Objective: Compare differences in pain pressure threshold (PPT) at an identified myofascial trigger point in the upper trapezius muscle amongst office workers after the application of various topic analgesic products.
Methods: Participants were recruited from their workplaces and assigned to a treatment arm, with both participant and investigators blinded. Baseline measurements of participants' PPT obtained, and VAS used to determine pain severity. One of 6 treatments was applied with re-assessment of PPT 7 minutes later. Comparison of the baseline and treatment scores was due using two-way repeated measures ANOVA. Trial registered retrospectively - NCT03939884.
Results: There were 120 participants, with 20 subjects randomly allocated to each arm. No significant between group differences in PPT were noted at baseline or post-treatment using repeated measures ANOVA (F(1,57) = 1.18; p = 0.32). Within group differences were observed (F(1,57) = 4.79; p = 0.03) for MuscleCare Roll-on without MSM, demonstrating the greatest increase in PPT from baseline to post-treatment. No differences in VAS were found between groups.
Conclusion: This study demonstrated that the non-pharmacologic, topical analgesic product (MuscleCare™) is at least as efficacious as a NSAID in the treatment of trapezius trigger point musculoskeletal pain.
Chronic pain can be debilitating and negatively affects activities of daily living [1-3]. While pain is complex, there may be a large portion related to musculoskeletal issues. Much of this pain is a result of myofascial pain syndrome (MPS), a syndrome in which pain from muscular trigger points causes pain in other parts of the body, a phenomenon commonly known as referred pain [4]. This may be due to increased nociceptive input from skeletal muscle or from central convergence around the dorsal horn neurons, which has resulted in multiple treatment options. Numerous studies have tried to provide insight into the best treatment for MPS with common treatment regimens including both pharmacologic and non-pharmacologic intervention [5].
Pharmacologic treatment has primarily revolved around the use of opioid agents for severe chronic pain, non-steroidal anti-inflammatory drugs (NSAIDs) and acetaminophen, or anti-depressive agents such as duloxetine and amitriptyline [6-8]. Williams, et al. (2014) found that, despite its widespread use, the usage of acetaminophen for low back pain did not result in clinically significant improvements in recovery time. Typically, these types of pharmacological treatments have been reserved for individuals with chronic severe pain or those who have failed to improve with conservative care. Those with mild to moderate pain may be treated with milder analgesic agents alone or in combination with a range of physical interventions such as exercise; soft tissue techniques including massage, myofascial release, trigger point release techniques; spinal manipulation; Tai Chi; Yoga; and either acupuncture or dry needling [5,9,10]. Various studies have found these techniques to be effective, however they often only provide temporary relief of the pain and still require patients to routinely follow-up with their practitioners [9,11-14].
A recent systematic review found that topical NSAIDs had a moderate effect at reducing pain immediately and up to 7 days, as well as scoring highly in terms of patient satisfaction [15]. This topical application led to decreased NSAID-associated adverse events such as gastrointestinal distress. However, this study did not address non-pharmacological topical applications. There is a lack of research for non-pharmacological topical applications within the treatment of myofascial pain, given that they may allow for multiple mechanisms of action including placebo while minimizing potential adverse events commonly associated with NSAIDs [7].
One non-pharmacological topical analgesic that has shown promise at treatment pain is MuscleCare™. A previous study has demonstrated that MuscleCare™ causes a significantly greater increase in pain pressure threshold (PPT) than other leading national brands including Biofreeze and IcyHot [16]. However, there are currently no data describing the efficacy of MuscleCare™ against any products including a non-steroidal anti-inflammatory drugs (NSAID) such as Voltaren®, which uses Diclofenac as its active ingredient.
The purpose of this study was to compare MuscleCare™, non-pharmacologic topical pain relief ointment, and the NSAID Voltaren®, in the acute treatment of myofascial pain in the upper trapezius. The objective of this study was to compare differences in pain pressure threshold (PPT) at an identified myofascial trigger point in the upper trapezius muscle amongst office workers after the application of various topic analgesic products.
This study involved pre-post measures. Subjects were using convenience sampling and randomly allocated to one of six possible treatment groups. The testing session occurred at the subjects' desks, which involved a baseline measurement followed by the application of the topical analgesic and then a follow-up measurement of PPT. All of this occurred within a 10-minute timeframe. Institutional ethics approval was obtained from the University of Toronto Research Ethics Board (33247) in confirmation with the Declaration of Helsinki on the use of human subjects.
Subjects included clerical and administrative staff who refrained from any form of manual labour over the course of the procedure. Subjects were excluded if they presented with any type of shoulder, neck, or back pain,absence of a palpable tender spot in the right upper trapezius region (identified by manual palpation by an indepdent assessor prior to enrollment), any history of allergic reaction to previous use of any analgesic topicals, any disorder of the skin over the right trapezius area, any previous surgery or major injury to the area of application. All subjects completed an informed consent.120 adult males and females between the ages of 21-65 wererecruited from variousworkplace environments, utilizing a sample of convenience for this study. Preliminary sample size calculations determined that this size would provide sufficient statistical power to detect any differences. All subjects consented to participate in the study, with 20 subjects randomly allocated to each treatment group (Figure 1).
A random number sequence of treatments was generated in Microsoft Excel by a blinded data analyst and each participant was randomly allocated to a single intervention group (see below). Concealment was accomplished by having files pre-prepared with the allocation contained in a sealed envelope which was opened by the examiner immediately prior to the application of the topical product in that session. Each product was kept in a generic white plastic container, all of the same size shape, and labeled from A-F. Blinding of topical odour unique to each treatment was achieved with the use of nose-clips (Figure 1).
Six topical preparations were assessed in this study: four were natural topical analgesics, one was a topical non-steroidal anti-inflammatory drug (NSAID), and one was a non-medicinal placebo which was used as the control. The natural topical analgesics comprised of four different preparations of MuscleCare™. Two were ointments and two were roll-on gels. Within each of those groups, one preparation was prepared without the active ingredient methylsulfonylmethane (MSM), an organosulfur compound. The NSAID-based product was Voltaren® and the non-medicinal placebo was a special preparation that was applied as a roll-on gel comprised of water, glycerin, and guar to emulate the feeling of the other products with none of the active ingredients. Products were placed in identical 3 oz generic white roll-on bottles or 0.5 oz white plastic containers and were labeled only with a letter. The coding for these preparations was kept from all examiners until conclusion of the study.
A research assistant provided all subjects with a noseclip to control for odour. After an independent examiner identified the presence of a myofascial trigger point and marked it on the skin, another blinded assessor applied the randomly assigned topical analgesic. The assessor performing the application performed adequate hand hygiene between subjects and donned gloves to eliminate the risk of cross-contamination. After the application of the topical analgesic, the area was lightly massaged to allow the topical ointment to absorb into the skin.
All topical analgesics were applied using the same process and same amount of product. This was done to help eliminate any biases and maintain blinding.
An assessor palpated the upper trapezius muscle in the samples and identified the presence of a myofascial trigger point, which was subsequently marked on the skin. If no myofascial trigger point was identified, the subjects were excluded. A score of tenderness at a myofascial trigger point (MTP) in the right upper trapezius and a visual analog scale (VAS) of pain was obtained. Following this, a manual pressure algometer (FPK20, Wagner Instruments, CT, USA) was used to assess tenderness in the trapezius muscle with all measurements were reported in kg cm-2 [17,18]. Vertical pressure was applied by the pressure algometer at a rate 1 kg cm-2 s-1 until the patient indicated the first presence of tenderness. Previous research has demonstrated reliability and validity for the use of pressure algometry in manual therapy [19]. The measurement was then repeated after application of one of the 6 topical preparations in the same manner. The difference of the two readings was used as the indicator of the efficacy of the treatment. A VAS from 0-10 was used to quantify pain in the upper trapezius pre- and post-application of the topical analgesic. The scale was presented to the subject immediately after tenderness was found through pressure algometry and they were instructed to rate the level of pain on a scale of 0-10. This measurement was then repeated with the second algometry reading and the difference of the two were used as an outcome of the efficacy of the treatment.
To minimize extraneous factors that may influence pain perception, subjects were asked to sit upright at their desk, in a comfortable position for the testing procedures. Both subjects and examiners wore a nose clip to ensure they were blinded to the product being applied. Pressure algometry measures were obtained to determine baseline tenderness, measured by pain pressure threshold (PPT) in the upper trapezius, as described previously [16]. A second examiner then applied one of the 6 topical preparations, determined by randomization, over the marked area. The substance was massaged into the skin for 10 seconds by the second examiner who donned gloves and performed appropriate hand hygiene between subjects. Following application, subjects were left for 7 minutes to allow for absorption and penetration of the preparation into the tissues. This timeline was based on expert opinion from local health care practitioners. During these 7 minutes, subjects were instructed to stay seated in the chair with minimal head movement. At the end of the 7 minutes, the first examiner returned to re-assess the PPT and VAS, using the same procedure.
Baseline descriptive statistics and analysis of change scores were conducted using a baseline ANOVA and two-way repeated measures ANOVA in Statistical Package for Social Sciences (SPSS) with a significance level set at 95% (p < 0.05).
All subjects consented to participate in the study, with 20 subjects randomly allocated to each treatment group (Figure 1). Of the 120 subjects, 54were male and the average age was 39 ± 3. Baseline subject demographics are presented in Table 1. Assessment of the PPT in the right upper trapezius both pre- and post-application is presented in Table 2.
 Figure 1: Consort diagram showing the random allocation of participants to one of 6 different pharmacologic treatments or a non-medicinal placebo.
View Figure 1
Figure 1: Consort diagram showing the random allocation of participants to one of 6 different pharmacologic treatments or a non-medicinal placebo.
View Figure 1
Table 1: Baseline demographics. View Table 1
Table 2: Assessment of PPT before and after treatment of one of 6 preparations. View Table 2
Baseline ANOVA revealed no significant differences in PPT between groups at baseline, or between groups using repeated measures ANOVA (F(1,57) = 1.18; p = 0.32) as shown in Figure 2. However, within-group differences were observed (F(1,57) = 4.79; p = 0.03) for MuscleCare Roll-on without MSM only (Product F), demonstrating the greatest increase in PPT from baseline to post-treatment (0.75 ± 0.006 kg/m2, p = 0.03) (Figure 3).
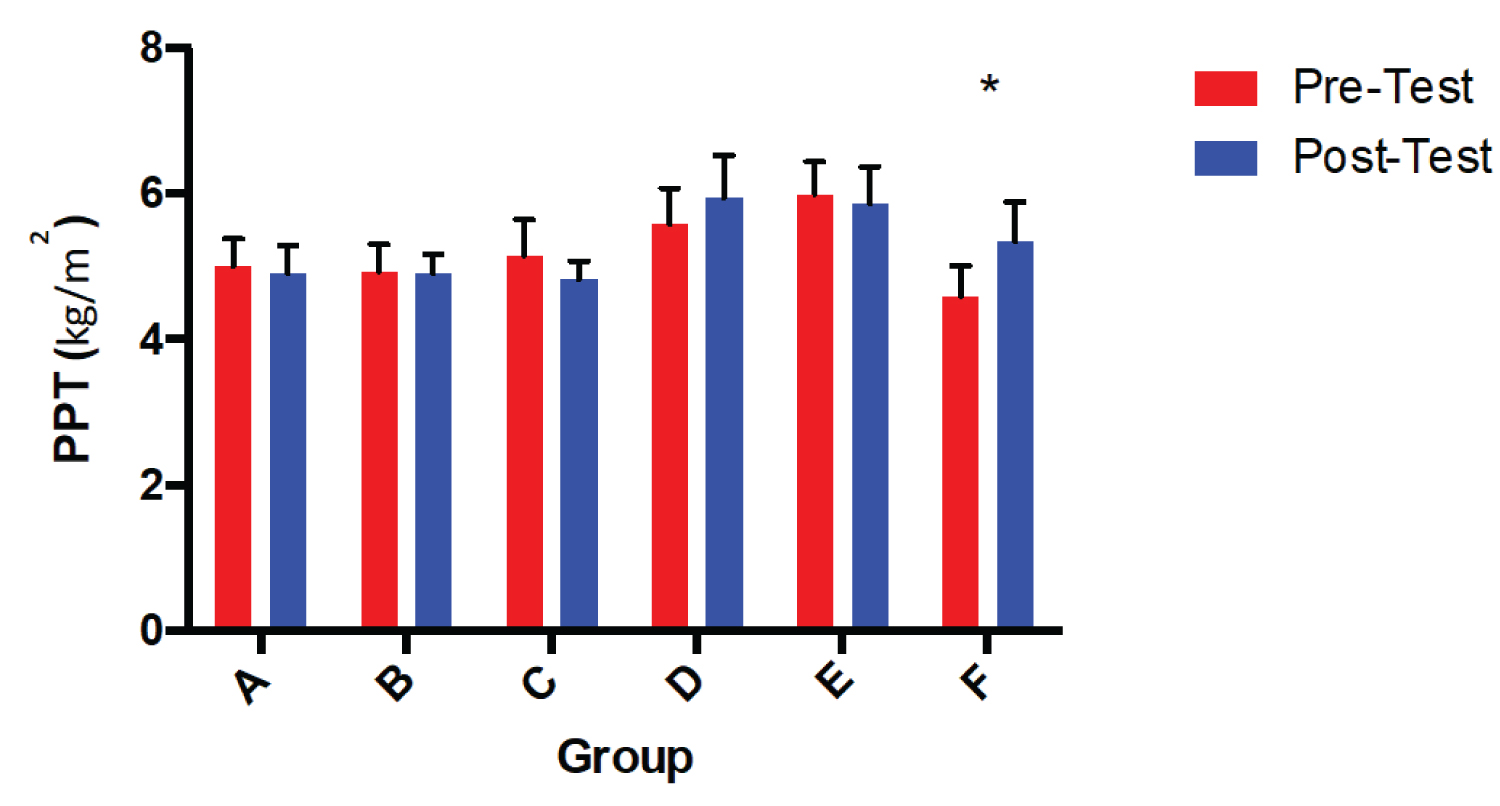 Figure 2: PPT from baseline to post-treatment in all six groups.
Figure 2: PPT from baseline to post-treatment in all six groups.
*Indicates a significant within group difference, (p = 0.03) A = MuscleCare Roll-on, B = Voltaren, C = MuscleCare Ointment, D = Non-medicinal Placebo, E = MuscleCare Ointment without MSM, F = MuscleCare Roll-on without MSM.
View Figure 2
 Figure 3: The difference in pressure point threshold from pre to post-treatment for all six preparations. *Indicates a significant within group difference (p = 0.03). A = MuscleCare Roll-on, B = Voltaren, C = MuscleCare Ointment, D = Non-medicinal Placebo, E = MuscleCare Ointment without MSM, F = MuscleCare Roll-on without MSM.
View Figure 3
Figure 3: The difference in pressure point threshold from pre to post-treatment for all six preparations. *Indicates a significant within group difference (p = 0.03). A = MuscleCare Roll-on, B = Voltaren, C = MuscleCare Ointment, D = Non-medicinal Placebo, E = MuscleCare Ointment without MSM, F = MuscleCare Roll-on without MSM.
View Figure 3
Assessment of the VAS of pain is shown in Table 3. There were no within or between group differences in VAS from pre- to post-treatment.
Table 3: Assessment of VAS before and after treatment of one of 6 preparations. View Table 3
This study demonstrated significant within-group differences for MuscleCare™ Roll-on without MSM, however there were no significant differences between groups. This suggests that this MuscleCare Roll-on without MSM preparation produces the greatest increase in PPT following treatment, however the reductive effect in terms of pain management was not significantly different between groups. In addition, the VAS of pain perceptions also showed no differences amongst any of the preparations used in this study. Despite these statistical differences, it is unclear if there is any clinical significance between groups.
To our knowledge, this is the first study to demonstrate that an all-natural product (MuscleCare™) is at least as effective as a product containing a NSAID (Voltaren®) at acutely improving PPT in the trapezius trigger point. There are numerous reasons why an all-natural product may be preferred to the use of NSAIDs. Firstly, there is no evidence to suggest these products produce any of the side effects that are seen with NSAIDs. This includes but is not limited to disturbances to the gastro-intestinal system [20] and the contraindications in patients with any cardiovascular risk factors [21] due to the reduction of prostaglandins that are responsible for maintaining vascular homeostasis and integrity [22]. Additionally, natural products like MuscleCare™ contain more active ingredients than pharmacologic products such as Voltaren®, which only contains Diclofenac sodium as an active ingredient. This allows for the possibility of synergistic effects between the ingredients, which could contribute towards increased benefit for the patient. Further research is needed to clarify these proposed mechanisms. Lastly the inclusion of Hydresia oils in MuscleCare™ may allow for increased delivery of active ingredients into the tissue [23], and thus creates the potential for a greater efficacy of the active ingredients and a greater reduction in pain for the patient.
Further research may be useful to determine if similar results are observed throughout various types of musculoskeletal pain, as well as across various timelines. While the healing process is similar across various musculoskeletal injuries, the effective of therapeutic interventions may not necessarily be similar. Furthermore, as per Quintner, et al. the underlying pathophysiological of trigger points is unclear [24].
Given the absence of detectable perception of pain reduction between topical agents, the benefits of a non-pharmocological product may offer a clinical advantage for health care practitioners who wish to avoid pharmacological interactions and limit the risk of adverse events associated with NSAID usage. The topical analgesics were all applied in a way that may not mimic real life clinical applications, so it is possible that patients may experience benefit if instructions were followed for all preparations.
Our study is not without limitations. The within group variability and the small sample size may have contributed to an insufficient power to detect significant changes using a 2-factor analysis. The data suggest that perhaps a larger sample size may have uncovered relationships that were not apparent with the power in this study. The within group differences only revealed a significant effect for change in PPT in the MuscleCare™ formulation, however a larger study is required to confirm if our failure to detect significant differences between group is the result of a type 2 error. In addition, although the analysis of these measurements in a working environment may allow for a more accurate assessment of the use of these preparations at reducing pain in an individual's day-to-day life, there may be some inherent limitations of not performing these analyses in a controlled environment. Lastly, we only assessed 1 product per individual, potentially introducing some between subjects variability, however we believed that there would be insufficient time between exposures to prevent contamination, thus. We do not believe that this significantly impacted our results as the test-retest method of pressure algometry has shown to be very reliable in many studies [19].
Pragmatically the topical preparations were all organized to provide the same dosage volume. This was done from a feasibility perspective to eliminate biases and maintain blinding. However, such preparations do not represent true life applications. It is possible that clinical or statistical differences may have been detected if all topical applications were applied according to the prescribed dosage and application parameters.
Further investigations should be completed with larger sample sizes and with measurements taken at varying times after the application of the products to confirm if other differences would be detected and to confirm the findings of this study. In addition, more information is required for a wider-range of individuals, including older and younger patients, and individuals with various musculoskeletal injuries. Such data may help to gain an understanding of the efficacy of non-medicinal topical analgesic products, such as MuscleCare™ in a greater range of chronic pain conditions.
This study demonstrates that a non-pharmacological topical analgesic (MuscleCare™ preparation without MSM) yielded the greatest increase in PPT post-treatment after seven minutes, demonstrating that an all-natural non-pharmacologic topical analgesic is at least as effective as some topical-NSAIDs (Voltaren®) when following the same procedures. Although the between group differences were not statistically significant, the within group differences may support the usage of non-pharmacological topical analgesics for the reduction of PPT associated with myofascial trigger points of the upper trapezius in sedentary office workers. Further research is needed to determine if these differences are observable across other musculoskeletal conditions, other timelines, and other patient populations.
Ethical approval was obtained from the Research Ethics Board at the University of Toronto. All participants provided written and informed consent prior to participating in the study.
This study contains no individual's personal information that would require consent to publish. Written informed consent was obtained from participants for publication of their results. The consent form is held by the author's and is available for review by the Editor-in-Chief.
All data generated or analysed during this study are included in this published article.
The authors declare that they have no competing interests.
This study was funded in full by Active and Innovation Inc.
RTS and JMG analyzed and interpreted the patient data and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
We would like to acknowledge Emily Vecchiarelli and Meghan Glibbery for their assistance with data collection and entry and all of the participants who agreed to participate in this study.