 gm/dl [normal 13-17 gm/dl], white blood cell count of 12500 per microliter [normal 4500-11000 per microliter], platelet count of 38 k/uL [normal 150-400 k/uL], Blood Urea Nitrogen of 101 mg/dL [normal 8-26 mg/dL], Creatinine 4.6 mg/dL [normal 0.5-1.5 mg/dL], INR 1.12 [normal ≤ 1.1], Creatine Kinase 7173 unit/L [normal 20-200 unit/L], and Lipase 230 [normal < = 61 U/L.] Liver chemistry tests were notable for aspartate aminotransferase 207 until/L [normal 9-48 unit/L], alanine aminotransferase 102 until/L [normal 5-40 unit/L], alkaline phosphatase 116 units/L [normal 56-119 units/L], total bilirubin 15.9 mg/dl [normal 0.2-1.2 mg/dL], and direct bilirubin 15.4 mg/dl [normal 0.0-0.3 mg/dL] (Figure 1 and Table 1). Right upper quadrant abdominal ultrasound showed normal liver size, no biliary dilatation, CBD diameter of 2 mm, and contracted gallbladder with sludge. CT abdomen and pelvis with oral contrast revealed small liver cysts without any biliary dilatation.
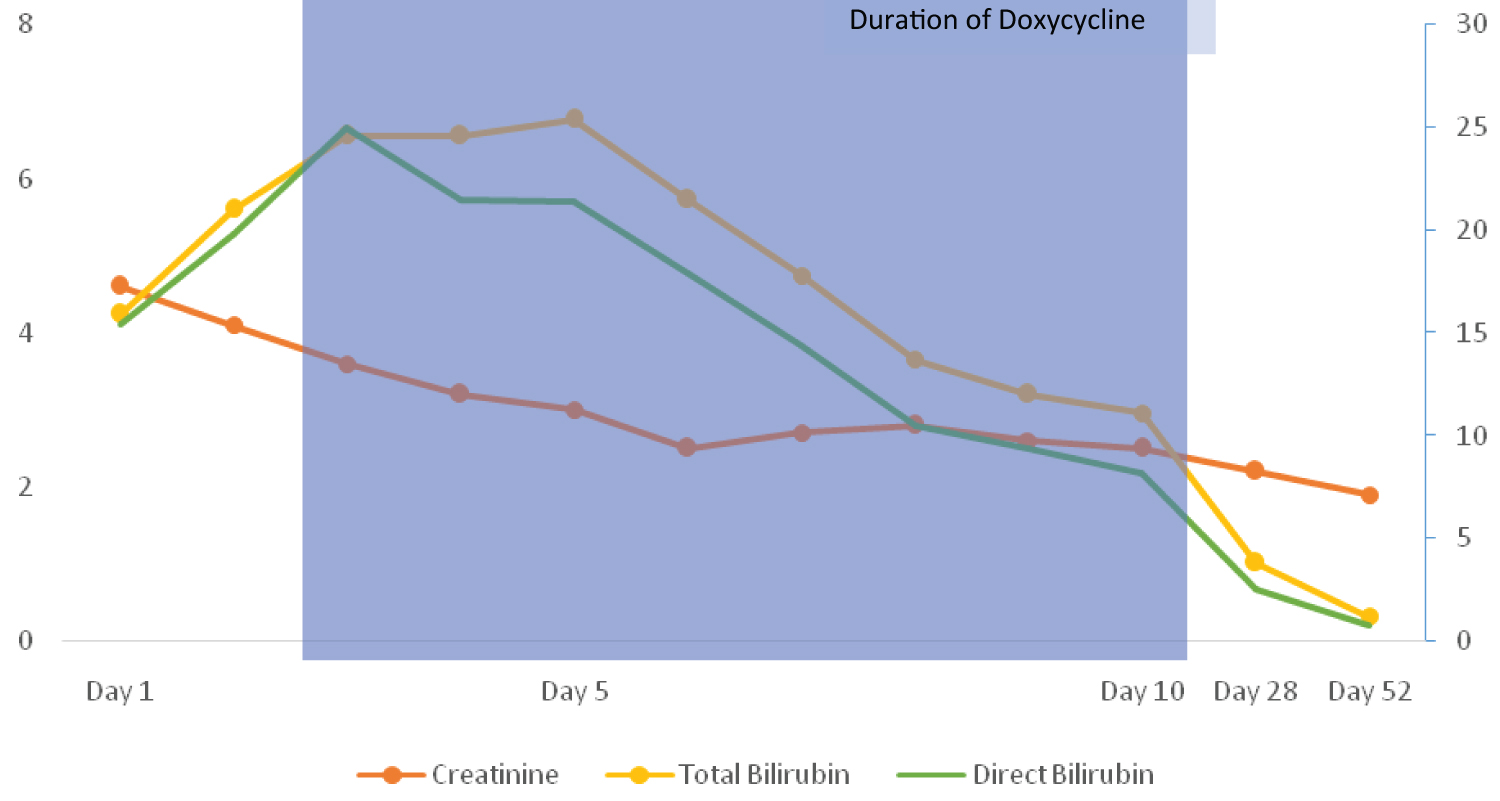 Figure 1: Creatinine and bilirubin trends.
View Figure 1
Figure 1: Creatinine and bilirubin trends.
View Figure 1
Table 1: Biochemical profile of abnormal variables. View Table 1
Patient was admitted to the intensive care unit, and his serum bilirubin continued to rise, reaching levels of 25.4 and 21.4 mg/dl for total and direct bilirubin, respectively. Viral serologies for Hepatitis A, B, C, E, Varicella, HIV, and CMV were all negative. Epstein Barr Virus IgG and IgM titers were elevated. Autoimmune workup including ANA, smooth muscle antibody, DNA antibody, and immunoglobulin G, were negative. MRCP abdomen showed multiple benign hepatic cysts with no biliary dilatation. Upon further questioning, patient reported exposure to rats' urine at his workplace, where he handled garbage without adequate protection, and noted to have multiple skin breaks and abrasions.
Leptospirosis Qualitative DNA PCR was sent and came back positive. Patient was started on oral Doxycycline 100 mg every 12 hours, with subsequent clinical and biochemical improvement (Refer to Table 1). Patient was discharged home to complete a 14-day course of doxycycline. Repeat liver chemistry tests during a follow up visit revealed normalization of his ALT, AST, total, and direct bilirubin.
Acute liver injury is a common clinical scenario faced by clinicians. The most common infectious agents that cause hepatic injury are the hepatotropic viruses (hepatitis A, B, C, D, and E); however, it is important to be cognizant of other infectious agents that can cause hepatic insult. Spirochetes, a group of spiral-shaped bacteria, including leptospirosis, syphilis, and borrelia burgdorferi, have been reported to cause liver injury [3]. The incidence of leptospirosis in the United States is low, with about 100-150 cases reported annually, and it is a nationally notifiable disease [4]. It usually occurs in tropical climate and rural settings where human exposure is higher. In an urban area like New York City, an average of 3 cases per year are reported. The clinical manifestations of leptospirosis infection start after an incubation period of 2 to 30 days. Most cases are either asymptomatic, or mild and self-limited, with less than 10% of cases progressing to severe illness with multiorgan failure. Common symptoms include fever, rigors, myalgias, headaches, conjunctival suffusion, non-productive cough, nausea, vomiting, and diarrhea [5]. Weil's Syndrome classically refers to renal and liver failure with jaundice. Leptospirosis infection can cause additional multi-organ dysfunction resulting in thrombocytopenia, rhabdomyolysis, aseptic meningitis and acute pulmonary haemorrhage [6].
Anicteric leptospirosis and Weil's Syndrome are two distinct forms of liver injury with leptospirosis, with more than 90% of the cases being anicteric. In anicteric disease, the infection is usually biphasic, with the first phase starting abruptly with a viral-like illness lasting 5-7 days associated with fever, myalgias, and conjunctival suffusion; an important diagnostic clue. After a brief period of symptom resolution, the second phase is characterized by nausea, vomiting, diarrhea, and abdominal pain lasting 4-30 days. During the second phase, hepatic involvement with elevated bilirubin and serum transaminases is rare however hepatomegaly has been reported [3]. Icteric leptospirosis (Weil's Disease) occurs in 5-10% of those infected. It constitutes jaundice in the acute phase for multiple weeks with transition to the second phase without resolution of symptoms. The second phase presents with periods of fever, renal impairment, and hepatic dysfunction. Liver injury most classically presents with an isolated direct hyperbilirubinemia with levels reaching up to 80 mg/dL with mild elevations in transaminases not exceeding 200 units/L3. Recent studies have shown that conjugated bilirubinemia could be due to hepatic infiltration of leptospirosis with subsequent disruption of hepatic intercellular junctions leading to leakage of bile from bile canaliculi into hepatic sinusoidal blood vessels [7]. Further multisystem destruction is attributable to high levels of leptospiremia that trigger systemic and local inflammatory responses. Patients with severe disease have been shown to have elevated IL-6 and TNF-alpha, indicating evidence of cytokine storm pathology [7].
Leptospirosis is diagnosed with clinical suspicion and confirmed with molecular tests, serology, culture and/or antigen detection. Initial laboratory settings can demonstrate mild leukocytosis with left shift, thrombocytopenia, hyponatremia, elevated creatinine, rhabdomyolysis, and hepatic derangements as mentioned above [8,9]. Serological methods of detection can miss infections as antibodies take 5-7 days to develop while nucleic acid detection via PCR can detect patients who have leptospiremia earlier in the disease course. Leptospirosis DNA can be detected in blood early in the disease course and in urine and CSF few days into the acute illness. It is recommended to use a combined serological and molecular approach for higher diagnostic sensitivity [10,11]. Culture of blood, CSF, and urine demonstrate variable results depending on timing of culture before antibiotics. Urine cultures have the highest sensitivity after second week of illness and remain positive for up to 30 days [12]. Treatment of leptospirosis varies based on mild versus severe presentation. Most cases are mild and self-limited, and often don't require antimicrobial therapy as patients don't seek medical care. Mild disease can be treated with doxycycline 100 mg twice a day for 7 days or azithromycin 500 mg once a day for 3 days. Severe disease is treated with intravenous antibiotics for seven days. Preferred antibiotics are Penicillin, Ceftriaxone, Doxycycline, and cefotaxime. Supportive therapy is often required in severe disease with renal replacement therapy, ventilation assistance, and blood product transfusion. Extracorporeal membrane oxygenation and plasmapheresis have been used sparingly [13-15].
Our case also highlights an unsettling trend of increasing cases in the Bronx borough of New York. As per New York City Health Communicable Disease Database, between the year 2000 and 2014 NYC saw an average of 1.92 cases per year; for a total of 27 cases. Over the years 2015-2019 (latest years of available data), there has been a threefold increase in reported cases, with average of 6.4 reported cases per year; 32 in total. Amongst the five boroughs, The Bronx was noted to have 43% of all cases during this period of increased cases [16]. Most infections also occurred in high poverty areas within each New York Borough and predominantly among men. This increasing trend of Leptospirosis in urban environments could be attributed to poor socioeconomic status [17] unsanitary waste handling and worsening rodent infestation [18,19].
Leptospirosis is a rare but an important cause of abnormal liver enzymes that is under recognized in urban settings. Leptospirosis infection should be considered in a patient with recent travel to endemic areas with exposure to bodies of water, animals or simply those who live in a poor socioeconomic region in an urban environment. Disproportionate direct hyperbilirubinemia and conjunctival suffusion are hallmark signs with progression to renal and liver failure characteristic of Weil's syndrome. The importance of considering leptospirosis as a differential for abnormal liver enzymes is prudent given high mortality rates in those with untimely diagnosis with progression to Weil's disease.
Informed consent was taken from the patient prior to starting our report.
All authors have no conflict of interest to declare.
None.