Background: Chronic low-grade inflammation is suggested to play a key role in the age-related decline in muscle mass, strength and physical performance also known as sarcopenia. The aim of this study was to investigate the potential associations between systemic markers of low-grade inflammation (CRP, IL-6 and TNF-α) and muscle mass, muscle strength, and physical performance in a geriatric out-patient population.
Methods: Geriatric patients were recruited from an outpatient fall clinic at Rigshospitalet Glostrup (n = 95, age +65 yrs.). Appendicular Lean mass (ALM) and ALM normalised to height (ALM/h2) were assessed by Dual energy X-ray Absorptiometry (DXA), muscle strength was evaluated by handgrip strength (HGS) and leg extension power (LEP) and physical performance by gait speed (GS) and 30s sit-to-stand test (30s STS). Inflammatory markers (hsCRP, IL-6 and TNF-α) were analysed by ELISA (Multiplex) and plasma concentration of each inflammatory marker was categorized as low, medium, or high when compared to reference values from a group of healthy older individuals who participated in The Copenhagen Sarcopenia Study.
Results: High levels of inflammatory markers were associated with poorer HGS also after adjusting for age and BMI. Men with high levels of IL-6 had 22.9% lower HGS than men with low levels of the cytokine, whereas women with high levels of hsCRP and TNF-α had 19.2% and 22.5% lower HGS, respectively.
Conclusion: Male geriatric patients having high plasma concentrations of hsCRP and IL-6 and female geriatric patients with high plasma concentrations of hsCRP and TNF-α have poorer HGS compared to those with lower plasma concentrations. These finding indicate that low-grade inflammation is associated with sarcopenia and underlines the need for focus on the area.
Inflammation, Sarcopenia, Muscle mass, Muscle strength, Geriatric patients
GP: Geriatric Patients; HO: Healthy Older Individuals; CRP: C-Reactive Protein; IL-6: Interleukin 6; TNF-α: Tumour Necrosis Factor Alpha; ALM: Appendicular Lean Mass; HGS: Handgrip Strength; LEP: Leg Extension Power; GS: Gait Speed; STS: Sit-To-Stand Test
As life expectancies increase and the global elderly population is growing, more and more people are affected from the physical decline that comes with older age [1]. The progressive loss of muscle mass and function associated with aging, also known as sarcopenia, is clinically important because it has been shown to increase the risk of physical disability, nursing home admission and mortality in home-dwelling older adults [2]. Consequently, sarcopenia was recognized as a separate condition of muscle disease and included in the IDC-10 in 2016 [3]. Yet, a better understanding of the underlying pathophysiological mechanisms leading to sarcopenia is still needed, to enable focused interventions to help people who are affected by sarcopenia and even to delay onset of the condition.
The causes of sarcopenia are multifactorial and include physical inactivity, altered endocrine function, insulin resistance, nutritional deficiencies, and inflammation [4]. Considerable evidence is now available pointing to the importance of increased cytokine levels for the development of sarcopenia [5-9]. Elevated concentrations of especially C-reactive protein (CRP) and pro-inflammatory cytokines such as interleukin 6 (IL-6) and tumour necrosis factor alpha (TNFα) have been demonstrated with aging [10-12]. Apart from being closely involved with the acute-phase inflammatory response these proinflammatory cytokines, produced by a wide range of human cells including macrophages, myocytes, and adipocytes, have a catabolic effect on muscle tissue [13,14]. IL-6 injections have been shown to induce skeletal muscle atrophy in otherwise healthy rats [15] and significant research has indicated that TNF-α on its own and through its stimulation of ROS (reactive oxidative species) and increased activation of nuclear factor kappa B (NF-κB) plays a key role in the development of muscle atrophy [16]. Besides direct catabolic effects of inflammation on skeletal muscle, indirect mechanisms such as inhibition of the expression and activity of hormones like growth hormone and IGF-I also contribute to muscle wasting [17]. Recent observations suggest an inhibitory effect of CRP upon the protein synthesis pathways [18], and the combined effect of inflammation and the decreased anabolism caused by declining concentration of sex hormones with old age only increases the progression towards sarcopenia. Additionally, high levels of the inflammatory markers CRP, IL-6 and TNF-α on their own have shown to be strong predictors of disability and to be associated with risk of falling [19-21].
The aim of this study was to investigate the relationship between plasma concentrations of CRP, IL-6 and TNF-α and skeletal muscle mass, strength, and physical performance in a group of patients from a Danish geriatric outpatient fall clinic.
This is a cross-sectional study conducted at Rigshospitalet-Glostrup, from June 2014 through May 2015. Geriatric patients (GP) were recruited from the geriatric out-patient clinic. The patients were primarily referred to the clinic for fall assessment or general geriatric assessment after discharge from the hospital. The exclusion criteria were age < 65 years, severe communicative problems, moderate-severe dementia, and inability to walk without mobility aids.
There is no clear consensus on when an older individual has low muscle mass or strength and no cut-off values for low-grade inflammation has yet been defined. To be able to relate our results regarding muscle mass, muscle strength and low-grade inflammation in GP and to establish cut-off values for low, medium, and high levels of each inflammatory marker we conducted a secondary analysis on data from healthy older individuals (HO) collected as part of the Copenhagen Sarcopenia Study, conducted at Rigshospitalet-Glostrup, from December 2013 to June 2016 after initially being recruited through the Copenhagen City Heart Study. The participants were characterized by living independently and being apparently healthy. Exclusion criteria were acute medical illness, surgery within the last 3 months, ongoing medication known to affect body composition, and a history of compromised ambulation or prolonged immobilization. More detailed information on the Copenhagen City Heart Study and the Copenhagen Sarcopenia Study has been described previously [22,23]. In the present study only data from participants aged 75 and older were included.
All participants gave a written informed consent before participation and the study was approved by the Ethics Committee of the Capitol Region of Denmark (H-3-2013-124).
Muscle quantity, in the form of appendicular lean mass (ALM), and visceral fat were assessed by Dual energy X-ray Absorptiometry (DXA), and ALM/h2 was calculated using the participant’s height. To assess muscle strength, measurements of the participant’s handgrip strength (HGS) and leg extension power (LEP) were conducted. Physical performance was evaluated using measurements of habitual gait speed (GS) and 30 seconds sit-to-stand performance (STS). For more detailed information on how the specific tests were conducted refer to Suetta et al. [23].
Blood samples were obtained from the antecubital vein of each participant on the day of clinical examination and after all the muscle assessments were conducted. Body temperatures were not checked before blood samples were taken. The plasma concentration of the proinflammatory cytokines IL-6 and TNF-α were measured using the Bio-Plex MAGPIX multiplex reader (Bio-Rad Laboratories, Inc) utilizing Luminex xMAP technology (Luminex) and data was analysed using the Bio-Plex Manager Software, Version 6.1, Build 727. The lower limit of quantification (LLOQ) for each biomarker was based on the expected concentration of the lowest standard following an assessment of the recovery-range of the lowest standard. A recovery range between 60-140% was deemed acceptable. Cytokine levels below the LLOQ were substituted by simple imputation of a value between 0 and the LLOQ based on the lowest accepted standard for the respective cytokine. The LLOQ for IL-6 was 0.48 pg/ml and for TNF-α the LLOQ was 3.24 pg/ml. Serum levels of hsCRP were assessed using latex-entrenched immune-turbidimetry analysis (Roche/Hitachi automatic instrument COBAS®) and the LLOQ for hsCRP was 0.15 mg/L.
Participants were divided into three categories of low, medium and high depending on the levels of each of the three inflammatory markers, hsCRP, IL-6 and TNF-α. The cut-off values were established using data from the group of healthy older individuals from The Copenhagen Sarcopenia Study divided by gender. This was done by dividing the inflammatory markers of the HO into tertiles for hsCRP and TNF-α, and because of the great number of IL-6-values below the LLOQ, categories of IL-6 levels were established by setting "low IL-6" as all IL-6 levels under LLOQ (IL-6 < 0.48 pg/ml) and by dividing the rest of the IL-6 values equally into groups of medium or high.
The statistics were performed using SPSS software (IBM SPSS Statistics 26) and all analyses were stratified by gender. Descriptive analysis was performed with presentation of the data as means ± SD. Independent t-tests were conducted to examine if there were any significant differences in age, anthropometric measurements, and muscle parameters between the two sexes and between the GP and the HO. The data distribution for the inflammatory markers was skewed and are therefore presented as medians with interquartile ranges. The differences of the concentrations of the inflammatory markers between the GP and the HO were assessed using Mann-Whitney Test and to assess the differences in the distribution of participants in the categories of the three inflammatory markers Chi-square post-hoc tests were conducted based on adjusted standardized residuals and compared with the adjusted p-value of 0.008.
An ANCOVA was performed on data from the GP to assess the means, adjusted for age and BMI, for each muscle parameter for every one of the three levels of the inflammatory markers (Table 2). In the ANCOVA, we also conducted pairwise comparisons using Bonferroni corrections to assess whether there were any significant differences between the adjusted means for the muscle parameters between the three levels of inflammation.
Table 1: Adjusted* means ± standard error (one-way analysis of covariance) of muscle mass and muscle strength according to low, medium, and high levels of inflammatory markers in male and female geriatric patients (GP). View Table 1
189 patients referred to the geriatric outpatient clinic were consecutively screened for inclusion, 18 patients were excluded due to following: Communicative problems/dementia (n = 10), Age < 65 years (n = 3) and no independent walking (n = 5). 82 of the eligible 171 patients signed informed consent and 80 completed the full assessment. Prior to this consecutive study a pilot feasibility study was conducted including 15 geriatric outpatients. The GP group consisted of both these groups, a total of 95 patients.
1306 participants aged 20-93 years were included in the Copenhagen Sarcopenia Study [23]. Only 262 of these participants were aged 75 and older and were included in this study on this criterion.
In total 357 participants were included consisting of 95 GP (32 men/63 women) and 262 HO (112 men/150 women) (Table 1).
Table 2: Comparison of muscle parameters and inflammation markers for geriatric patients (GP) and healthy older individuals (HO). View Table 2
No significant difference was observed in age, height, weight, or BMI between the GP and the HO men and women (Table 1). However, men in the GP group had significantly more visceral fat than the male HO (21.22 ± 11.24 kg vs.16.59 ± 9.75 kg, p = 0.023). No difference in visceral fat was observed between groups for women (10.70 ± 7.98 kg vs. 9.42 ± 6.41 kg, p = 0.218). Despite, there being no difference in muscle mass between the GP and HO group for men and women, both the male and the female GP scored significantly lower in all the physical tests than the male and the female HO.
Moreover, men in the GP group had significantly higher concentrations of IL-6 (0.58 (0.24; 0.89) pg/ml vs. 0.24 (0.24; 0.50) pg/ml, p = 0.001) and TNF-α (21.63 (15.40; 28.68) pg/ml vs. 17.05 (13.56; 21.79) pg/ml, p = 0.033) compared to men in the HO group. And for women we found similar results between the groups not only for IL-6 (0.24 (0.24; 0.89) pg/ml vs. 0.24 (0.24; 0.24) pg/ml, p < 0.001) and TNF-α (20.76 (15.58; 24.52) pg/ml vs. 17.07 (12.54; 21.12) pg/ml, p = 0.001) but also for hsCRP (2.13 (0.78; 4.50) mg/L vs. 1.30 (0.72; 2.72) mg/L, p = 0.008).
The percentage distribution of participants with low, medium, or high levels of each inflammatory maker is displayed in Figure 1a-f. Of the female GP, 54.0% demonstrated elevated concentrations of hsCRP (> 2.05 mg/L) compared to 33.3% in the HO group. The difference between the two groups was significant (p = 0.005) which was also the case for women with high IL-6 (28.6% vs. 10.0%, p < 0.001) and high TNF-α (57.1% vs. 32.7%, p < 0.001). For men, the only significant difference was between the number of men with low IL-6 (< 0.48 pg/ml) in the GP and HO groups (40.6% vs. 72.3%, p < 0.001).
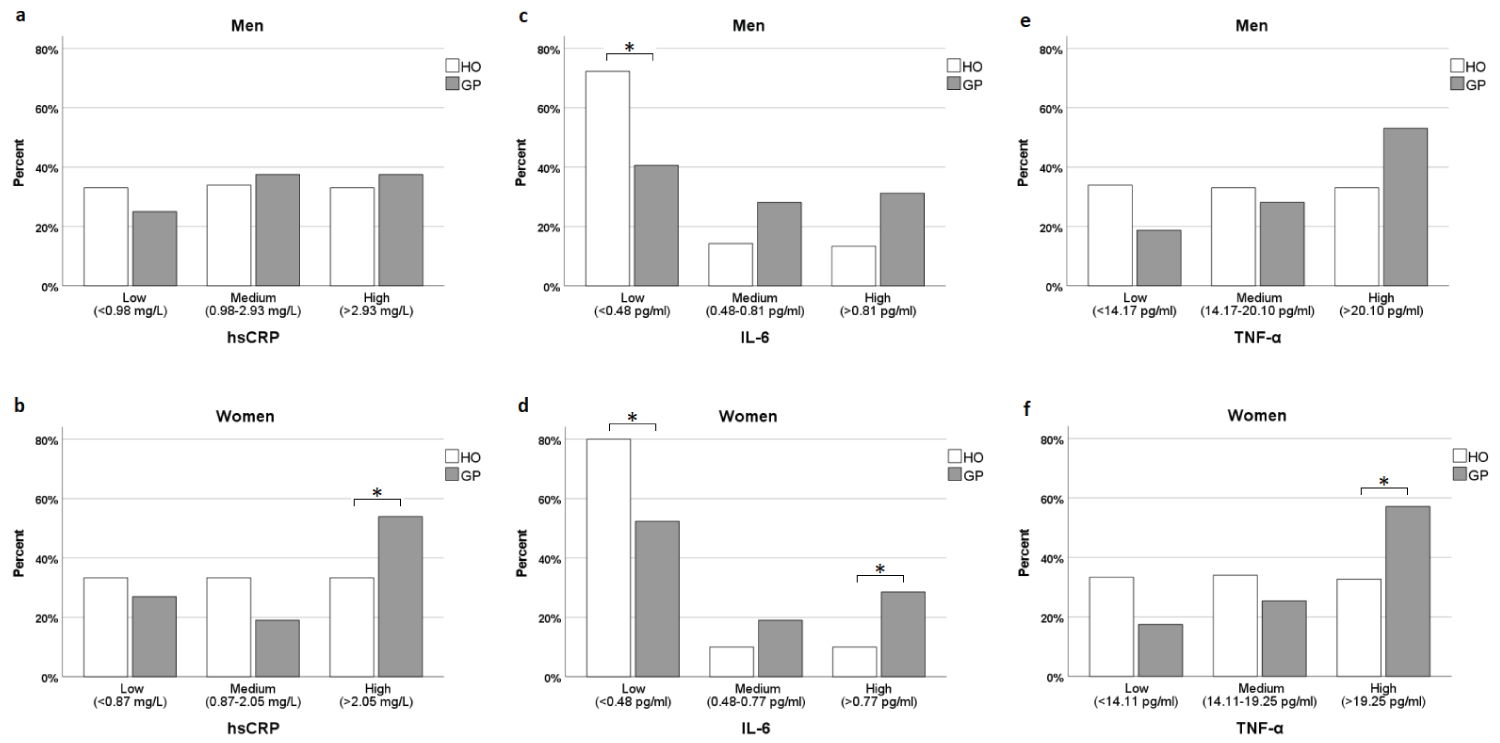 Figure 1 a-f: Percentage distribution of participants with low, medium, and high levels of each inflammatory maker. *P < 0.008. View Figure 1
Figure 1 a-f: Percentage distribution of participants with low, medium, and high levels of each inflammatory maker. *P < 0.008. View Figure 1
In Table 2 adjusted means for the different muscle parameters of the geriatric patients are shown for three levels (low, medium, and high) of each inflammatory marker. Notably, there was a 22.94% reduction in HGS in men having high levels of IL-6 compared to those with low levels and men with high hsCRP had reduced HGS compared to men with medium levels of hsCRP (29.63 ± 1.75 kg vs. 36.35 ± 1.74 kg, p = 0.033). Additionally, women with high hsCRP and high TNF-α had a 19.21% and a 22.46% lower HGS, respectively, compared to women with low levels of hsCRP and TNF-α. Apart from a reduced LEP in men with medium IL-6 levels compared to men with low IL-6 levels (114.6 ± 18.2 W vs. 179.7 ± 15.9 W, p = 0.032) ALM, ALM/h2, GS, LEP or 30s STS were not significantly different between the levels of inflammatory markers for neither men nor women.
In the present study we compare group of frail non-hospitalized elderly with a group of healthy older individuals combining assessments of muscle mass, muscle strength and physical performance in addition to plasma concentrations of inflammatory markers. Our results confirm a relationship between markers of low-grade inflammation (hsCRP, IL-6, and TNF-α) and hand grip strength in a population of geriatric patients from a Danish geriatric outpatient fall clinic.
In accordance with our hypothesis, our study population consisting of GP had reduced hand grip strength (HGS), Leg Extension Power (LEP), 30s STS performance and gait speed (GS) compared to healthy individuals of similar age, factors that all contributes to the diagnosis of sarcopenia. However, to our surprise, there was no difference in appendicular muscle mass between the two groups in men and women, respectively. It could be conceived that the GP group in the present study was not yet affected on muscle mass which is known to be affected at a later timepoint than muscle strength and physical function [23]. A possible explanation for this preliminary decline in muscle performance could lie on the potential influence of inflammation upon the muscle fiber contractility, where especially TNF-α amongst other cytokines plays a considerable role [24]. Regarding the plasma concentrations of the three inflammatory markers both hsCRP, IL-6, and TNF-α were significantly higher in the GP group compared to the HO group. Previous findings have demonstrated higher plasma concentrations of CRP, IL-6, and TNF-α in sarcopenic opposed to non-sarcopenic individuals [25-28].
Furthermore, we investigated the relationship between different levels of inflammatory markers and skeletal muscle mass, strength, and physical performance in the GP group. Of note, we found that men having high levels of IL-6, had significantly reduced HGS (22.94%) compared to men with low levels of IL6, and men with medium levels of IL-6 had 36.23% lower LEP compared to men with low levels of IL-6 (Table 2). In contrast, there was no difference between levels of IL-6 and any muscle parameters for women in this study. In line with our findings in men, a systematic review by Tuttle and colleagues found a negative correlation between plasma IL-6 and HGS. However, in contrast to our study, it found that this was true for both genders although the association was found to be larger in males than in females [29]. Only women with high levels of TNF-α in the present study had significantly lower HGS (22.46%) compared to individuals with low levels of TNF-α (Table 2). The same gender difference was seen in Visser, et al. where they found a significant association between higher circulating levels of TNF-α and lower HGS in women but not in men [5]. Focusing on hsCRP, men with high levels of the inflammatory marker had significantly lower HGS compared to men with medium levels of hsCRP and women with high hsCRP had a 19.21% lower HGS compared to women with low levels of hsCRP. Other studies have found similar results for CRP and HGS [30-32] for both genders and a big, population-based, cross-sectional study from Korea from 2018 found a significant correlation between CRP and HGS in men only [33].
Somewhat surprisingly, levels of inflammatory markers did not seem to be related to GS or STS in the present study in contrast to previous literature where a negative correlation between one or more of the three inflammatory markers (CRP, IL-6, and TNF-α) and GS [31] and STS [30,31] is reported. However, patient populations as well as methods for measuring muscle function and physical performance can vary considerably from study to study making comparison difficult. Another consideration when comparing studies is the fact that some studies have introduced an inflammatory profile, combining levels of several cytokines (e.g., IL-6 and TNF-α) to create an overall measure of cytokine status [5,8]. All these different methods to measure, analyze, and combine data make it very difficult to compare data from different studies in this field of research.
The fact that the study only found a significant correlation between inflammation and HGS could possibly be explained by HGS being a more direct measure of muscle function, whereas LEP, GS and STS are more overall measures of physical function. Therefore, it could be hypothesized that negative effects of inflammation are missed when using tests that rely on factors other than muscle function (e.g., balance, mobility, and cognition). In addition to this, HGS is less influenced by age-related degeneration of the joints which might downplay the relationship between muscle function and inflammation when assessing LEP, GS, and STS.
The biggest limitation of this study is the limited size of our patient population (GP group), which prevented a further subdivision into age groups, e.g., 65-74 yrs., 75-84 yrs. and > 85 yrs. Furthermore, as we measured gait speed one of our exclusion criteria were that the patients had to be able to walk independently to be included in the study, which meant we had to leave out the most fragile patients in the recruitment of participants to the GP group. As these patients were the ones with the poorest physical performance and were probably most affected by sarcopenia, it can be speculated that our findings perhaps would be stronger had these patients been included. Also, unfortunately, it was not possible to adjust for comorbidities as this information was not available in the healthy population. Additionally, the study design has its limitations. A cross-sectional study cannot conclude causality or temporality, but only an association between the matters of interest. Few longitudinal studies concerning the relationship between inflammation and muscle strength and function have been conducted [26,34,35]. Westbury and colleagues found in 2018 that in a group of 336 men and women of the Hertfordshire Cohort Study (HCS) born between 1931 and 1939, higher CRP, but not IL-6 or TNF-α, was associated with an accelerated decline in grip strength from baseline with a median follow-up time of 10.8 years later [34]. Moreover, Schaap and colleagues found that high IL-6 (> 5 pg/mL) and high CRP (> 6.1 μg/mL) were associated with a 2 to 3-fold greater risk of losing more than 40% of muscle strength after three years, in a group of 986 men and women (mean age: 74.6 yrs.) of the Longitudinal Aging Study Amsterdam (LASA) [35]. Importantly, these findings suggest that plasma concentrations of CRP and IL-6 perhaps can be used to predict loss of muscle strength and function.
Despite the limited number of patients in the present study we found that patients from a geriatric outpatient clinic had higher plasma concentrations of hsCRP, IL-6 and TNF-α compared to healthy individuals of similar age. Notably, high levels of hsCRP, IL-6 and TNF-α were associated with lower hand grip strength. Where other studies investigating inflammation and muscle mass and strength comes to similar conclusions there is a lack of information regarding the pathophysiological pathways for the influence of inflammation on the development of frailty and sarcopenia. As a future implication, assessment of inflammatory markers may be used as predictors of early signs of declines in muscle mass and strength, however larger studies are needed to clarify this aspect.