Introduction: Hepatitis E virus (HEV) is one of the most common causes of acute hepatitis in endemic areas, yet least diagnosed aetiologies of acute viral hepatitis.
Objective: Highlights the importance of early consideration of HEV in all patients with acute hepatitis who have originated from endemic regions.
Methods: The clinical course of a patient admitted for acute fulminant hepatitis complicated by multiorgan failure is described.
Results: Demonstrated improvement in his clinical status after hepatic encephalopathy medications, haemodialysis for acute oliguric kidney injury and total plasma exchange (TPE) for bilirubin and protein bound toxin removal.
Conclusion: The identification of this unusual extrahepatic complication of Hepatitis E virus is important and might consider TPE early for acute cholestatic fulminant hepatitis.
The World Health Organization estimates that HEV causes 20 million new infections annually, with more than 3 million cases of acute hepatitis and over 55,000 deaths [1]. The prevalence rates of hepatitis E antibody are higher in resource-limited countries as compared with developed countries (10 to 70 versus 1 to 21 percent) [2]. A small proportion (0.5 to 4 percent) of HEV-infected persons develop acute hepatic failure [3]. Acute hepatic failure carries a high mortality, resulting in an overall case fatality rate of 0.5 to 3 percent [4]. In general, cholestatic hepatitis resolves spontaneously within weeks to months with no sequelae [5]. Recovery is marked by viral clearance, an increase in IgG anti-HEV titres, and a decrease in IgM anti-HEV levels [6]. The coexistence of Hepatitis E infection in G6PD patients can result in severe haemolysis, renal failure, and other complications such as fulminant hepatitis. It may consequence in morbidity and mortality, if critical attention to the patient is not provided immediately [7].
34-years-old male patient presented with agitation, history of low grade fever, dark coloured urine with low volume, jaundice and right sided upper abdominal pain for last 2-3 days. He is alcoholic, no significant medical history, no family history of liver disease nor significant sexual or drug use history. On general physical examination, blood pressure of 160/70 mmHg, heart rate 120/min, respiratory rate 17/min and temperature of 37 ℃. The patient was delirious, dehydrated, jaundiced. Neurological examination revealed asterixis, exaggerated deep tendon reflexes, and bilateral withdrawal planter reflex. On abdominal examination there was tenderness in right hypochondrium and epigastric region, no signs of chronic liver disease. Haematology revealed haemoglobin 6.6 gm/dl, WBC 21000/cmm (neutrophils 65%, lymphocyte 20%, eosinophil 0.2%), platelets 258,000 ml. Biochemistry panel revealed total serum bilirubin of 879 u mol/l with a direct bilirubin of 592 u mol/l mg/dl. The alanine aminotransferase (ALT) was 4059 U/L, serum aspartate aminotransferase (AST) was 16447U/L, alkaline phosphatase was 207 U/L, lipase 364 IU/L and creatinine 144 umol/l (Figure 1). A urinalysis was unremarkable with the exception of protein and presence of bile. He was admitted in critical area, intubated for air way protection and mechanical ventilated. A liver ultrasound revealed no biliary tree dilatation and patent hepatic vasculature. Presumptive diagnosis alcohol induced liver injury with the differential diagnoses including acute viral hepatitis, autoimmune liver disease, toxic hepatitis. He was treated supportively with intravenous hydration, ursodeoxycholic acid, cholestyramine, lactulose enema, Rifaximin and N-acetylcysteine and methylprednisolone 100 mg for possible drug induced liver injury. On 3rd day of admission the patient's renal function continued to progressively deteriorate with increasing azotemia and oliguria, serum creatinine escalated upto 500 umol/l and urea was 16 mmo/l, haemoglobin 5 gm/dl, indirect and direct Coombs test were negative., lactate dehydrogenase was 3755 U/L and serum ferritin was 8000 ng/ml (Figure 1). On peripheral blood smear absolute neutrophilia with cytoplasmic toxic granulations, schistocytes. Reticulocyte 10% and serum haptoglobin < 0.1 g/L indicate evidence of intravascular haemolysis. Malaria Parasite test, Leptospira serology and paracetamol level negative. A hepatitis screen was conducted, and negative results included hepatitis A, B and C, human immunodeficiency virus, Epstein-Barr virus, cytomegalovirus. Iron studies, and ceruloplasmin normal. Serological markers for autoimmune causes of hepatitis negative, G6PD normal 5.99 units/gHb but in patient with acute haemolysis and reticulocytosis G6PD may appear falsely normal. With all above examination and workup the diagnosis of fulminant hepatitis with possibility of G6PD deficiency was determined, haemolytic anaemia, rhabdomyolysis as CPK was high, bilirubin nephropathy complicated by acute kidney injury. In the presence of oliguria; volume overload and azotemia, haemodialysis was started on continuous veno-venus hemodiafiltration, effluent dose > 30 ml/kg per hour, predilution replacement fluid 2000 ml per hour, dialysate fluid 2000 ml per hour, ultrafiltration 1-2 ml/kg per hour, blood flow 150-200 ml/min. Dialysate solution composition; sodium 138 mmol/L, potassium 2 mmol/L, chloride 113 mEq/L, calcium 1.5 mmol/L, bicarbonate 35 mEq/l, glucose 5.55 mmol/L, osmolarity 293 mOsm/L, maintaining the filtration fraction below 25% to avoid filter clotting as no anticoagulation was given. On day 5 of hospital admission biochemistry panel revealed progressive rise in total serum bilirubin to 1090 umol/l with a direct bilirubin of 736 umol/l mg/dl, the patient was still in hepatic encephalopathy with high serum ammonia level 89 umol/L and so we started TPE for removal of inflammatory molecules, bilirubin and any possible protein bound toxin. TPE was performed daily against 10 units FFP and albumin was used as replacement fluid. Treatment duration was about 2.5 hours and TPE was performed on 3 consecutive days until clinical recovery of hepatic encephalopathy. On day 7 of hospital admission, the patient improved clinically and extubated, his laboratory parameters were improving in comparison with admission time (total serum bilirubin declined from 1090 umol/l to 519 umol/l, ALT from 4095 U/L to 200 U/L and AST from 1644 U/L to 122 U/L. Due to the evidence of intravascular haemolysis and acute hepatitis repeated serum ceruloplasmin was sent and slit lamp examination to exclude Wilson disease was done and hepatitis E virus antibodies screen was pending as the patient was coming from endemic area. The follow up of serum lipase was rising to 2299 IU/L suggesting acute pancreatitis, so contrast-enhanced computed tomography of the abdomen showed mild edematous pancreatitis. Over the course of 2 weeks hospital admission, the patient's metabolic parameters gradually improved total bilirubin declined to 304 umol/l, AST was 157 U/L, ALT was 154 U/L, lipase 429 IU/L and reticulocyte count fell to 0.8% (Figure 1). He was maintained on regular haemodialysis due to persistent oliguria, the HEV serology IgM came positive on day 12 of hospital admission, given his improving LFTs and general condition, he was discharged home on day 14 of hospital admission to continue haemodialysis.
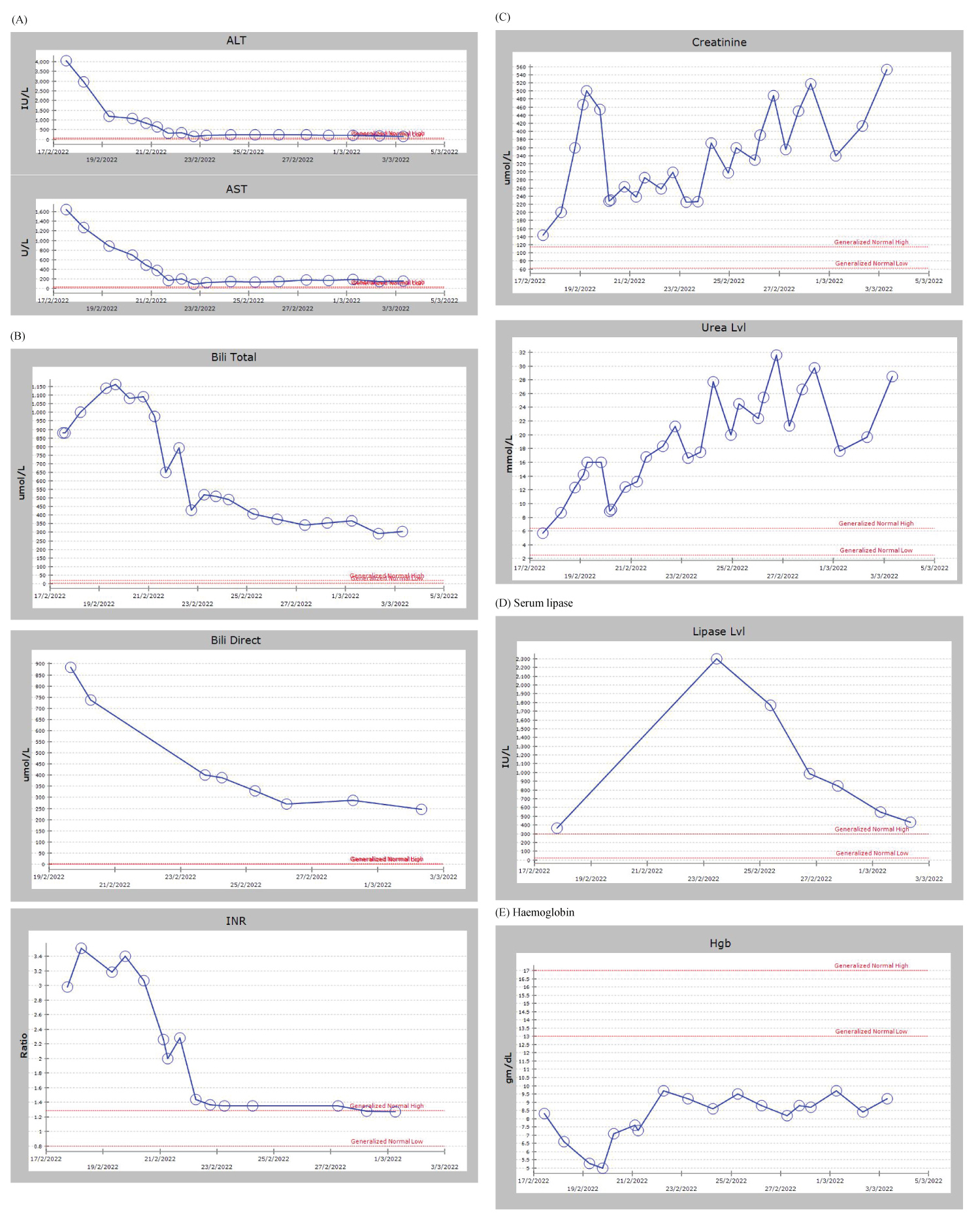 Figure 1: Graphical representation of biochemical trends over 14-day admission showing (A) ALT, AST, (B) bilirubin (total & direct), (C) Serum creatinine & Urea, (D) Serum lipase, (E) Haemoglobin INR.
Figure 1: Graphical representation of biochemical trends over 14-day admission showing (A) ALT, AST, (B) bilirubin (total & direct), (C) Serum creatinine & Urea, (D) Serum lipase, (E) Haemoglobin INR.
ALT: Alanine Aminotransferase; AST: Aspartate aminotransferase; INR: International Normalized Ratio.
View Figure 1
Studies have found the main cause of liver tissue injury in acute hepatitis E patients may be the host immunity [8]. Under normal conditions, the symptoms of acute hepatitis are mainly manifested by the rapid increase of serum liver enzymes as well as jaundice similar to our case who showed features of sever fulminant liver cell failure. In a previous study on HEV epidemic they found that among 230 patients 24 (10.4%) had developed acute liver failure (ALF). Four (1.8%) among them died due to multiorgan failure with acute kidney injury, and all were pregnant [9]. Our case showed hepatitis E virus IgE positive and IgG > 25 IU/ml results consistent with acute hepatitis E virus. This case highlights the importance of early consideration of acute HEV especially in endemic regions. Case reports have shown improved LFTs after corticosteroid administration in patients with acute liver failure from HEV infection [10]. Our patient was started on corticosteroid therapy because a as there is some evidence to suggest that steroid therapy improves prognosis in patients with ALF [11]. However, the steroid trial was ineffective in improving our patient's liver function and clinical status. N-acetyl cysteine was also administered to our patient because it may improve transplant-free survival in patients with ALF with low grade encephalopathy [12]. In patients suspected with viral hepatitis along with unexplained anaemia, intravascular haemolysis, should suspect either acute viral hepatitis induced haemolysis or Wilson disease, repeated serum ceruloplasmin was 0.140 g/l borderline and normal slit lamp examination, giving low possibility of Wilson disease. Acute Viral Hepatitis induced haemolysis is usually mild, rarely there is a drop in haemoglobin level more then 1-2g/dl [13]. However, the presence of hepatitis E in G6PD deficient patient can lead to severe haemolytic anaemia, could be the cause of intravascular haemolysis in our case. Acute kidney injury in our case may be due to excess haematin and bilirubin may lead to obstruction of renal tubules, precipitating oliguric acute renal failure. Diagnosis of G6PD deficiency can be a challenge because G6PD levels can be normal throughout the course of illness and immediately after an event of haemolysis as in our patient. False normal values of G6PD are due to the fact that, old RBCs which are deficient in G6PD are haemolyzed earlier while the new RBCS have normal G6PD content. It is advisable to repeat G6PD levels 8-10 weeks after the haemolytic event [14]. Our patient had TPE for 3 consecutive session to improve systemic inflammation and hepatic encephalopathy. Several studies have shown that the presence of a systemic inflammatory response syndrome is associated with a worsening of hepatic encephalopathy and a particularly poor prognosis in ALF [15,16]. Extracorporeal removal of excess circulating proinflammatory molecules might be a therapeutic option. In this context, therapeutic plasma exchange (TPE) has shown beneficial effects in some studies [17,18]. Various case series and uncontrolled studies have shown that TPE is safe in patients with ALF and can significantly reduce both the concentration of ammonia and the grade of hepatic encephalopathy as well as improvements in hemodynamic [17,18]. It combines two treatment principles in a single intervention: (a) replacement of the excretory and metabolic functions of the failing liver via supplementation of the lacking proteins and (b) removal of deleterious damage-associated molecular patterns and cytokines thus modulating the pathological overwhelming immune response that counts responsible for the associated multiorgan failure. Our case was started on continuous renal replacement therapy (RRT) that might be superior to intermittent RRT in ALF patients as shown by Cardoso, et al. [19] our patient had acute kidney injury either due to intravascular haemolysis, bile cast nephropathy or rhabdomyolysis. The pathophysiology of renal injury in haemolytic anaemia is due to excessive intravascular haemolysis and precipitates of haemoglobin dimer within the renal tubules due to its relative insolubility [20]. Haemoglobin is then reabsorbed along the proximal tubular by the megalin-cubulin receptor system and dissociates into heme and globin within the tubular cytoplasm. Increased intracellular concentrations of heme leads to cellular injury and damages organelles as mitochondrion [20]. Bile cast nephropathy could be another precipitating factor for acute kidney injury in our patient, it is a rare and poorly understood entity characterized by progressive renal insufficiency in the setting of elevated serum bile salts and hyperbilirubinemia. Elevated total bilirubin levels, typically greater than 20 mg/dL, are reported in cases of bile cast nephropathy [21]. This excess bilirubin in kidney tubules believed to cause oxidative damages tubular toxicity and injury. Elevated levels of bile salts have been demonstrated to have negative chronotropic and ionotropic effects, resulting in cardiovascular instability and decreased renal perfusion and pre-renal azotemia. This is further exacerbated by changes in endovascular reactivity believed to be due to the prevalence of endotoxemia, hypoalbuminemia, and nitric oxide-mediated mechanisms resulting in decreased peripheral vascular resistance and decreased renal blood flow, resulting in ischemia to the kidney [22]. Owing to the rarity of this condition, there are currently no accepted treatment guidelines. Dialysis and plasmapheresis may additionally be of utility via the filtration of inflammatory cytokines, bilirubin and bile acids [23]. These methods of blood and/or plasma filtration have served utility in patients with acute liver failure and acute on chronic liver failure via the filtration of inflammatory cytokines, bilirubin and bile acids, among other compounds such as amino acids and free fatty acids [23]. On discharge he was maintained on regular intermittent haemodialysis waiting for renal recovery.
Treatment with TPE in case of acute viral hepatitis E with life-threatening complications, might lead to improvement in hemodynamic and reduced organ dysfunction.
Informed consent was obtained from the patient.
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
The authors declare that they have no conflicts of interest.