Living donor renal transplant with grafts having complex vascular anatomy is technically difficult with higher complications. We herein present our experience of complex vascular anatomy living donor renal grafts as compared to grafts with simple vascular anatomy.
The is a retrospective comparative analysis of a prospectively maintained database of all the patients undergoing live related renal allograft transplant from January 2015 till Dec 2019. All adult transplants with graft with complex vascular anatomy were included and deceased donor and pediatric transplants were excluded.
There were 422 eligible transplant patients out of which 92 (21.8%) patients had grafts with complex vascular anatomy and 330 (78.2%) patients had single renal artery and vein. There were no major intra-operative complications. Warm ischemia time and operating time were significantly less in single artery group (p < 0.001). There was no difference in terms of urine output, fall in serum creatinine levels, delayed graft function (4.2% vs. 4.3%), primary graft non function (1% vs. 0.6%), urine leak (2.1% vs. 3%) and hospital stay.
Renal transplant with grafts with multiple renal vessels have equivalent outcomes as compared to simple vascular anatomy. Complex vascular anatomy living donor transplants should be done in high volume centers by experienced surgeons.
Renal transplant, Complex vascular anatomy, Recipient outcome
Chronic kidney disease is an increasing global public health problem, both in developed and developing countries [1]. The incidence of end stage renal disease in India is 151-232 per million per year. However, the renal transplantation rates are only 3.25 per million per year [2]. Deceased organ donation is still not commonly performed in India due to various social, cultural and religious issues and more than 95% of renal donations are live related. Ideally a living donor graft should have a simple vascular anatomy with single artery and vein for best outcomes [3]. Renal transplantation using grafts with multiple renal vessels are technically difficult with increased warm ischemia time; mean operative time and a higher risk for vascular and urological complications when compared to grafts with single renal artery [3,4]. Kidneys with multiple vessels in live renal donors are avoided and simple vascular anatomy grafts are preferred. However in live related renal allograft transplant, the donor pool is limited and ethical issue and legislation in India does not give much choice for recipients having donors with complex vascular anatomy. Using renal grafts with multiple vessels is one approach to expand the donor pool for renal transplantation [5-7].
There are few studies that have evaluated the influence of multiple renal vessels on graft function especially in living renal transplants. We herein present our experience with renal grafts with complex vascular anatomy in live related donors and its effect on recipient outcomes as compared to renal donors with simple vascular anatomy.
This is a retrospective analysis of data from a prospectively maintained database from January 2015 till December 2019 of all patients undergoing live related renal allograft transplant in a single surgical unit at a tertiary care center. Deceased donor and pediatric renal transplants were excluded from the study.
Donor demographic details, CT angiography findings and intraoperative details were recorded. The side of donor nephrectomy was decided on the basis of principle of leaving the better functioning kidney with the donor and choosing the kidney with the least complicated vascular anatomy. Standard guidelines for work-up of donor and recipient were followed.
Donors in whom left kidney was retrieved were operated on laparoscopically irrespective of vascular anatomy. At the time of laparoscopic donor nephrectomy, attempt was made to preserve all the renal arteries as well as veins. Initially at perfusion attempt was made to perfuse all arteries irrespective of their size. Decision to sacrifice or use the arteries was made at the time of bench dissection after perfusion. The detailed steps of laparoscopic donor nephrectomy have been given in our previous publication [8]. Similarly, donors in whom it was decided to take right kidney based on the vasculature, open donor nephrectomy was performed following the same principles as stated above.
Kidney was implanted preferentially in the right iliac fossa irrespective of the side of donor nephrectomy. If the right side was not available (previous transplant, vascular issues) than the kidney was implanted in left iliac fossa. In patients with single renal artery, vein and ureter, arterial anastomosis was done with either external iliac artery in an end to side fashion or with internal iliac artery in an end to end fashion depending upon the vessel caliber and surgeon preference. Venous anastomosis was always done with external iliac vein. Different combination of anastomosis was performed in patients with complex anatomy renal grafts according to the recipient vascular anatomy and vascular anatomy of graft kidney. In majority of cases, main renal artery with accessory or polar arteries were anastomosed to external iliac artery in an end to side fashion or internal iliac artery in an end to end fashion or variety of other methods as described in the results (Table 1). The renal vein in all the cases was anastomosed to external iliac vein in an end to side fashion. Only the larger single vein was anastomosed in all patients after ligating the smaller caliber vein if present. All the anastomosis were done using 6-0/7-0polypropylene sutures. The grafts were placed extraperitoneally in the right or left iliac fossa. Urinary tract continuity was established by modified Lich-Gregoir technique of extra vesical ureteroneocystostomy over a stent especially in patients with complex vascular anatomy.
Table 1: Type of arterial reconstruction performed. View Table 1
Outcomes were compared in terms of donor operative time, warm ischemia time-I & Rewarming time, cold ischemia time, recipient operative time, on table graft function and postoperative graft outcomes in terms of urine output, daily fall of serum creatinine and hospital stay in days till the time of discharge. Complications like delayed graft function, primary non-function, vascular thrombosis; urine leaks and need for surgical intervention in postoperative period (bleeding, hematoma, obstruction, thrombosis) were also noted.
Triple drug immunosuppression including tacrolimus, mycophenolate mofetil and prednisone was given to all patients in the standard dosage [9]. The trough blood concentration of 10-12 ng/ml for tacrolimus was maintained in the postoperative period. Induction agents like IL-2 receptor blocker basiliximab or antithymocyte globulin were used based on standard criteria [9].
Warm ischemia time-I & Rewarming time as well as cold ischemia time was calculated in all cases. Warm ischemia time-I was calculated from the clamping of renal artery in donor till the perfusion of kidney with histidine-tryptophan-ketoglutarate solution (Custodial©) solution. Cold ischemia time was calculated from the time of infusion of cold preservation solution to removal of kidney from ice for anastomosis. Rewarming time was calculated from the time of removal of graft kidney from ice to restoration of blood circulation after anastomosis of vessels in recipient. Operating time was calculated from the skin incision to the skin closure.
Acute rejection was suspected in patients with rise in serum creatinine more than 30% from baseline levels with or without clinical signs and symptoms of oliguria, graft tenderness and fever. A confirmatory biopsy was done in all cases whenever rejection was suspected. Methylprednisolone was used as the primary therapy in case of rejection at the dose of 10 mg/kg/day for 5 days.
Delayed graft function was defined as requirement for dialysis within one-week post-transplantation. Similarly, primary non-function was defined as an aggravated form of reperfusion injury, which resulted in irreversible graft failure without detectable technical or immunological causes.
Continuous variables were described as mean values and range, and categorical data by proportions. Paired t-test was used to compare measured data of paired samples. Scores of two paired groups was compared using Wilcoxon signed rank test while scores of two unpaired groups was compared using Mann Whitney U test. Fisher test was used for two unpaired groups with dichotomous data. A p value of 0.05 or less was taken as significant. Post-transplant urine output, serum creatinine levels, post-operative complications and hospital stay were analyzed using statistical analysis STATA and graphics software for windows 10.
During the study period, 483 renal transplants were performed at our center out of which 61 patients were excluded (pediatric and deceased donor transplants). Total of 422 patients were included in the study (Figure 1). Demographic and clinical profile in both the groups was comparable (Table 2). 92 (21.8%) patients underwent kidney transplantation with grafts with multiple renal arteries and 330 (78.2%) patients with a renal graft with single renal artery and vein (Figure 1).
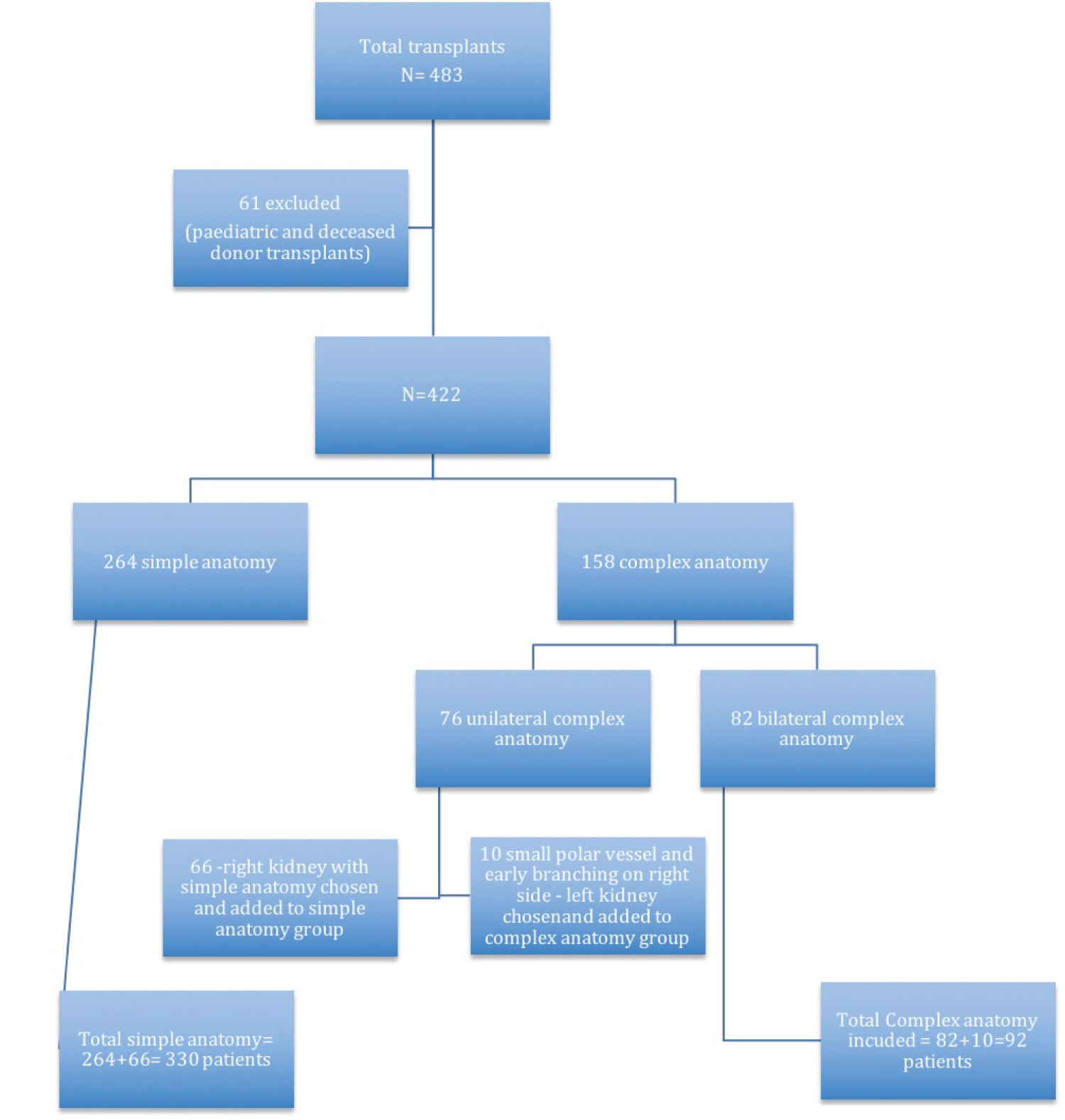 Figure 1: Study design.
View Figure 1
Figure 1: Study design.
View Figure 1
Table 2: Demographic profile of donors and recipients. View Table 2
Most of the patients with complex vascular anatomy had a main renal artery and an accessory renal artery (58.6%) and the remaining had either an upper or lower polar artery. Double renal vein was seen in 17 patients.
Different methods of renal artery reconstruction were used depending upon the graft arterial anatomy (Figure 2, Figure 3 and Figure 4). There were 62 (67.4%) grafts with double artery. Bench surgery was done in 8 cases (8.6%) in which ex vivo pantaloon (side to side) anastomosis was done to create a common lumen and the arterial anastomosis was done with the external iliac artery. Upper polar artery was ligated in 8 (8.7%) grafts, as the size of the artery was < 3 mm with < 10% area of perfusion. In the remaining patients both arteries were separately anastomosed to the external iliac artery in an end to side fashion.
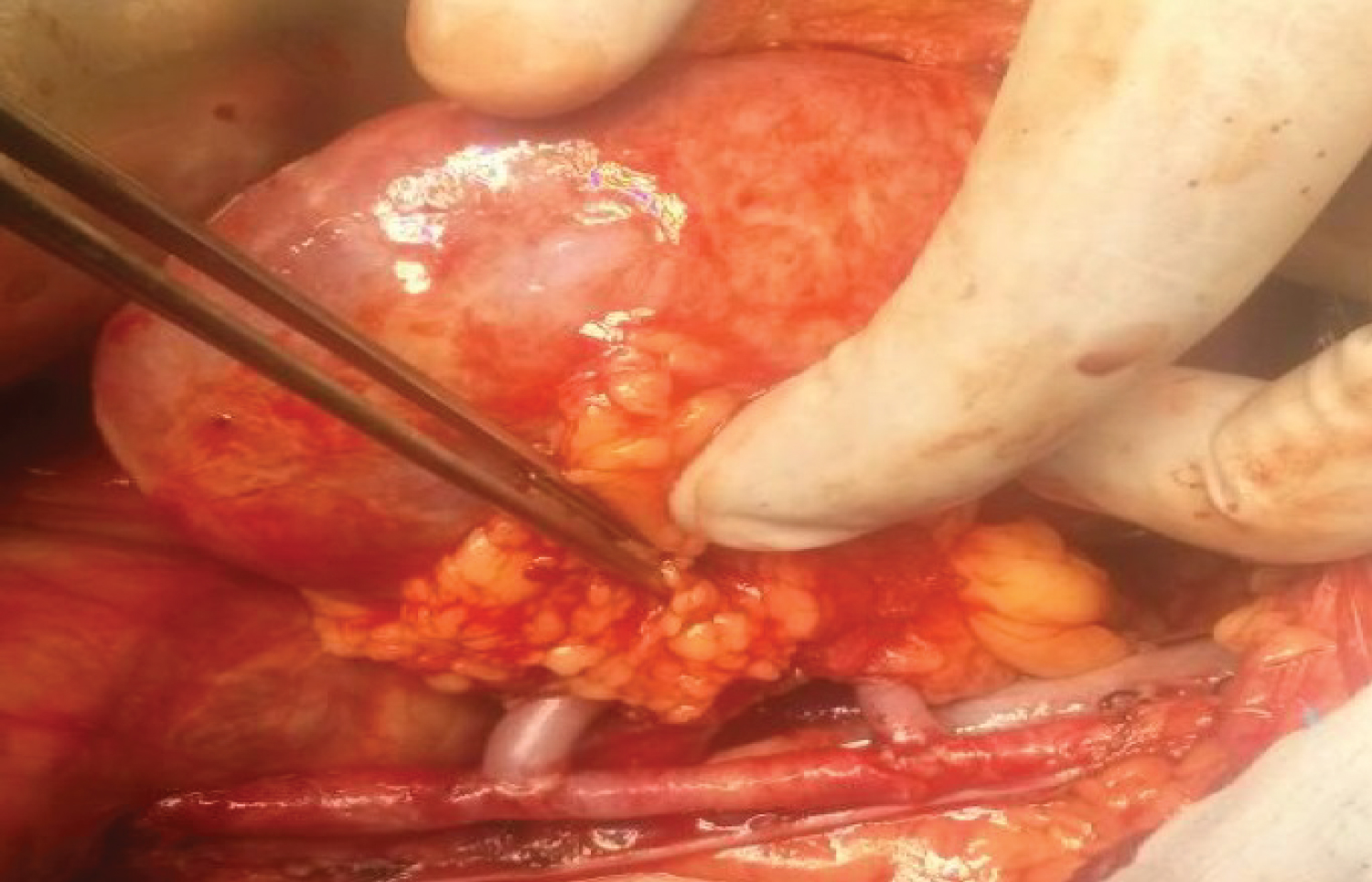 Figure 2: Showing the anastomosis between Accessory/Main Renal Arteries with External Iliac Artery.
View Figure 2
Figure 2: Showing the anastomosis between Accessory/Main Renal Arteries with External Iliac Artery.
View Figure 2
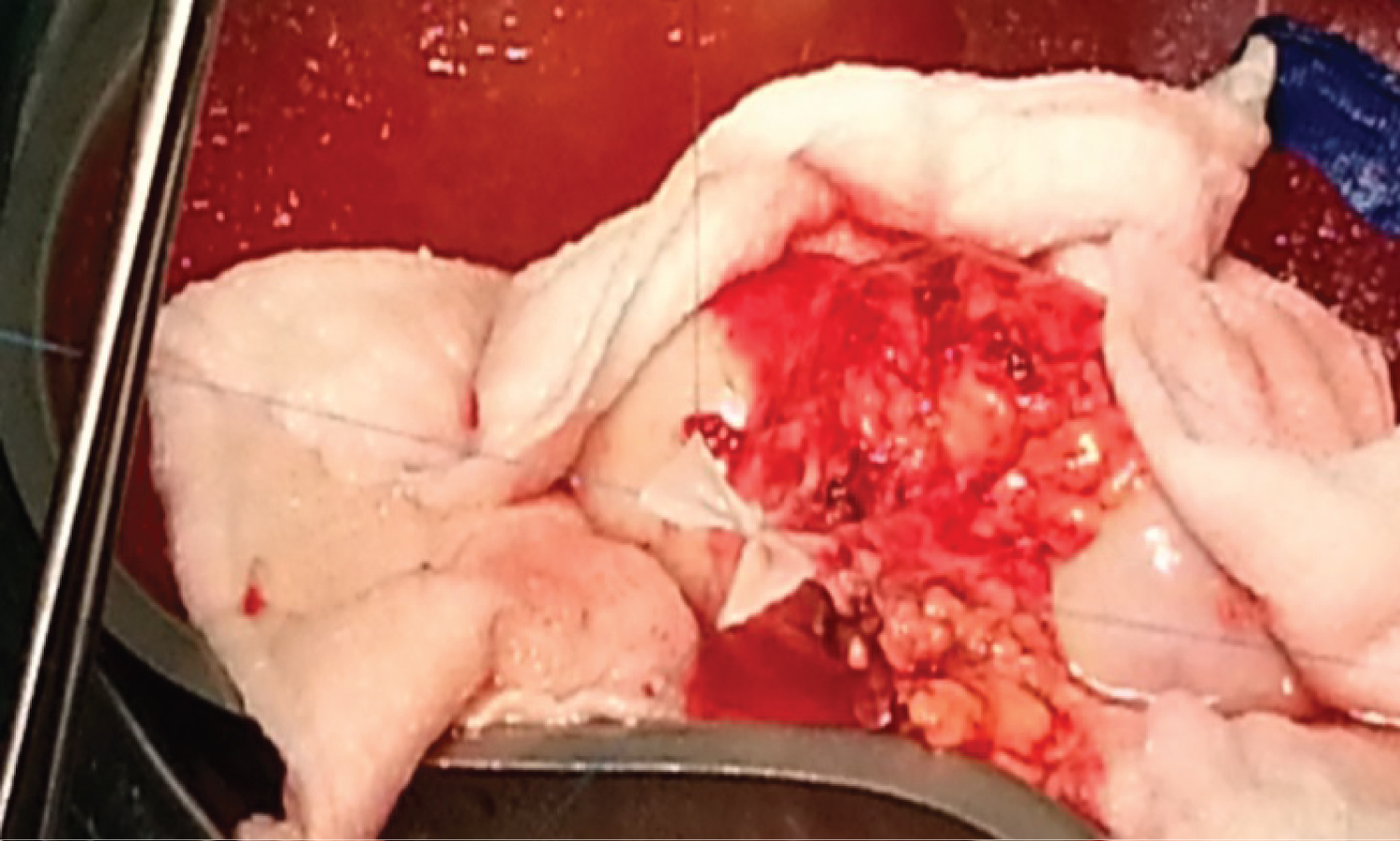 Figure 3: Showing the ex vivo side to side anastomosis of MRA with Accessory Renal Artery (Pantaloon).
View Figure 3
Figure 3: Showing the ex vivo side to side anastomosis of MRA with Accessory Renal Artery (Pantaloon).
View Figure 3
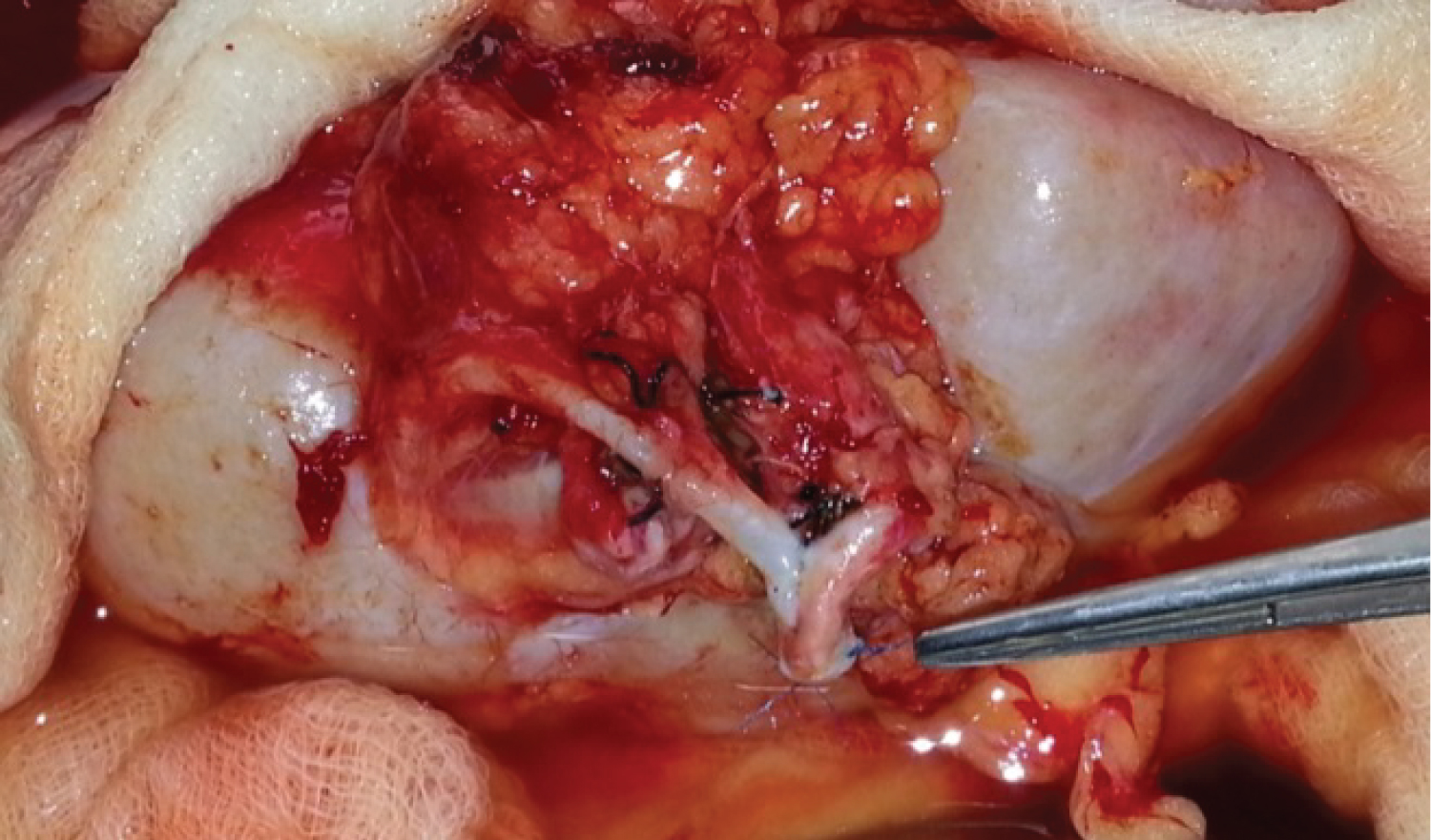 Figure 4: Showing the side-to-side anastomosis of MRA with Accessory Renal Artery (Pantaloon) with EIA.
View Figure 4
Figure 4: Showing the side-to-side anastomosis of MRA with Accessory Renal Artery (Pantaloon) with EIA.
View Figure 4
There were 30 (32.6%) grafts with triple renal artery. In three patients two hilar vessels were joined using the pantaloon technique and were anastomosed with the internal iliac artery in an end-to-end anastomosis. The third artery was anastomosed separately with the external iliac artery. In 10 (10.7%) patients with triple renal artery, the main renal artery and lower polar artery were anastomosed to the external iliac artery and the upper polar artery to the internal iliac artery. In 15 (16.3%) patients with upper polar artery, the size of the artery was < 3 mm with < 10% area of perfusion, which was ligated on bench dissection. In 2 (2.2%) patients the main and the accessory renal arteries were anastomosed separately with the external iliac artery in an end to side manner (Table 1). In the 17 (18.5%) donors who had double renal veins, smaller caliber vein was ligated and single venous anastomosis to external iliac vein was performed (Figure 2, Figure 3 and Figure 4).
Warm ischemia time-I & Rewarming time and mean operative time were more in complex renal anatomy transplants (Table 3). There were no major intraoperative complications in both the donors as well as recipients. All except seven graft kidneys (2 in complex anatomy group and 5 in simple anatomy group) showed immediate urine production following declamping. There was no difference in the urine output and fall in serum creatinine levels in the postoperative period in both the groups (Table 4).
Table 3: Warm ischemia time, cold ischemia time and operating time. View Table 3
Table 4: Short term outcome of graft kidney and patient survival. View Table 4
Post-operative morbidity (Table 4) in terms of delayed graft function (4.2% vs. 4.3%), re-exploration rate (4.3% vs. 4.5%) and primary graft non function (1% vs. 0.6%) were comparable between the two groups. One patient required graft nephrectomy due to spontaneous graft rupture in simple anatomy group where hemostasis could not be achieved. One patient developed hyper acute graft rejection, which was confirmed on histology and required graft nephrectomy in complex anatomy group.
Arterial thrombosis (Table 4) was seen in 2 (2.1%) patients in complex anatomy group and 6 (1.8%) patients in simple anatomy group. Both the patients in complex anatomy group had on table thrombosis in the accessory/polar vessel. In the simple anatomy group five out of six patients with arterial thrombosis required graft nephrectomy and only one graft could be salvaged after reperfusion and redo anastomosis. On routine DTPA performed on POD5, 5 patients in the complex anatomy group showed non-perfusion in the area supplied by the smaller caliber vessels. All the grafts were salvaged in these patients albeit with a higher baseline serum creatinine levels at the time of discharge from the hospital.
Ureteric complication (Table 4) in form of urine leak was seen in 2.1% patients in the complex anatomy group compared to 3% of patients in simple anatomy group. The urine leak was diagnosed by drain fluid creatinine more than twice that of serum value or DTPA evidence of leak and confirmed by diagnostic per cutaneous nephrostogram or CT urogram. All patients were managed by Foley's catheterization, pigtail drainage of collection, image guided antegrade stenting and two patients from the simple vascular anatomy group required Boari flap reconstruction. The stents were removed after 6 weeks and none of these patients developed long term stricture. There were in total 4 mortalities, one (1%) in complex anatomy group and three (0.9%) in simple anatomy group patients. However, none of the mortality was related to graft vascular anatomy. Two patients died because of dengue hemorrhagic shock syndrome, one died secondary to fungal pneumonia with sepsis and one patient with delayed graft function had massive intracranial bleed due to hypertensive crisis while on dialysis (Table 4).
The hospital stay was comparable between the two groups (13.5 days vs. 14.2 days, p value = 0.31) (Table 4).
Kidney transplantation is the treatment of choice for patients with end-stage renal disease. During the last three decades, graft and patient outcomes have significantly improved because of standardization in surgical technique and improvements in immunosuppressive therapy, organ preservation, and antimicrobial therapy. Live kidney donation has become increasingly important over the years and accounts for more than 95% of kidney transplantation in India [2]. In India the deceased donation program is still in infancy because of various social, cultural and religious reasons. Due to the persistent donor organ shortage and increasing incidence of end-stage renal disease [1], there is a worldwide trend in accepting so called extended criteria live kidney donors (i.e. obese donors, older donors, donors with hypertension). Vascular multiplicity in live kidney donors is considered another extended criterion, because of the premise that it is associated with higher (surgical) complication rates in the recipient. According to current evidence and guidelines, single renal vascular anatomy is preferred in living kidney donors, although arterial or venous multiplicity should not be considered as an absolute contra-indication for live kidney donation [10-17]. Multiple renal arteries are found in 18% to 30% of all potential kidney donors. Although the use of these grafts has been associated with an increased incidence of vascular and urologic complications, their long-term outcome has not been studied well especially in live donation. The impact of accepting this graft is the increase of the number of graft suitable for transplantation, reducing the waiting list. We retrospectively reviewed our experience with multiple renal arteries and compared both short- and long-term outcomes of kidney grafts single renal arteries.
Computed tomography angiography is an effective method for evaluating the renovascular morphology of the donor [18]. Grafts with anatomic variations, such as double ureters and multiple renal vessels pose a challenge to the transplant surgeons in both living as well as deceased donor transplant as these vessels are of smaller diameter and anastomosing them is difficult and time consuming [19-21] which in turn leads to longer warm ischemia time and higher incidence of delayed graft function. In a systemic review and meta-analysis [10], the mean warm ischemia time-I & Rewarming time was more in multiple renal artery group as compared to single renal artery group and there was higher incidence of delayed graft function. It seems obvious that performing multiple vascular anastomoses entails an additional time, which is likely to prolong the rewarming time. Although the warm ischemia time was more in our study also in patients with multiple renal arteries but the incidence of delayed graft function was not higher in our study. Similar observation was reported by Vazquez, et al. [11] where warm ischemia time-I & Rewarming time and mean operative time was more but the graft outcome in patients with multiple renal artery was not affected.
Various methods of reconstruction of multiple arteries have been reported in the literature. It has been shown in literature that the method of reconstruction does not affect the graft outcomes. The technique of reconstruction should be based on the size and origin of the vessels of the graft kidney as well as the status of the vessels of the recipient. Upper pole arteries less than 3 mm can be safely ligated. It has been shown that it does not affect the graft outcomes and even if anastomosis of such small vessels is done, generally it leads on to the thrombosis. Ashraf, et al. [12] reviewed the records of 105 live related kidney transplant, in which 33 grafts had multiple renal arteries. Patients were subdivided in to 3 groups according to the vascular reconstruction technique: Group A (n = 72) (68.6%) comprised recipients with single-renal artery allografts anastomosed end-to-side with external or end-to-end with internal iliac artery in a single anastomosis pattern, group B (n = 6) (5.7%) comprised recipients with multiple renal-artery allografts anastomosed end-to-end with internal iliac or end-to-side to external iliac artery with single anastomosis (multiple renal arteries were converted to single artery after bench reconstruction), and group C (n = 27) (25.7%) comprised recipients of multiple renal artery allografts, implanted with multiple arterial anastomosis. He, et al. [13] in his retrospective study found that variety of surgical reconstruction techniques can be used including ligation of upper pole arteries < 3 mm in diameter with equal patient and graft survival. Davari, et al. [14] in his study also concluded that graft survival was excellent with sequential multiple anastomoses with multiple arteries converted to a single artery by bench reconstruction. In our study, we used similar combinations of arterial anastomosis between renal graft arteries and recipient vessels in complex anatomy group as described previously. However, in majority of cases anastomosis was done with external iliac artery and in few cases upper polar artery was ligated (< 2 mm size). In eight cases, on bench arterial reconstruction was done to make a common conduit for anastomosis (Ex vivo side to side, pantaloon reconstruction). Thus, the selection of the technique arterial reconstruction should be individualized based on the caliber and length of multiple renal arteries.
Generally single venous anastomoses is preferred for the graft because unlike arteries, the venous drainage of multiple renal veins is interconnected and ligation of smaller veins has not been shown to be associated with higher incidence of venous thrombosis or other complication [12]. Lim, et al. [15] in his retrospective study showed no complications during intraoperative period in both the donor and recipient groups with multiple renal veins. In our study all patients underwent single venous anastomosis between graft renal vein and recipient external iliac vein. Those patients where renal grafts had double veins underwent single venous anastomosis after ligating the smaller caliber vein. No venous anastomoses related intraoperative or post-operative complications were seen in the recipient group.
Recipient graft outcomes in terms of mean creatinine level are an important measure of the success of a technique of transplantation. Ashraf, et al. [12] in his retrospective study showed that mean creatinine level at 1 month after renal transplantation were 1.21 and 1.12 for grafts with multiple renal artery and single renal artery respectively (p = 0.215). Khanna, et al. [16] in their retrospective study also noted no significant difference in graft function and patient survival at 1, 3 and 5 years following renal transplant between single renal artery group and multiple renal artery group. The present study has also shown similar result and the primary graft outcomes in terms of urine output, fall in serum creatinine and delayed graft function was comparable in both the groups.
One of the important complications associated with graft with arterial multiplicity is a higher incidence of ureteric complication and arterial thrombosis due to smaller caliber of vessels resulting in graft loss. In 2008, Kok, et al. [22] investigated the live kidney donor cohort in a single center from 2001 until 2005 regarding vascular multiplicity and reported, an increased incidence of urological complications after renal transplant in donors with complex arterial anatomy. Osman, et al. [17] in a study of twelve hundred live renal transplant patients has evaluated the incidence of vascular and urological complications in detail. There were 34 (2.8%) vascular and urological complications in their study and thrombotic complication (p = 0.04) was significantly associated with multiple renal arteries. Roza, et al. [21] has also reported a higher incidence of graft loss of 4.7% in their study of complex anatomy renal transplants. In our study, the incidence of urological, vascular complication and graft loss were similar in both the groups.
The major limitation of the study is that it is a retrospective study with only short-term outcome evaluation.
The extended criteria live donors (older donors, donors with hypertension) are accepted world over to overcome the shortage of organ donors. Kidney donors with complex vascular anatomy have shown to be associated with higher complication rate but with refinements and advances in surgical techniques, vascular and intra-operative complications related to multiple renal vessels can be minimized and excellent graft and patient survival can be achieved. To conclude, grafts with multiple renal vessels have equivalent outcomes as compared to simple vascular anatomy but should be done in high volume centers and by experienced surgeons.
None of the authors have any financial disclosure and no conflict of interest. This study has not been published elsewhere.