Kidney transplant recipients (KTR) and liver transplant recipients (LTR) are at risk for elevation in serum potassium post-transplant. Patiromer is approved by the Food and Drug Administration for the treatment of hyperkalemia, but there is limited safety and efficacy data for its use in solid organ transplantation.
We retrospectively reviewed adult KTR and/or LTR transplanted between October 2015 and September 2019 treated with patiromer.
17 KTR, 13 LTR, and 2 simultaneous liver-kidney recipients received patiromer post-transplant. Mean serum potassium (mEq/L) was significantly reduced at approximately 1 week and 1 month after starting therapy (5.5 ± 0.4, 4.9 ± 0.5, 5.1 ± 0.5; p < 0.001). Side effects included diarrhea in two patients and nausea in 1 patient. Mean tacrolimus level (ng/ml) at 1 week post-patiromer initiation (8 ± 3.1 vs. 7.8 ± 2.8, p = 0.7) and 1 week post-discontinuation of therapy (7.9 ± 3 vs. 9.2 ± 3.3, p = 0.3) were not significantly different.
Patiromer is an effective medication for hyperkalemia in post-abdominal transplant recipients, with a favorable side-effect profile. Additional monitoring of tacrolimus levels may be warranted for patients taking patiromer due its potential to affect absorption of tacrolimus.
Kidney transplant, Liver transplant, Patiromer
Hyperkalemia is a common complication post-abdominal transplant with a reported incidence of 22-44% post-kidney transplant and nearly 20% post-liver transplant [1,2]. Post-transplant renal dysfunction and use of medications that decrease renal potassium clearance (e.g., calcineurin-inhibitors, sulfamethoxazole-trimethoprim, angiotensin-converting enzyme inhibitors) contribute to the higher risk of hyperkalemia [3]. Patiromer is a medication approved by the Food and Drug Administration for the treatment of hyperkalemia in adults. It is a non-absorbed cation polymer that exchanges calcium for potassium in the lumen of the gastrointestinal tract, which increases fecal excretion of potassium [4]. Patiromer has been described as a viable alternative to other agents for the treatment of hyperkalemia such as sodium polystyrene sulfonate, with perhaps a more tolerable side effect profile. In clinical trials, patiromer was shown to effectively and safely lower serum potassium levels [5,6]. However, transplant patients or others receiving calcineurin inhibitors, were excluded from these studies. As a consequence, the role of patiromer in the context of transplantation is not well understood.
Data on use of patiromer in kidney transplant recipients (KTR) is limited to one case report with two patients and one small case series [7,8]. No prior studies have evaluated the safety and efficacy of patiromer in liver transplant recipients (LTR). This study aims to review the use of patiromer in LTR and expand on the limited data in KTRs. Our objective was to provide a comprehensive description of the efficacy and safety of patiromer in abdominal transplant recipients, and describe its potential impact on post-transplant immunosuppressant dosages and levels.
We retrospectively analyzed the electronic medical records of adult kidney and/or liver transplant recipients age ≥ 18-years-old transplanted between October 1, 2015 and September 1, 2019 prescribed patiromer at any time after transplant. The primary outcome was resolution of hyperkalemia post-initiation of patiromer. Secondary outcomes included incidence of adverse effects (nausea, diarrhea, constipation, hypomagnesemia, hypercalcemia), rejection and mortality. Tacrolimus levels and adjustments to tacrolimus dosages were also analyzed. The lab value drawn closest to each specified time point was recorded. The University of Nebraska Medical Center Institutional Review Board approved this study. The requirement of individual consent was waived.
Baseline characteristics, laboratory measurements, medication dosages, and clinical outcomes were summarized using descriptive statistics, including mean ± standard deviation (SD) and median (range). Dependent sample t-test was performed for normally distributed continuous variables and presented as mean ± SD. Statistical significance was defined at a p-value of < 0.05.
Thirty-two subjects were initiated on patiromer in the inpatient or outpatient setting. Seventeen patients (53%) were KTR, 13 (41%) were LTR, and 2 (6%) were simultaneous liver-kidney (SLK) transplant recipients. Demographics of these patients are described in Table 1. The most common indication for transplant was diabetic kidney disease in KTR (32%) and non-alcoholic steatohepatitis in LTR (27%). Three LTR and 2 KTR developed biopsy proven acute rejection post-transplant. A total of 20 patients received hemodialysis post-transplant: 10 KTR, 9 LTR, and 1 SLK transplant recipient. Sixteen of these patients had stopped hemodialysis prior to patiromer initiation, while four were started on hemodialysis at the time of patiromer discontinuation. At the time of patiromer initiation, 28 patients were taking calcineurin inhibitors, 14 were taking sulfamethoxazole-trimethoprim for pneumocystis prophylaxis, 2 were taking lisinopril for blood pressure control, and one patient was taking sacubitril-valsartan due to heart failure.
Table 1: Patient demographics. View Table 1
Patiromer was initiated at a median of 257 days (5-980) post-transplant and the majority of patients were initiated at 8.4g daily. Six patients required an increase in dosage to 16.8g daily, and one patient required further dose escalation to 25.2g daily. Patiromer was eventually discontinued in 21 patients (64%). Seventeen of these patients stopped patiromer due to improvement in renal function and resolution of hyperkalemia, 4 were initiated on dialysis, and 1 stopped therapy due to diarrhea. One patient developed diarrhea, and another reported nausea, but neither required discontinuation of therapy. Median duration of therapy was 87 days (1-835). 12 patients (38%) were still taking patiromer at the time of this analysis.
Mean serum potassium levels over the course of patiromer therapy are shown in Figure 1. Potassium (mEq/L) significantly decreased from time of initiation of patiromer compared with 1 week and 1 month later, and at discontinuation of therapy (5.5 ± 0.4, 4.9 ± 0.5, 5.1 ± 0.5, 4.4 ± 0.5, p = 0.001). Serum creatinine (μmol/L) was not significantly different prior to patiromer initiation compared to 1 week later or at discontinuation of patiromer (2.1 ± 0.2, 1.8 ± 1.4, 1.8 ± 0.9; p = 0.3, p = 0.7). Serum magnesium (mEq/L) (1.8 ± 0.35, 1.7 ± 0.2, 1.7 ± 0.2; p = 0.2) and calcium (mEq/L) (9.3 ± 0.65, 9.4 ± 0.7, 9.3 ± 0.8; p = 0.7) were similarly unchanged before, during and after patiromer therapy.
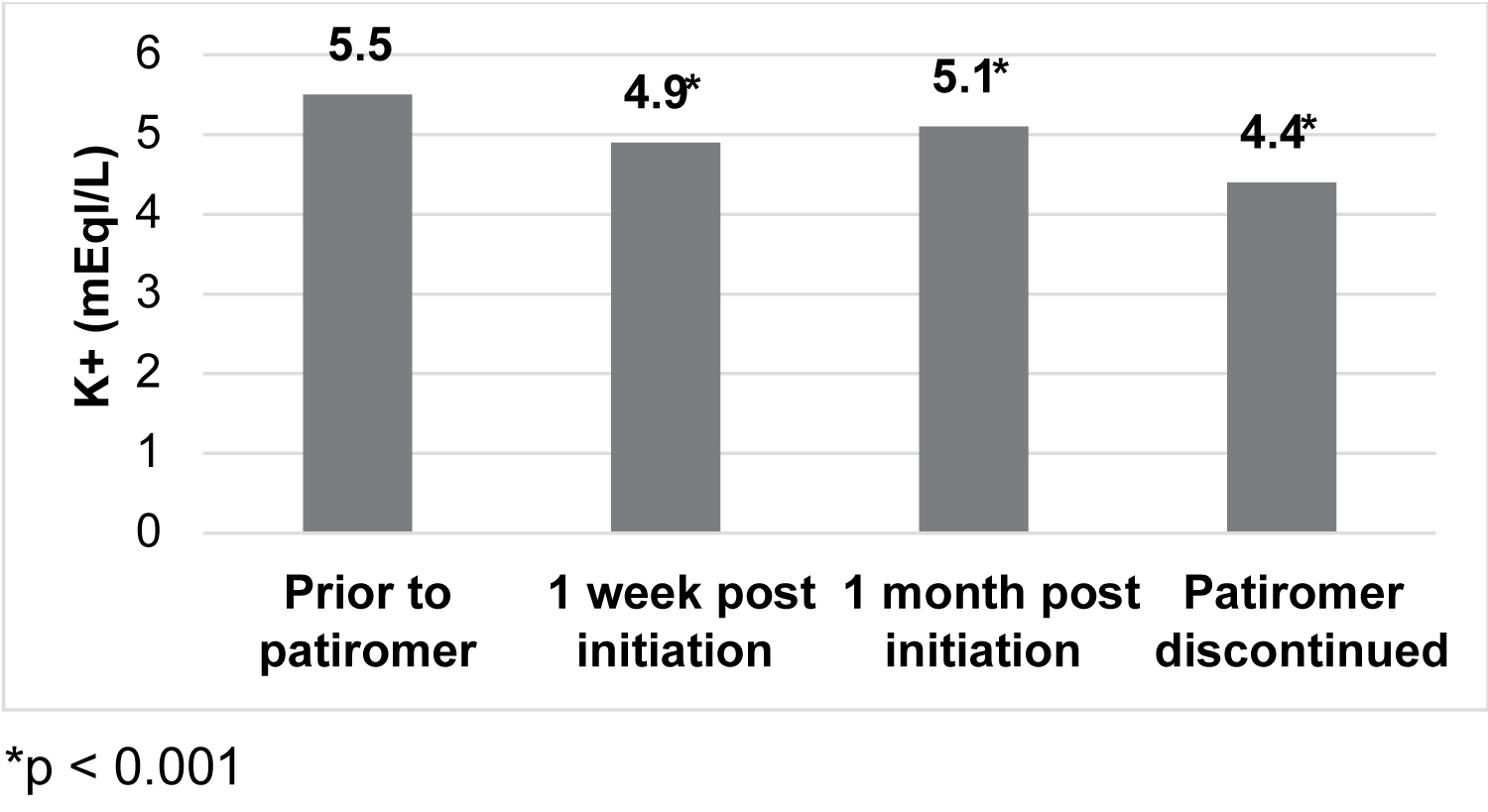 Figure 1: Mean serum potassium pre and post-patiromer initiation.
View Figure 1
Figure 1: Mean serum potassium pre and post-patiromer initiation.
View Figure 1
Mean tacrolimus dosage per day and 12-hour trough level prior to initiation of patiromer, 1 week post-patiromer initiation, at time of patiromer discontinuation, and 1 week post-discontinuation of therapy is presented in Figure 2. There was no significant difference in level or dosage over the course of patiromer therapy. No biopsy proven rejections occurred post initiation of patiromer. Two LTR and one SLK transplant recipient died, all greater than one year post-transplant and none while on patiromer.
 Figure 2: Mean tacrolimus dosage per day and 12-hour trough level pre and post-patiromer initiation and discontinuation.
View Figure 2
Figure 2: Mean tacrolimus dosage per day and 12-hour trough level pre and post-patiromer initiation and discontinuation.
View Figure 2
This is a retrospective study evaluating 32 kidney and/or liver transplant recipients who received patiromer for the treatment of hyperkalemia. It is the first study to evaluate the use of patiromer in liver and SLK transplant recipients, and adds to the limited data available in KTR. Patiromer appears to be an effective therapeutic option in this patient population, as demonstrated by the significantly reduced serum potassium at one week and one month post initiation of therapy. The drug was well tolerated with gastrointestinal side effects reported in only 9% of patients, and there was no significant effect on serum magnesium or calcium levels.
The package labeling for patiromer recommends other medications be taken at least 3 hours before or 3 hours after patiromer due to its potential to bind medications and limit their absorption [4]. Timing of medications is an important point of education for the transplant population to ensure the administration of patiromer does not negatively impact other medications, especially their immunosuppression. Although not statistically significant, in patient's taking tacrolimus we observed a decrease in their mean 12-hour trough level one week after initiation of therapy, despite an increased mean dosage per day. After patiromer discontinuation, mean tacrolimus level trended non-significantly higher without any change in the mean tacrolimus dosage. While many factors can influence immunosuppression levels, this trend is notable, suggesting the need for increased monitoring when starting, adjusting, or stopping patiromer. It also highlights the need to educate patients on proper timing of medications. At our institution, we recommend patients take patiromer with lunch to minimize potential interaction, though midday medication timing may impact adherence.
This study has limitations inherent to its retrospective nature. Data collected depended on the accuracy of the documentation in the electronic medical records. Medication reconciliation records at clinic visits were used to document patiromer and immunosuppression doses, and medication administration records were used to document administration of these medications during inpatient admissions. Due to the small cohort, conclusions could not be made regarding the effects of patiromer exposure on rejection and mortality.
In conclusion, patiromer appears to be a safe and efficacious medication for the treatment of hyperkalemia in KTR and LTR. This is the largest study to date in solid organ transplant recipients, and the first in LTR and SLK transplant recipients. We noted minimal GI intolerance and little effect on other electrolytes, making it an appealing option for post-transplant hyperkalemia. We recommend enhanced monitoring of immunosuppression after initiation, titration, or discontinuation of patiromer.
The authors have no conflicts of interest to disclose.
A.S. and C.M. designed the study, A.S. collected the data. A.S., E.L., S.W., C.M. contributed to the analysis of the results and writing of the manuscript.