In this study, we examined the spread of SARS-CoV-2 infection and the dose-dependent efficacy of the COVID-19 vaccine Covishield (Oxford/AstraZeneca). During the period between December 2020 and February 2021, we tested the level of natural infection among individuals by estimating the reactivity of their sera towards SARS-CoV-2 spike (S) and nucleoprotein (N) proteins. The seropositivity of the population in different communities ranged from 17% to 51%, depending on their connectivity to the nearest metropolis Kolkata (population 14.85 million), the disease epicenter. We further found that while 90% of the people administered with two doses of Covishield developed antibody against SARS-CoV-2, only 55% developed antibody after one dose. Primarily those who had developed antibody through natural infection remained seropositive after the first vaccine dose in contrast to those with undetected infection. Our experimental findings not only contribute to a better understanding of COVID-19 epidemiology in India but also pave the way for an effective vaccination strategy involving the individual history of infection.
COVID-19, the abbreviated form of 'Coronavirus Disease-2019', is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. World health Organization declared COVID-19 as pandemic in March 2020 [2]. COVID-19 began in Wuhan in China in late 2019 and spread all over the world by the middle of 2020. As of May 27, 2021, ~3.5 million people died of COVID-19 and an additional ~170 million people around the world were known to have been infected [3]. However, both the numbers might be higher in reality since many countries have underreported and infections and deaths [4] and a large number of asymptomatic infected people have never been tested for the infection.
Coronaviruses belong to a large family of viruses. The family now has seven established human coronaviruses including SARS-CoV-2 [5]. The first two Human Coronaviruses (HCoV)-OC43 and -229E were isolated in the1960s [6,7]. These two viruses cause only mild respiratory problem and common cold. Four more human coronaviruses were found by 2012. Human coronaviruses HCoV-NL63 and HCoV-HKU1 are not pathogenic. But the other two -SARS coronavirus 1 (SARS-CoV), which emerged in November 2002, and MERS (Middle East Respiratory Syndrome) coronavirus (MERS-CoV), which emerged in 2012, [8,9] are highly pathogenic. Fortunately, they disappeared quickly. Finally, SARS-CoV-2 emerged in late 2019 [10,11].
The genome of the coronavirus is a single stranded ribonucleic acid (RNA) polymer [12,13]. The advantage of having an RNA genome is that some of the very essential proteins are generated just after the SARS-CoV-2 enters the host (human) cell. The viral surface protein, Spike (S), attaches through its receptor-binding domain (RBD) to the host cell receptor for entry into the cell. Membrane (M), envelope (E) and nucleocapsid (N) are other major structural proteins. N protein is involved in the transcription and replication of viral RNA and packaging of the encapsidated genome into virions. Both N and S are highly immunogenic and are used for the diagnosis of SARS-CoV-2 infection [14]. Generation of circulating anti-viral antibody in the host after viral infection or virus specific vaccine administration is essential for blocking viral entry into host cells [15]. The detection of SARS-CoV-2-specific antibodies in the blood plasma of unvaccinated individuals is a measure of host infection by the virus [16].
Most COVID-19 vaccines use S protein as the target with the idea that antibody generated against the S protein will successfully block viral entry into the host cell [17-19]. Vaccine shortages have started to impact the global vaccination efforts [20]. Thus although vaccinating as many people as possible and as quickly as possible without allowing emergence of antibodyevading variants is the best way out of the pandemic situation, efficacy of fractional vaccination of the previously infected individuals [21] is now being discussed as part of a protection plan [22,23]. However, such examination of efficacy has only been carried out in three industrialized nations (U.S.A., Italy, and Israel) with the mRNA vaccines (Pfizer-BioNTech and Moderna) [24-28].
In India, the first wave of infection and death reached a peak around September-October, 2020 [3]. In February, cases reached to its lowest level but a much stronger second wave largely caused by a mutant (B.1.617.2) that has already been detected worldwide is currently sweeping through the country. Although mass vaccination started in the country in late February, only about 14% of the population has received the vaccine so far.
In this study, we examined the severity of COVID-19 in the Indian population and evaluated the seropositivity of individuals after both natural infection and one or two doses of AstraZeneca's Adenovirus Vaccine Covishield. We noted that the disease severity in a certain population was controlled at least partly by its socioeconomic status. We also observed a positive correlation between natural infection and development of circulating antibody. While the first dose of vaccine helped maintain seropositivity in previously SARS-Cov-2 infected individuals, the second dose of vaccine was critical for development of antibody in those people with undetected SARSCov2 infection. Overall, these results provide a basis for modifications of policies in determining the dosage requirement of the Covishield vaccine.
N gene (Nucleocapsid protein gene) was synthesized and cloned in pUC 57 (Ampr) followed by subcloning in BioBharati pHIS-TEV vector (Cat# V0020) between Bam HI and Xho I restriction sites. Then E. coli (Rosetta DE3) cells were transformed with pHisTEV vector containing histidine taggedN gene. Cells were grown in LB media containing ampicillin (100 µg/ml) and chloramphenicol (17 µg/ml) at 37 ℃ till the light scattering value at 600 nm reached 0.6. Then the culture was induced with 0.5 mM IPTG at 37 ℃ for 4 hours. The cells were centrifuged and the pellet was resuspended in Buffer A [25 mM Tris-Cl (pH 7.5), 300 mM NaCl, 0.5% Triton-X 100, 5% Glycerol, 2 M Urea] and sonicated. The suspension was centrifuged at 12500 rpm for 30 minutes at 4 ℃. Supernatant was loaded onto 1 ml Ni-NTA agarose beads pre-equilibrated with Buffer A at 4 ℃. Flow through was discarded. Beads were washed 3 times with 10 column volume of Buffer A + 20 mM Imidazole. Protein was eluted using Buffer A containing 250 mM imidazole. Collected fractions were analyzed using 12.5% SDS PAGE. Purified protein concentration was estimated and stored as 50% glycerol stocks at -80 ℃.
S-RBD was obtained from a commercial source (Genscript, Catalog #Z03514-100) and as a gift from Dr. Gene Tan of J. Craig Venter Institute (La Jolla, USA).
Horseradish peroxidase (HRP) was first dissolved in water and activated with 0.25 M sodium periodate at room temperature for 30 minutes followed by purification by gel filtration in Sephadex G-25 column. Purified activated HRP was then conjugated to protein A/G at 4:1 molar ratio in the presence of 0.5 M carbonate buffer, pH 9.6 for 16 h at 4 ℃. Next, Protein AG-HRP was dialyzed against 0.1 M PBS, pH 7.5. The dialyzed Protein AG-HRP was then concentrated and mixed with BSA-Glycerol mix so that the final concentration of protein is 1 mg/ml. Goat antihuman IgG/A/M antibody HRP conjugated was obtained from Invitrogen (Catalog # A18847).
Roughly two drops of blood were from the finger-tip of each subject by inserting a sharp needle. The age range of all subjects, both males and females, was from 20 to 80 yr with nearly 50% with age below 45 yr. Samples were incubated overnight at 4 ℃ followed by centrifugation to separate bold sera. Sera were preserved frozen at -20 ℃ until ELISA.
ELISA was performed following the method published [29] with some modifications using a test kit developed inhouse. Briefly, 100 ng of S-RBD was coated in each well of 96 well high binding ELISA plates (JetBioFil, China). Medium binding ELISA plates were used for serological test against N protein test. The antigen was incubated for 16 h at 4 ℃ followed by washing with PBS (pH 7.5). The antigen coated plates were then blocked with 1% BSA mixed with stabilizing agent and again kept for 16 h at 4 ℃. Then the plates were washed with 1X TBST before addition of sera. Each serum was diluted in 1:100 ratio and incubated for 90 minutes at room temperature before washing with 1X TBST. The secondary antibody (Protein AGHRP) was mixed in 1:5000 dilution and kept at room temperature for 45 minutes followed by washing with 1X TBST. The plates were developed according to standard TMB method and OD was measured at 450 nm in BioRad Mark microplate reader five mins after the reaction was stopped. Usefulness of A/G-HRP was further confirmed using HRP conjugated antihuman IgG/A/M at 1:10,000 dilution.
The cutoff was determined using sera from 22 pre-COVID samples considered as negatives. The mean of the OD values of the negative control replicates plate plus three times the standard deviation of the OD value distribution gave the cutoff. 95% confidence intervals were calculated by using the formula x̄ ± z (s/√n), where x̄ is the sample mean, z is 1.96, s is standard deviation, and n is sample number. P-values between pre-COVID and post-COVID samples (Figure 1B and Supplementary Figure 1B) were calculated by one sample one tailed t-test. Briefly, tstatistic was calculated by using the formula (x̄ - μ)/sx̄, where x̄ is sample mean, μ is pre-COVID mean, and sx̄ is s/√n (s is sample standard deviation and n is sample number). Then ttable was used to determine the pvalue range from the t-statistic. Samples from different communities were compared to that of Kolkata (Figure 2) using unpaired two-sample one-tailed ttest in Microsoft Excel. Samples from the same set of individuals obtained at two different times (Figure 3C, Figure 3D and Supplementary Figure 2) were compared using paired two-sample onetailed t-test in Microsoft Excel. Results obtained with protein A/G and IgG were compared using paired twosample twotailed t-test in Microsoft Excel. Samples were analysed for the distribution plot with Graph Pad Prism version 8.3.1 software.
 Figure 1: Experimental design. (A) Samples were collected from the city of greater Kolkata, and the villages of Lakshminarayanpur, Bhadur, and Mukutmanipur in the districts of South 24 Parganas, Hooghly, and Bankura, respectively. The aerial distances of these places from Kolkata central are shown; (B) Distribution of Sproteinspecific antibody level in preCOVID and RTPCR positive samples; p-value and 95% confidence interval of the distributions are shown.
View Figure 1
Figure 1: Experimental design. (A) Samples were collected from the city of greater Kolkata, and the villages of Lakshminarayanpur, Bhadur, and Mukutmanipur in the districts of South 24 Parganas, Hooghly, and Bankura, respectively. The aerial distances of these places from Kolkata central are shown; (B) Distribution of Sproteinspecific antibody level in preCOVID and RTPCR positive samples; p-value and 95% confidence interval of the distributions are shown.
View Figure 1
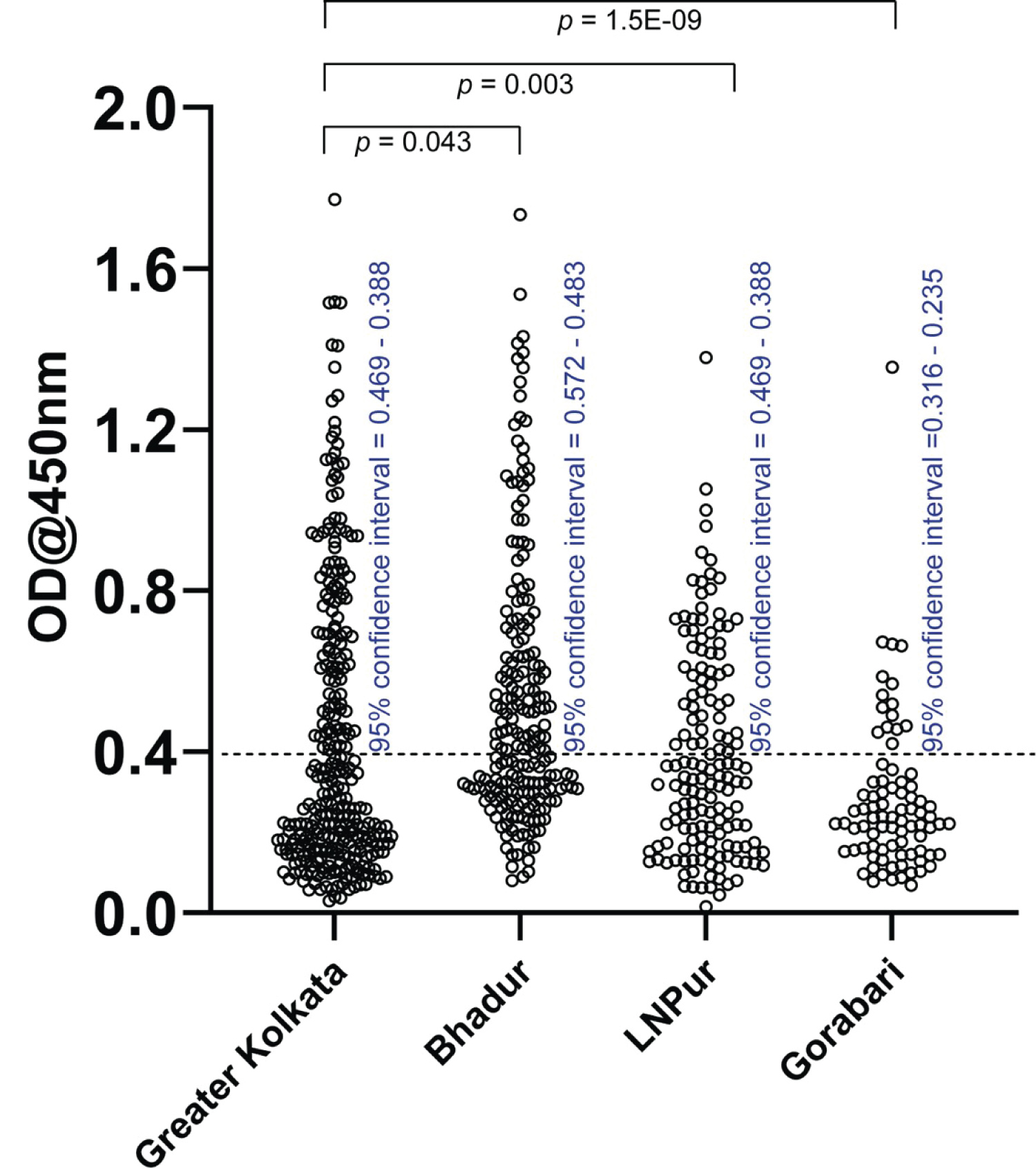 Figure 2: Seroreactivity of samples collected from the four regions Sample sizes for greater Kolkata is 292 (total population ~14.85 million), Bhadur 203 (total population ~3000), Lakshminarayanpur 150 (total population ~4000), Gorabari 83 (total population ~1100); 95% confidence intervals and p values for comparing individual rural distributions to that of Kolkata are shown.
View Figure 2
Figure 2: Seroreactivity of samples collected from the four regions Sample sizes for greater Kolkata is 292 (total population ~14.85 million), Bhadur 203 (total population ~3000), Lakshminarayanpur 150 (total population ~4000), Gorabari 83 (total population ~1100); 95% confidence intervals and p values for comparing individual rural distributions to that of Kolkata are shown.
View Figure 2
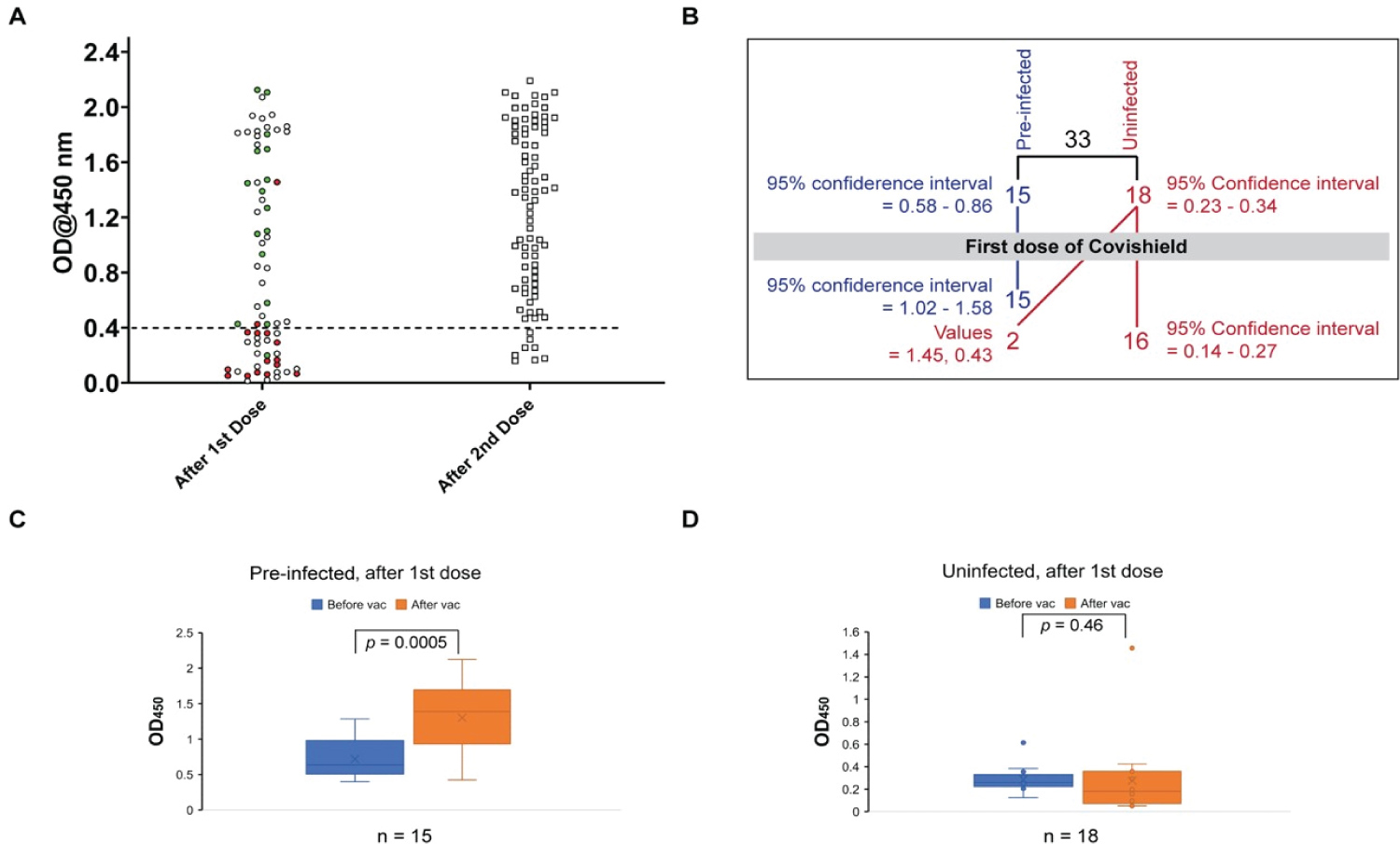 Figure 3: Antibody development through SARSCoV2 infection and vaccination. (A) Distribution of seroreactivity levels of people after the first (left plot) and the second (right plot) dose of AstraZeneca's Covishield vaccine; -the red circles indicate previously uninfected individuals, green circles previously infected individuals, and hollow circles individuals with no available prior history; (B) Distribution of seroreactivity levels of individuals with known history of infection after the first dose of Covishield vaccine showing development of seropositivity in only 2 out of 18 previously uninfected individuals; (C) Distribution of seroreactivity levels of previously infected individuals before and after the first dose of Covishield showing significant enhancement in seropositivity upon vaccination;; the horizontal line within the boxplot indicates median value and the 'x' average; (D) Distribution of seroreactivity levels of previously uninfected individuals before and after the first dose of Covishield showing no significant change in seropositivity after vaccination.
View Figure 3
Figure 3: Antibody development through SARSCoV2 infection and vaccination. (A) Distribution of seroreactivity levels of people after the first (left plot) and the second (right plot) dose of AstraZeneca's Covishield vaccine; -the red circles indicate previously uninfected individuals, green circles previously infected individuals, and hollow circles individuals with no available prior history; (B) Distribution of seroreactivity levels of individuals with known history of infection after the first dose of Covishield vaccine showing development of seropositivity in only 2 out of 18 previously uninfected individuals; (C) Distribution of seroreactivity levels of previously infected individuals before and after the first dose of Covishield showing significant enhancement in seropositivity upon vaccination;; the horizontal line within the boxplot indicates median value and the 'x' average; (D) Distribution of seroreactivity levels of previously uninfected individuals before and after the first dose of Covishield showing no significant change in seropositivity after vaccination.
View Figure 3
 Supplementary Figure 1: Experimental design. (A) Boxplot demonstrating distribution of seroreactivity values obtained with Protein A/G and IgG showing no significant difference between the two:- The horizontal line within the boxplot indicates median and 'x'average; (B) Distribution of seroreactivity levels for N-protein-specific antibody in pre-COVID and RT-PCR positive individuals showing the cut off for seropositivity being about 0.2.
View Supplementary Figure 1
Supplementary Figure 1: Experimental design. (A) Boxplot demonstrating distribution of seroreactivity values obtained with Protein A/G and IgG showing no significant difference between the two:- The horizontal line within the boxplot indicates median and 'x'average; (B) Distribution of seroreactivity levels for N-protein-specific antibody in pre-COVID and RT-PCR positive individuals showing the cut off for seropositivity being about 0.2.
View Supplementary Figure 1
 Supplementary Figure 2: Distribution of seroreactivity levels for N-specific antibody of RT PCR positive individuals showing gradual loss of N-specific antibody over time.
View Supplementary Figure 2
Supplementary Figure 2: Distribution of seroreactivity levels for N-specific antibody of RT PCR positive individuals showing gradual loss of N-specific antibody over time.
View Supplementary Figure 2
An Institutional Review Board (IRB) on Human Subjects was constituted following the guidelines of the Indian Council for Medical Research (ICMR). The IRB read and discussed the research proposal, and provided certificate of approval for the research conducted in this study. Samples were collected following the protocol approved by the IRB. Samples were collected from donors after receiving their signed consent. Gram Panchayat (Village Council) leaders along with the local non-government organization helped assemble donors in the rural places and were present during sample collection. In Kolkata, donor groups were assembled by various non-government organizations.
This study was focused on understanding the first wave of SARS-CoV2-infection through the estimation of seroprevalence and efficacy of the Covishield vaccine among a portion of the Indian population. We chose people from four different areas within the southern part of the Indian state of West Bengal (Figure 1A). The study was conducted in two parts. In the first part, encompassing the period from the December 2020 to February 2021, we aimed at determining the fraction of the population under study infected by the SARS-CoV-2 virus. Additionally, we were interested in finding out if there was any correlation between infection rate and socioeconomic status. In the second part, conducted in the middle of April, we tested people after one or both doses of the Covishield vaccine for potential seropositivity, approximately 30 days after vaccination. In the greater Kolkata spanning around 60 square miles, we collected samples from 6 small areas. We also included three rural areas- Lakhminarayanpur in South 24 Paraganas district (66 km from Kolkata), Bhadur in Hooghly (110 km from Kolkata) and Gorabari in Bankura (250 km from Kolkata). The idea was to investigate if people were living further from Kolkata are least affected by the virus. About 65% of the total number of people tested belonged to the working class (described in Table 1).
Table 1: Summary of number of test subjects in four different places. View Table 1
Serological tests were conducted against two different viral structural proteins, nucleoprotein N and Spike (S). For the spike protein, we used only the receptorbinding domain (RBD) of S (SRBD), the protein domain of the virus that directly interacts with host cell receptors. We used two different secondary antibodies - goat anti human IgG monoclonal and protein A/G. Both produced similar results (Supplementary Figure 1A). Since protein A/G gave a lower background, we chose HRP conjugated protein A/G as the detection probe. Using 22 samples collected before the pandemic we determined the cutoff to be 0.4 for S- RBD (Figure 1B). To determine the specificity of the assay we also tested 29 RTPCR positive samples of patients showing a broad spectrum of disease severity, which included hospitalization of 14 people (1 died later). The antibody titer of 28 of these samples was above the cut off, barring two samples, both of which in fact were positive for N (the cut off for N protein was 0.2 - Supplementary Figure 1B).
In order to assess the level of SARS-CoV-2specific antibody generated through natural infection we collected around 300 samples from Kolkata between December 15 and February 19 of which nearly 36% were females and 64% males. Nearly 25% of these samples belonged to the working-class population and 75% to the middle class. Serological tests were conducted with both the SARS-CoV-2 N- and S-specific antibodies (Table 1 and Supplementary Table 1). However, during the course of the study, we observed a significant reduction of N-specific antibody over time (Supplementary Figure 2). An earlier report indicates that the Nspecific antibody may disappear quickly from the circulation [30]. This led us to rely on only the data from the SRBD tests. Within a total of 292 samples from Kolkata tested against SRBD, 42% was found to be seropositive (Figure 2 and Table 1). We observed no preference in infection between male and female or between the working class and middle class of different age groups (Table 1).
Supplementary Table 1: Density grades in male group. View Supplementary Table 1
Thereafter, we tested 150 people comprising of both males and females in the village of Lakshminarayanpur (population ~4000, District: South 24 Paraganas), 66 km from Kolkata (Figure 2 and Table 1) and found 39% seropositivity for the S protein. Similarly, among 198 people living in Bhadur (population ~3000, District: Hooghly), 110 km from Kolkata nearly 51% turned out seropositive for SRBD (Figure 2 and Table 1). Additionally, among 83 people in the village of Gorabari (population ~1100, District: Bankura), which is nearly 250 km away from Kolkata only around 17% of the population turned out seropositive (Figure 2 and Table 1). Like Kolkata, we found no preference of infection based on gender or economic status. Although Bhadur (Hooghly) is further from Kolkata than Lakshminarayanpur (South 24 Parganas), transportation of people between the district of Hooghly and the city is Kolkata is likely more common as reflected by the difference in the average GDP per capita (₹70639 in Hooghly vs ₹58808 in South 24 Parganas) [31]. We further noted that while none from the working class population in our study was severely sick more than 50 people from middle class exhibited severe sickness including hospitalization and death. We did not receive any reports of comorbidity.
Overall, through our estimation of SRBD specific seroprevalence in a total of 734 subjects comprising Kolkata and adjoining rural areas (Table 1), we found that SARS-CoV-2 infection rate ranged between roughly 17% and 51%. Our experimental results are suggestive of a correlation between the infection rate and connectivity to Kolkata within approximately ~150 km radius around the city.
In order to evaluate the efficacy of the Covishield vaccine we conducted serological tests after the first and second doses of vaccination. Initially, we tested 71 people (27 females) about one month after the first dose of vaccine (Figure 3A, left plot). In this group, we included 33 people of the phase I study group, of who 15 were Santibody positive and 18 were negative (Table 2 and Figure 3). The preinfected people showed some enhancement of the level of circulating antibody. However, only 2 out of 17 with undetected infection exhibited enhancement of antibody level above the cutoff of 0.4. Additionally, out of the 38 people, who were not tested in phase I, 25 exhibited seropositivity. Since the majority of these people were from Bhadur where ~51% of population had natural infection, it is possible that a large proportion of those 25 people who were not included in the phase I study were also naturally infected. Based on these observations, we concluded that only a small fraction (~1015%) of the uninfected population exhibits seropositivity after the first dose of vaccine. Failure to develop detectable levels of circulating antibody after one month following the first dose of vaccine suggests that a second booster dose is essential for protection.
Table 2: Summary of the effect on seroreactivity of one and two doses of vaccination (A) Table showing distribution of individuals with one dose of vaccination; (B) Table showing distribution of individuals with two doses of vaccination. View Table 2
We also tested 83 people about one month after the second dose of vaccination. Only 8 out of these 83 subjects failed to develop antibody (Figure 3A, right plot and Table 2). This data conforms to the high efficacy of two doses of the Covishield vaccine. It is, however, possible that the higher seropositivity percentage after the second vaccine dose is at least partly contributed by an earlier natural infection. Taken together, our results suggest that one dose of Covishield vaccine could maintain the circulating antibody level after natural SARSCoV-2 infection.
Serological studies conducted in this study after the first wave of COVID-19 indicate that up to nearly 50% of the population in different communities in and around Kolkata could have been infected with SARS-CoV-2 by February 2021. Constant travel between Kolkata, the disease epicenter, and nearby places might have had induced spreading of the infection within roughly a radius of 150 km. An indepth survey revealed that although Lakshminarayanpur and Bhadur had nearly similar fraction of the population infected as Kolkata, severity of the disease resulting from infection was more in Kolkata than in the other places. Perhaps the higher density of infected people in the city caused repeated infections leading to severe illnesses. Disease severity was also found to be least among the working class group, be it in village, small town or city suggesting that severity or mildness of disease outcome from infection could be linked with physical labor. This conclusion is drawn based on our observation of zero hospitalization or severe sickness of all working class people included in this study compared to at least fifty cases of disease severity including hospitalization and two deaths among the middle class.
Other populationbased studies in India on the seroprevalence of SARS-CoV-2 antibodies have been reported. However, these studies were done either before or around the first peak of SARS-CoV-2 infection [32-34]. Moreover, these studies targeted only the viral N protein, not the S protein, which generates longer lasting circulating antibodies [30].
Anti-COVID-19 vaccination started in February and a small fraction of population in the state of West Bengal received one or two doses of vaccine by April 2021. Accordingly, in order to determine the presence of circulating COVID-19 specific antibodies, we tested a few individuals in these two groups of vaccinated people. Similar studies have been done with other vaccines but not with Covishield [22,26-28]. Our study demonstrates that while the population that was already infected exhibited enhancement of circulating antibodies after the first dose of Covishield, in the population with undetected infection, only 1015% showed seropositivity after the first dose. This is not to imply that the first dose was not effective, because after the second dose around 90% people exhibited circulating antibody. In this context it is worth mentioning that although circulating antibody potentially provides immediate protection against infection, it is the response of the memory B and T cells to infection that bestows longterm immunity [35]. Thus, having undetectable antibody may not necessarily indicate ineffectiveness of the first dose of the Covishield vaccine.
Taken together, our study suggests that the potential for seroprevalence after natural infection and vaccination is quite the same. Therefore, people with antibody after natural infection perhaps could wait to be vaccinated giving way to the other group with no apparent infection or circulating antibody. However, making such decision requires testing people within one to two months after infection. Although India is the biggest producer of vaccine, it is facing vaccine shortfall [20]. A thoughtful knowledge-based vaccination program might at least partly mitigate the problem.
India is currently going through the second wave of infection predominantly by new SARS-CoV-2 mutant(s), after the rampant mixing of people in large gatherings. These mutants appear to be more virulent and rapid spreaders. The question that remains to be answered is whether the current vaccines generated against the original virus (wild type) can protect people infected with the second wave mutants or other unseen mutants.
Authors thank the following nongovernment and government organizations whose support was vital to complete the study: Bhadur Gram Panchayat (Naba Pal), Kolkata Hridayam (Kajal Sinha), Indranarayan Saradar (Lakhminarayanpur Patna Vidyasagar Sevakendra and Sisuthirtha) and Bandhu Ek Asha (Nupur and Pritam), and Agradut Polly Unnayan Samiti (Umasankar Mukhopadhyaya and Sarit Mohanti). Authors also thank the following people for their support: Dr. Prabhas Sadhu Khan, Dr. Subhas Ghosh and New Avenue Nursing Home, Dr. Ratna Chattopadyay, Dr. Pradipta Dasgupta, Dr. Mahua Sen, Dr. Sandip Ghosh, Dr. Archya Sengupta, Dr. Manisankar Ghosh, Manab Sengupta, Biren Bhattacharjee, Prashanta Kundu, Rabi Meddya, Satadeepa Kal, Sreyashi Maity and Asit Chowdhury (Kolkata); Batakrishna Dhara, Bankim Ghosh, Tarasankar Ghosh, Sabitri Mapui, Riju Malakar, Ranajit Kotal, Dipsankar Ghosh (Bhadur), Prabir Santra and Pratap Santra (Gopinathpur) for help in sample collection. GG and MS thank Mr. Umasankar and Mrs Jyotsna Ghosh for partial financial support and constant encouragement to conduct the project.