Chronic kidney disease is a public health problem that causes severe complications to the patient. The major damage is the loss of muscle mass, with a consequent reduction in the functional capacity that is associated with hypertension and increased oxidative stress. This study aimed to evaluate the effect of intra-dialysis resistance training on the hemodynamic parameters, oxidative stress and quality of life in patients with chronic kidney disease.
The volunteers underwent a physical training during the hemodialysis sessions. The hemodynamic parameters, oxidative stress, the quality of life, the functional autonomy and one repetition maximum (1 RM) were evaluated.
The data did not show alterations in the hemodynamic parameters, nitric oxide or lipid peroxidation after 12 weeks of resistance training. We observed significant differences in the quality of life in the following dimensions of the Kidney Disease Quality of Life - Short Form (KDQOL-SF™) questionnaire after 12 weeks of resistance training: Functional capacity, physical aspects, vitality, social and emotional aspects and mental health (P < 0.05). The time needed to execute the functional autonomy test was improved after the resistance training, and strength was increased significantly in all the exercises, as assessed by the 1 RM test (P < 0.001).
In conclusion, after 12 weeks, the intra-dialysis resistance training did not modify the hemodynamic parameters neither the oxidative stress profile of these patients, but improved their quality of life. These data show that intra-dialysis resistance training is safe and can provide many physical and emotional benefits to chronic kidney disease patients.
Resistance exercise, End stage kidney disease, Nitric oxide, Lipid peroxidation, Hemodialysis
Chronic kidney disease (CKD) is a public health problem, and epidemiological studies have shown that millions of people are on renal replacement therapy (RRT) worldwide [1]. In Brazil, according to the Brazilian Society of Nephrology, more than 100,000 CKD patients were on RRT in 2013 [2].
Compared to healthy people or non-dialyzed CKD patients, end-stage renal disease (ESRD) patients experience muscle loss, lower exercise tolerance and physical fitness levels, reduced strength and functional capacity, and excessive fatigue [3]. The physical inactivity, sarcopenia and reduction of physical capacity are associated with increased mortality in these populations. Sedentary ESRD patients have 62% higher risk of mortality in one year compared with those who are the most active [4].
Cardiovascular disease is a major cause of death in CKD patients, and these patients have endothelial dysfunction, which is considered an early mediator of the atherosclerotic process [5]. Nitric oxide (NO), an important physiologic vasodilator and one of the factors involved in the anti-atherosclerotic process, is deficient in this population [6]. The reduction in NO levels is associated with oxidative stress [7] and contributes to cardiovascular events [8]. Cakir-Atabek, et al. have demonstrated, in healthy men, the reduction of oxidative stress after training with resistance exercises [9]. However, few data are available regarding the impact of training in ESRD patients especially in those underwent hemodialysis (HD). A disease such as CKD markedly reduces the quality of life during the years [10].
This study aimed to evaluate the effects of resistance training (RT) during dialysis sessions in the hemodynamic parameters, oxidative stress and health-related quality of life in CKD patients.
A total of 20 CKD patients on hemodialysis were recruited from Dialysis Unit of Universidade Federal de Sao Paulo, Sao Paulo, Brazil. Patients of both sexes, on treatment for at least 3 months by arterial-venous fistula were approached to participate in the study. Exclusion criteria included age less than 18 years, presence of anemia, myocardial infarction, angina or heart failure in the 3 months before the study, or comorbidities that could compromise the execution of the exercises.
This study was reviewed and approved by the Ethics Advisory Committee of the Universidade Federal de São Paulo (#1143/10). Informed consent was obtained from all individual participants included in the study.
This prospective study comprise 12 weeks of resistance training (RT) performed 3 times a week during HD sessions. Before starting the RT, all the patients were evaluated by a cardiologist through transthoracic echocardiography and a treadmill protocol with the intensity based on their physical and clinical conditions. All the tests were performed 1 week before the training. Three patients were excluded because they presented increased blood pressure and arrhythmias during the treadmill test, totalizing 17 patients assessed in this study. More over body composition analysis was performed in the inter-dialysis period by the plethysmography method with the Bod Pod® system, at baseline, for all patients.
The RT session was composed of exercises for the upper limbs (shoulder press, biceps curl and triceps curl) using dumbbells, for the lower limbs (leg extension, hip flexion, leg curl and hip abduction) using ankle weights or elastic band (Thera Band®).
A one-repetition-maximum test (1 RM) was performed to determine the workload used in the exercises using dumbbells and ankle weights, as described by MacArdle, et al. [11]. Prior to this test, the volunteers performed an exercise session without the use of load, to learn the correct technique and execution of the movement. The RT was applied for a period of 12 weeks; the first 3 weeks were for adaptation to training with a load of 40% of the 1 RM, and the other 9 weeks included intensity training corresponding to 60% of the 1 RM, performed three times/week, one hour after the start of the HD session and lasting approximately 30 minutes. During the first week the volunteers performed leg curl and hip abduction using a green elastic band with a resistance similar to 2.7 kg, between the 2nd to 6th week with a grey band (3.5 kg), and between 7th to 12th week with a blue band (4.5 kg).
The model used alternated segments with unilateral movements; the movements for the upper limbs were executed with the non-fistula arm, and the movements for the lower limbs were executed simultaneously. Prior to all the RT sessions, the volunteers performed warm-up, followed by exercises with three sets of 12 repetitions with one-minute rest between sets.
The exercises were performed between the 75 and 105 minutes of the HD session. A 1 MMC test was performed to evaluate the strength before and after the 12th week of the RT.
Anautomatic blood pressure monitor BPA 100 (Microlife®, USA) was used for measuring the systolic blood pressure (SBP), diastolic blood pressure (DBP) and heart rate (HR) as validated by Bonso, et al. [12]. The mean blood pressure (MBP) and double product (DP) were calculated. The measurements were performed before, in the beginning and at the end of the RT, in the last HD session of the week, at baseline, 1st, 6th and 12th weeks.
Blood samples (5 mL), were collected in the beginning and at the end of the RT, in the last HD session of the week, at baseline, 1st, 6th and 12th weeks, for analysis of oxidative stress markers. The determination of Nitric oxide (NO) in the plasma was performed with a Nitric Oxide Analyzer by chemiluminescence (NOA™ 280, Sievers Instruments, Inc, Boulder, CO, USA) [13]. Thiobarbituric acid reactive substances (TBARS) [14] were determined by the colorimetric method; the absorbance was read at 534 nm in a spectrophotometer (Sunrise TM Tecan, Männedorf, Switzerland). The calculations of TBARS concentrations were performed using the extinction coefficient 1.56 × 105 M-1cm-1, and the results were expressed in nmol/L.
The assessment of quality of life was performed by Kidney Disease and Quality-of-Life Short-Form (KDQOL-SF™) questionnaire and functional autonomy protocol. KDQOL-SF™ consists of eight fields for the SF-36 dimensions and 11 specific fields concerning CKD as described where else [15]. For each dimension, a score-value from 0 (worst health status) to 100 (best health status) was obtained. The method of the questionnaire completion was previously explained to the patients by the researchers, and the questionnaire was applied before and after the RT period.
The Latin American Group of Development for Maturity (GDLAM) protocol was used before and after RT to assess the functional autonomy of the patients, based on four activities of daily living, including: walking 10 meters (W10m), standing from a seated position (SSP), lifting from the prone position (LPP), and standing from a chair and moving around the house (SCMH). The execution of each activity was measured in seconds, and the final score of functional autonomy (IG) was measured [16].
The data were expressed as the mean ± standard deviation (SD) or the median (Interquartile) when appropriate. Paired Student's t-test or the Wilcoxon test was used to compare the variables before and after RT. In the repeated measure ANOVA analysis, the Sidak post hoc was used. The differences were considered statistically significant when p < 0.05, and all the analyses were performed using SPSS 18.0 software for Windows (SPSS Inc., Chicago, USA).
The clinical and demographic characteristics of the patients were shown in Table 1. Hypertension and diabetes were the more common conditions which caused CKD. The patients were on HD for approximately two years. Considering BMI, no patient had protein energy wasting, overweight and obesity were found in 12% and 24% of the patients, respectively. There was no patient with anemia. The ejection fraction was within the normal ranges and left ventricular hypertrophy was observed in 4 (23%) patients.
Table 1: Clinic and demographic characteristics of hemodialysis patients. View Table 1
There were no significant differences in hemodynamic and oxidative stress parameters during the study (Table 2 and Table 3), except HR, (Table 2), that showed differences After EXE at 1st, 6th and 12th vs. baseline (85.6 ± 15.4, 87.6 ± 19.2, 87.3 ± 17.2, vs. 80.3 ± 14.1, P < 0.05). The KDQOL-SFTM scores were depicted in Table 4. There was a significant increase in six dimensions of the SF-36 after RT, as follows: functional capacity, physical aspects, vitality, social aspects, emotional aspects and mental health. There was a trend to improve the dimension related to pain. The eleven CKD specific dimensions did not show differences, however, there was a trend to improve the symptoms/problems dimension.
Table 2: Hemodynamic parameters: Systolic blood pressure (SBP), diastolic blood pressure (DBP), heart rate (HR), mean blood pressure (MBP) and double product (DP) in the baseline and 1st, 6th and 12th weeks of resistance training. View Table 2
Table 3: Determination of Nitric Oxide concentration and plasma lipid peroxidation using the TBARS before and after exercise, in the baseline and in the 1st, 6th and 12th weeks of Resistance Training. View Table 3
Table 4: Scores of dimensions: Quality of life questionnaire (KDQOL-SFTM). View Table 4
There was a significant reduction of the time necessary to perform the GDLAM protocol tests after 12th weeks of RT (Figure 1). There was an decrease in SSP time (11.0-14.9) vs. 8.9 (7.9-11.9) seconds; p = 0.01]; in LPP [3.8 (2.0-4.7) vs. 2.3 (1.9-3.8) seconds; p = 0.001]; in SCMH [47.3 ± 1.2 vs. 43.1 ± 7.7 seconds; p = 0.02] and in IG [28.9 (24.3-33.7) vs. 25.1 (21.9-30.1) seconds; p = 0.01]. One patient could not execute the SSP test. Moreover 1 RM showed a significant increase in the strength after RT in all exercises (p < 0.01): Shoulder press (9.76 ± 2.39 vs. 6.65 ± 1.66), biceps curl (10.65 ± 2.91 vs. 6.97 ± 1.97), triceps (6.88 ± 1.97 vs. 4.21 ± 1.91), leg press (13.88 ± 4.4 vs. 6.94 ± 2.51) and hip flexion (9.88 ± 6.33 vs. 6.06 ± 5.44; Figure 2).
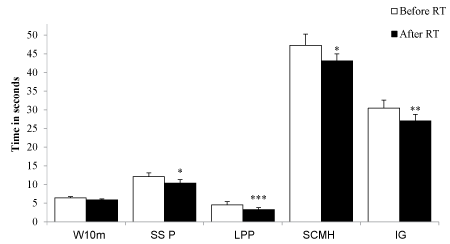 Figure 1: The "Grupo de Desenvolvimento Latino Americano da Maturidade" (GDLAM) functional autonomy test was conducted before and after 12 weeks of RT. W10m = walking 10 meters; SSP = stand up from a seated position; LPP = lift from the prone position; SCMH = standing up from a chair and moving around the house; IG = functional autonomy score. The data were expressed as mean ± SEM. The statistical analysis before and after RT was realized using paired Student's t-test. * = P < 0.05 vs. before, ** = P < 0.01 vs. before and *** = P < 0.001 vs. before. View Figure 1
Figure 1: The "Grupo de Desenvolvimento Latino Americano da Maturidade" (GDLAM) functional autonomy test was conducted before and after 12 weeks of RT. W10m = walking 10 meters; SSP = stand up from a seated position; LPP = lift from the prone position; SCMH = standing up from a chair and moving around the house; IG = functional autonomy score. The data were expressed as mean ± SEM. The statistical analysis before and after RT was realized using paired Student's t-test. * = P < 0.05 vs. before, ** = P < 0.01 vs. before and *** = P < 0.001 vs. before. View Figure 1
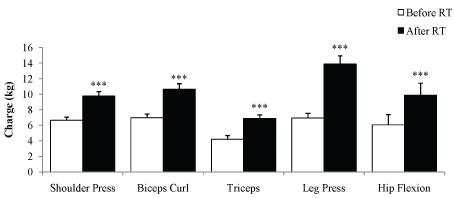 Figure 2: 1 maximum repetition test (1 MR) in Kg, performed before and after 12 weeks of RT. Paired Student's t-test. ***P < 0.001 vs. Before RT. View Figure 2
Figure 2: 1 maximum repetition test (1 MR) in Kg, performed before and after 12 weeks of RT. Paired Student's t-test. ***P < 0.001 vs. Before RT. View Figure 2
All patients completed the study. During the study, 4 incomplete exercise sessions occurred, due to hypotension (2 episodes), cramp and excessive inter-dialysis volume gain with high blood pressure and dyspnea.
The present study demonstrated that 12 weeks intra-dialytic RT improves quality of life, functional autonomy and strength, without altering hemodynamic parameters or oxidative stress.
Some studies had evaluated the effect of exercises performed during HD on blood pressure with conflicting results. These studies had shown reduction on blood pressure after physical exercises [17,18]; on the other hand, in others, similar to present data, no effect was observed [19]. These differences could be partially explained by the characteristics of the studied population and the intensity and type of exercise used. Most of the studies that had a beneficial effect performed aerobic exercises or combined aerobic and resistance exercises [17,18]. It has been suggested that the aerobic exercise improve blood pressure, reduces the use of antihypertensive drugs and increases the plasma level of HDL cholesterol [20]. Although there are fewer studies, it appears that some have suggested that RT could improve the blood pressure since it is combined with aerobic activities [18].
The consideration of regular physical activities in the management of chronic diseases, has focus on the reduction of cardiovascular disease and stroke risk [21]. The kidney is the main supplier of L-arginine for physiological NO synthesis, and renal complications could induce a reduction in L-arginine concentrations, leading to injuries in the vasculature. Low levels of l-arginine can result in 'uncoupling of NOS enzymes' [22], which can be one of the reasons that NO did not change in our study after RT. But, it is known that aerobic exercises can decrease endothelin-1 (ET-1) concentrations, a potent vasoconstrictor produced by endothelial cells, which might contribute to the increased production of NO [23]; this increase in the NO production may be possibly through increasing blood flow and endothelial stretching during the exercise [24]. However, Park and Omi [25] have shown that resistance exercise or swimming for 8 weeks was enough to reduce the ET-1 levels and increase levels of NO; in our study we did not observe increases in NO levels, similar to other researchers, and its mechanisms are not clear and remain to be elucidated [26].
Patients undergoing HD have increased TBARS levels because of the inflammatory process resulting from HD treatment [27]. A study performed by Wilund, et al. showed that four months of aerobic exercise performed on a bicycle during HD decreased the TBARS in the exercised patients compared with the control group, and this finding could be attributed to regulation of the antioxidant enzyme activity promoted by physical exercise [28].
In the present study a significant increase in six of eight dimensions were observed, indicating that RT intra-dialysis could be an useful tool in these patients, for increasing quality of life. The results of Oh - Park, et al. [29] were similar to our results with respect to improvement in the dimensions of functional capacity, physical aspects and mental health; the first study used aerobic exercise during HD. In these studies the authors did not find significant improvement in the dimensions relating to the social aspects, emotional aspects and vitality, which is one of the benefits of physical exercise.
This study was the first to use the GDLAM method of assessment in CKD patients. Chronic disease could decrease functional capacity, and we used the GDLAM test of functional autonomy [30], which is typically applied in the elderly; we used this test because of the similarities of comorbidities, the accuracy and the easy of application. The specific characteristics of this test do not induce exhaustion or fatigue in the patient. We found significant changes in the scores on the functional autonomy index, showing improvement on their daily activities such as walking without help, standing up from a chair, and going up and down stairs. RT for CKD patients can minimize the loss of muscle mass that is frequent in these patients, improving muscle strength.
The RT increased strength in almost all the exercises assessed with the 1 MR test, which occurred probably because of neuromuscular and organism adaptation. Similar results were observed by Headley, et al. [31], using dynamometry for the assessment of quadriceps strength.
The small number of patients enrolled in this study is a limitation. However, given the reported benefits of RT on HD patient health, further investigations should be conducted on a larger number of patients to test other types and periods of exercises.
In summary, our study showed that twelve weeks of inter-dialysis RT did not modify the hemodynamic parameters in CKD patients during HD, neither in terms of NO synthesis or TBARS. The training protocol was sufficient to change the quality of life in the patients, which was demonstrated by increasing scores on the KDQOL-SFTM questionnaire, improvement in the functional autonomy test and improvement in strength observed in the 1 MR test after the RT. Our study indicates that RT can be performed safely during the first hours of HD sessions, providing great benefits to patients in their daily living activities and leading to an improved quality of life. We suggest the implementation of such exercises as a routine in the dialysis clinics.
To Margaret Gori Mouro for technical support. This research did not have financial support.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
The authors declare that they have no conflict of interest.