Background: Musculoskeletal disorders (MSKD) create a significant burden on individuals and society. Physical activity is an effective way of self-managing MSKD. Global health policy routinely recommends the instigation of management approaches to support people with MSKD to engage in physical activity. However, there is considerable heterogeneity in outcome measurement and establishing whether interventions are effective or not.
A way of addressing this is to develop a core outcome set (COS). This study aims to identify a COS for exercise and physical activity interventions for people with MSKD.
Methods: Guidance from the COMET initiative was followed. The study had two phases.
1) A systematic search of effectiveness studies investigating interventions that aim to increase physical activity levels in people with MSKD was conducted. All outcome concepts evaluated were extracted from the included studies.
2) Patients with MSKD and expert stakeholders then participated in an online and workshop-based consensus process. In accordance with other COS development studies, 70% agreement was required for a concept to be included in the final COS.
Results: Phase 1: 25 studies were identified from the systematic searches and 50 conceptually different outcomes were extracted.
Phase 2: 14 group members were recruited to the consensus phase. Function, Patient satisfaction, Physical activity, Quality of Life, Pain, Cost-effectiveness, Self-efficacy, Knowledge to plan future exercise, Utilisation of health services were the concepts that reached the 70% threshold for inclusion in the final COS.
Conclusions: This study provides a COS that could provide an outcome measurement strategy in interventions that aim to increase exercise and physical activity in people with MSKD.
Musculoskeletal diseases, Physical activity, Consensus, Review, Core outcome set
CINAHL: Cumulative Index to Nursing and Allied Health Literature; COMET: Core Outcome Measures in Effectiveness Trials; COS: Core Outcome Set; EPA: Exercise and Physical Activity; ERS: Exercise Referral Scheme; MEDLINE: Medical Literature Analysis and Retrieval System Online; MSKD: Musculoskeletal Disorder; OA: Osteoarthritis; PICO: Population, Intervention, Comparator, Outcomes; pMSKD: People Living with a Musculoskeletal Disorder; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RCT: Randomised Controlled Trial
Musculoskeletal disorders (MSKD), such as low back pain, arthritis, or osteoporosis, are the 2nd largest cause of years lived with disability [1]. They create a significant burden on individuals and health economies, with 28.4 million working days lost in 2019 due to MSKD [2]. The prevalence of MSKD is expected to continue to rise [3]. MSKD are a diverse group of disorders that have a range of pathophysiological causes but are linked anatomically [4] and by the similar pain and dysfunction they cause leading to physical, biological, psychological and social consequences for people living with MSKD (pMSKD) [5-7].
There are management strategies that are specific to each of the many MSKD to target specific structures or physiological processes, however a core management approach across MSKD is to support an increase in exercise and physical activity (EPA) levels [8,9]. EPA is recognised to be beneficial for pMSKD and to prevent onset also, however despite these potential benefits pMSKD are twice as likely to be inactive compared to those without MSKD [2]. This is due to a range of biological, psychological and social factors [10] and demonstrates the increased difficulty that pMSKD are faced with when trying to access EPA. To attempt to address this, EPA interventions are a commonplace management strategy [11,12], employed by health services, aiming to support pMSKD to become more active and reduce the burden of their MSKD whilst improving uptake and maintenance of EPA. A wide range of EPA interventions have been used to support pMSKD to engage in EPA. These include walking [13], self-management programmes [6], exercise referral schemes [14] and home based EPA interventions [15].
A key issue with EPA interventions for pMSKD is the lack of a defined approach to outcome measurement [10]. One systematic review investigating exercise and physical activity (EPA) interventions that were designed to improve EPA uptake in pMSKD found limited evidence that participants were more likely to increase their level of moderate intensity EPA compared to usual care [14]. One major limitation reported in this review and others was the inconsistency of outcomes used in interventional studies [14,16]. Further studies have reported this leads to difficulty in comparing or pooling data between studies due to heterogeneity of outcomes [17,18]. Consequently, this creates challenges for clinicians, healthcare organisations and policy makers when applying health care research in practice as it is difficult to understand if EPA interventions are effective and worth investment [19]. Improvements in policy for MSKD also advocate increased consumer-centred outcomes to support understanding of interventions that support pMSKD [11].
To reduce variation in outcome measurement and support improved consistent outcome measurement, it is recommended that a COS, or an agreed standardised collection of outcomes, be developed for EPA interventions [20]. The COS should represent the minimum that should be measured and reported in trials of interventions [19]. This will support benchmarking and comparison of similar services to reduce variation in delivery. The Core Outcome Measures in Effectiveness Trials (COMET) initiative provides guidance and resources to support COS development using a standardised approach [19] and the core outcome set-standards for reporting (COS-STAR) checklist provides guidance for reporting [21]. COS have been produced for related subject areas, such as for clinical trials for non-specific low back pain [18], standardized outcome reporting in low back pain [22], and rehabilitation in musculoskeletal disorders [23]. However, searches of the (COMET) database identified that no COS has been developed to measure the effectiveness of EPA interventions that support pMSKD.
Though there are similarities with other COS focused on MSK conditions, no other COS can be found that specifically support interventions that aim to improve EPA in people with MSKDs. The need to support EPA in pMSKDs is well documented. This study aims to produce a COS to support standardisation and understanding of what outcomes are important when supporting pMSKDs into EPA.
The COMET Handbook was followed to undertake this COS development [19], which also recommends to follow the COS-STAR checklist [21], which this study has done.
A two-phased COS development method was used. Phase 1 was a systematic search of the literature undertaken in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines [24] and the Cochrane handbook for systematic reviews of interventions [25]. All outcome concepts from effectiveness studies of EPA interventions that have a primary outcome of increasing EPA in pMSKD were sourced. Phase 2 was a consensus process undertaken in line with COMET guidelines [19] using expert patients and stakeholders to agree which concepts derived from the phase 1 systematic searches are most important and must be included in the final COS. The consensus phase consisted of an online stage, followed by a face-to-face consensus workshop.
A search strategy was undertaken in May 2017 to identify EPA intervention studies that had a primary outcome of increasing physical activity. The PICO (population, intervention, comparator, outcome) that provides the basis for the systematic search is presented in Table 1.
Table 1: PICO for systematic review. View Table 1
Five databases were searched in September 2017: MEDLINE (EBSCO), CINAHL (EBSCO), Cochrane (Wiley), SportDiscus (EBSCO), Web of Science (Thomson Reuters, now Clarivate). Titles, abstracts, and full-text articles were screened independently by two authors (AT, TP). Any non-concordance that could not be resolved between the two reviewers were discussed and a consensus reached; any outstanding disagreements were resolved using a third author (SM). Articles were included if they were a randomised controlled trial, included populations with MSKD, and a primary outcome measure to evaluate physical activity participation was used. Non-English studies, studies including participants < 18-years-old, studies with physical activity not as a primary outcome, and studies with no EPA interventions were excluded. Since the study team were only interested in identifying outcome measures used within included trials, quality assessment was not required.
A data extraction form, aligned with COMET guidance [19], was developed to capture essential information from included studies. Data extracted included study characteristics, outcome concepts and outcome measurement instruments utilised. The data extraction form was piloted by two authors (AT, TP) to test for consistency and utility. The outcome concepts used in each study were then extracted by one author (TP) and checked for accuracy by a second author (RM). Outcome concepts were reviewed by three authors (AT, SM, RM) for duplication, relevance, and definition to create a list of mutually exclusive outcome concepts. This list of outcome concepts was taken forward to phase 2.
Phase 2 was a consensus approach split into an online phase and a workshop phase, the aim of which was to agree which of the outcome concepts derived from phase 1 must be included in the final COS. This phase was conducted according to COMET guidance [19]. This guidance suggests that the selection of participants for consensus activities should be pragmatic but relate to the context of the COS being developed. Consequently, patients who had participated in an EPA intervention that aimed to increase their level of activity were invited to participate in this study, along with expert clinicians, researchers, health service managers, and commissioners. Ethics approval was sought and granted by the NHS Health Research Authority and Sheffield Hallam University.
An online questionnaire, participant information sheet, and an instructional video about the consensus process were produced (all were available in paper form at request for participants in the consensus process). The recruited participants were directed to online resources to access participant information and the questionnaire. The questionnaire asked participants to rate the importance of each outcome concept in relation to assessing the effectiveness of an EPA intervention. Outcome concepts were rated using a 9-point Likert scale that ranged from 1-9, with 1 = not important to 9 = extremely important. The questionnaire was electronically submitted and one author (RM) collated results.
Concept ranking following the voting results was undertaken between three members of the study team (AT, SM, RM). Concepts were placed into one of three groups: Consensus of high importance for inclusion in the COS, consensus of low importance for inclusion in the COS, or consensus not achieved. Ranking was based on judging the mean, median and mode of the total scores from voting of importance on a 1-9 Likert scale. One author (AT) had the final decision if non-concordance occurred between the three authors.
A consensus workshop was convened for all the participants who had participated in the virtual stage. The workshop participants were supplied with clear instructions as to the content and aims of the workshop. In accordance with COMET guidance this included requesting the group to consider if any further outcome concepts not derived from phase 1 should be included.
The voting and ranking from the online questionnaire were presented to workshop participants. Participants were then divided into three groups, this included a group of exclusively patients (to ensure patients did not feel intimidated by professionals), with the remainder of the participants divided into two groups. Following this, participants were invited to discuss those concepts that did not reach consensus during the online stage. Participants were then asked to vote electronically using 'yes' or 'no' responses as to whether each concept must be included in the final COS or not. All voting results were sent electronically and anonymously to the study team, where results were immediately collated and presented back to the workshop participants.
Participants were then reorganised into three mixed groups consisting of both patients (as it was clear they had confidence to voice their views) and healthcare professionals to ensure the broadest possible discussions took place in the next round of discussions. Participants were asked to vote for a second time on which of the outcome concepts must be in the final COS, using 'yes' or 'no' responses as to whether each concept must be included in the final COS or not. All voting results were immediately collated by the study team.
To be considered suitable for inclusion in the COS, 70% of participants had to agree that the concept must be included in the COS. Although it is an arbitrary figure, a 70% cut off has been used in the development of other COS [26], and COMET discuss a 70% threshold representing the majority in a consensus group [19]. The study team agreed that 70% would result in a manageable list of final concepts, whilst not being too stringent to exclude key outcomes [19].
Following removal of duplicates, 3500 records were screened, leading to 48 full text studies being retrieved, of which 23 were excluded resulting in 25 studies being included for data extraction (Figure 1).
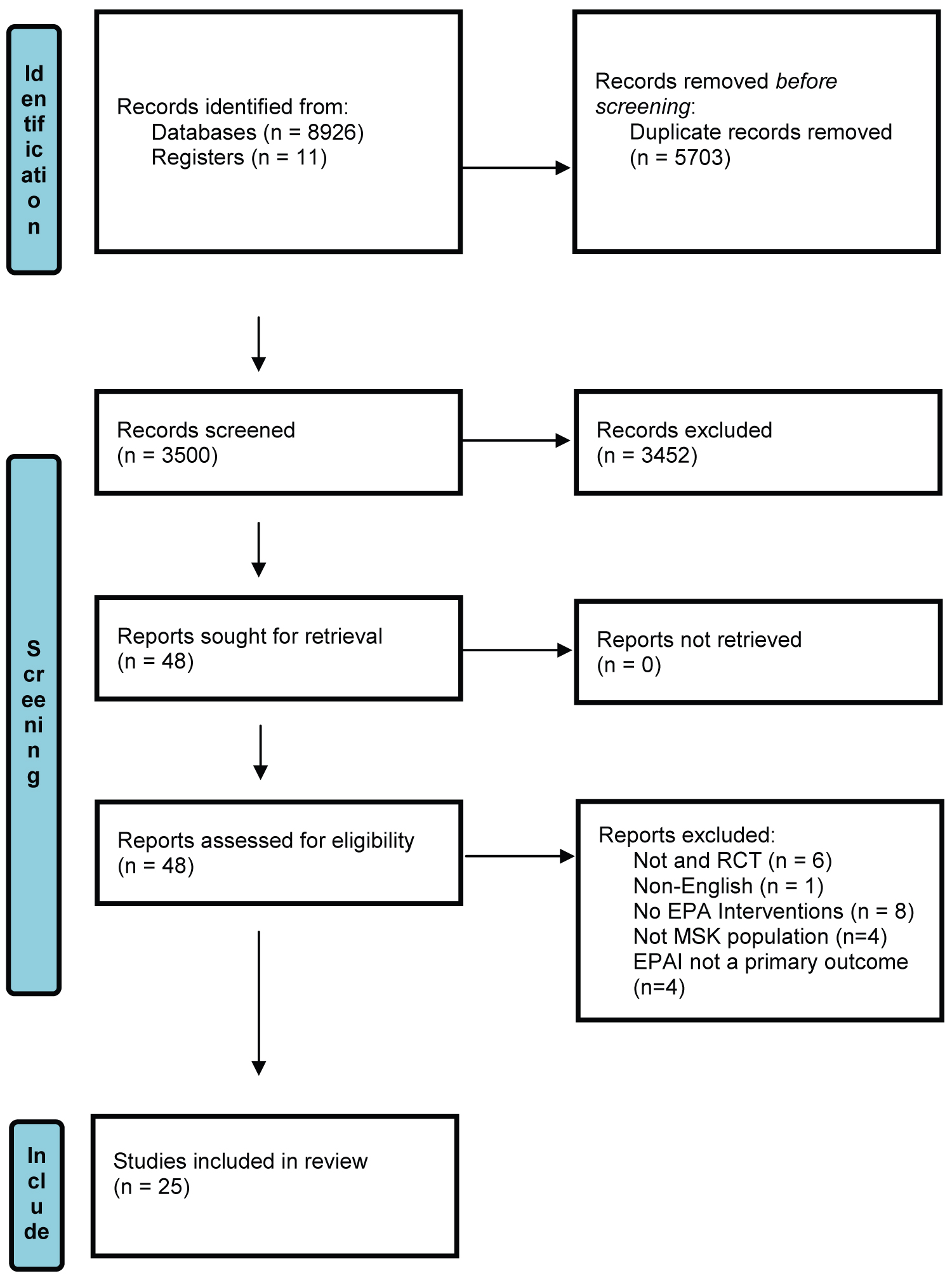 Figure 1: PRISMA flowchart of study selection [24].
View Figure 1
Figure 1: PRISMA flowchart of study selection [24].
View Figure 1
The 25 papers (Table 2) extracted exhibited wide-ranging properties. Populations studied within these studies were: Knee osteoarthritis (OA) (n = 8) [27-34], Low back pain (n = 3) [35-37], after total knee or hip replacement (n = 3) [38-40], sedentary populations with MSKDs (n = 2) [41,42], Hip OA (n = 2) [43,44], Neck and shoulder pain (n = 1) [45], Ankle sprain (n = 1) [46], after primary lumbar disc surgery (n = 1) [47], shoulder impingement syndrome (n = 1) [48], depression, overweight or obese (n = 1) [49], fibromyalgia (n = 1) [50], Lumbar radicular pain (n = 1) [51].
Table 2: Extracted study characteristics and outcome concepts. View Table 2
Studies were predominantly hospital-based physiotherapy clinics (n = 15) [27,30-32,34-37,39,40,43,44,47,48,51]. Others were set in leisure centres (n = 2) [41,42], Hydrotherapy pools (n = 2) [33,38], parks and forests (n = 2) [49,50], adult day centres (n = 1) [28], universities (n = 1) [29], work place (n = 1) [45], accident and emergency department (n = 1) [46].
Studies were mainly from western regions of the globe: Europe (excluding Scandinavia) (n = 12) [28,30,34,36,38,40-43,46,48,49], Scandinavia (n = 5) [39,44,45,47,50], Australasia (n = 3) [27,31,37], United States (n = 2) [29,51], Asia (n = 2) [33,35], Brazil (n = 1) [32].
Concepts extracted from the included studies were found to have considerable levels of duplication as some concepts such as pain, function and patient experience were common outcomes. In total, 50 different outcome concepts were extracted from the included studies (Table 3). All studies had physical activity as a primary outcome.
Table 3: Concepts extracted from the literature, ranked following the online vote. View Table 3
Sixteen participants were recruited to the consensus phase. This included expert patients (n = 7) with MSKD who had participated in an EPA intervention, senior physiotherapists (n = 3) with extensive knowledge of MSKD and EPA interventions, researchers (n = 2) with experience in this field, a healthcare manager (n = 1), a healthcare commissioner (n = 1), a public health representative (n = 1), and an exercise professional (n = 1) specialising in supporting people with MSKD. All 16 participants completed the online questionnaire.
Table 3 shows the 50 outcome concepts after participants completed the virtual phase questionnaire. A pragmatic balance between mean, median and mode data was used to guide ranking. Ranking was undertaken to guide discussion in the workshop, i.e., to identify which concepts that had not reached a consensus of high or low importance in the COS. These findings were taken forward to the consensus workshop.
One patient and one public health representative were unable to attend the workshop. All other participants attended resulting in 14 participants. Initially participants were asked if there were any outcome concepts they would add to the list. One additional concept was identified by the group prior to the first voting round, this was: Referral/utilisation of health care services.
Following small group discussions, participants were asked to vote (yes or no) which of the 51 outcome concepts should be included in the final COS. Following this, groups were reorganised, and further discussion of key concepts took place. A second additional outcome concept was identified at this stage: 'Knowledge to plan further exercise'. This concept was discussed and added to the concept list. Participants were asked to vote (yes or no) for a second time for which of the 52 outcome concepts must be in the COS.
Table 4 presents the overall ranking of the 52 concepts after two rounds of voting. Nine outcome concepts reached a 70% threshold after the second voting round. The remaining 43 outcome concepts were ranked below the agreed 70% consensus level required for inclusion in the COS. The 9 concepts with 70% or more consensus were presented to and ratified by the group as the final COS.
Table 4: Final outcome concept rankings. View Table 4
As far as we are aware, this is the first study to define a COS for EPA interventions to support pMSKD. The systematic searches identified 50 independent concepts that were used for investigating the effectiveness of EPA interventions in RCTs. A further two concepts were added to this list during the consensus workshop stage. Following consideration of these 52 outcome concepts, workshop participants arrived at a consensus that nine must be included in the COS. These were: function, patient satisfaction, physical activity, quality of life, pain, cost-effectiveness/healthcare costs, self-efficacy, knowledge to plan future exercise, referral/utilisation of health services.
The final COS contains concepts that are frequently used in studies of effectiveness of EPA interventions in pMSKD, with 6 out of 8 of the most frequently used concepts (Physical activity, pain, function, quality of life, patient satisfaction, cost-effectiveness) extracted included in the COS. Physical activity was used as an outcome concept in all the studies as its inclusion in effectiveness studies was a pre-requisite in the systematic search phase of this study.
The inclusion of two concepts ('self-efficacy' and 'knowledge to plan future exercise') demonstrates the need for behavioural change concepts to be measured as part of EPA interventions. Self-efficacy is defined as 'people's beliefs about their capabilities to produce designated levels of performance that exercise influence over events that affect their lives' [52]. The need for integration of self-efficacy theory into EPA interventions has been advocated for some years [53]. However, how this might be achieved is not well understood as there could be numerous internal and external factors that affect self-efficacy [53]. Positive insights for integrating self-efficacy are available. In a recent systematic review and meta-analysis [54] it was found that EPA interventions participants were twice as likely to increase EPA in interventions that prioritised self-efficacy. Although self-efficacy was included in the final COS with a strong consensus (78.6%) it was only extracted twice from the studies derived from the phase 1 systematic searches. This perhaps indicates a disconnect between the design of interventions in the literature compared to the consensus group in this study that included patients.
'Knowledge to plan future exercise' was one of the two concepts derived by the consensus workshop participants. This concept was felt to be important as it represents whether EPA intervention participants have gained sufficient knowledge to plan and adhere to ongoing EPA independently. This is supported in behavioural change literature with knowledge and gaining information thought to be a key part of behavioural change interventions to support ongoing adherence [55,56]. Both self-efficacy and knowledge acquisition are core parts of behavioural change models such as the Transtheoretical Behavioural change model (TTM) [57], the WHO behavioural change model to support adherence [58], and also the behavioural change wheel [59].
The other concept to be derived by the consensus workshop participants was referral/utilisation of healthcare services. This relates to services that pMSKD access related to their condition, and if EPA interventions can change an individual's level of healthcare utilisation. Although the systematic searches in phase 1 of this study did not provide studies specifically reporting on healthcare utilisation, this seems to be a regularly used outcome in studies about MSKD [60-62]. Cost-effectiveness/healthcare costs were also included within the final COS. The consensus workshop participants deemed referral/utilisation of healthcare services' to be distinct from this. However, studies investigating interventions for MSKD have included utilisation of healthcare services to establish costs or cost effectiveness of services [63,64]. When applying this COS in practice, consideration of combining these two concepts into one could be considered.
Although it appears no other studies have developed a COS for supporting EPA interventions in pMSKD, COS has been developed in similar populations or interventions. Core outcome domains have been produced for MSKDs. One such being a core outcome domain development for clinical trials for non-specific low back pain [18]. Two hundred and Eighty global experts participated in the Delphi study including 15 patients, with a steering group of low back pain experts defining outcome concepts and having a final say on the COS. Four outcome domains - Physical functioning, pain intensity, health-related quality of life and number of deaths - resulted. A COS for exercise interventions for people living with multiple sclerosis have been produced [65]. An international consensus meeting was used to identify the COS from the International Classification of Functioning disability and health core sets [66]. Both these COS development studies show how derivation of a COS can be done by similar but nuanced methodology.
COMET [19] recommends that the size and members of expert consensus panels should be pragmatically considered with reference to the subject in question. The size of the consensus group (n = 14) in this study is low compared to other COS consensus groups. In some COS development studies international panels have been used [18,67]. However, the range of participants in this study was representative of the interested parties related to EPA interventions for pMSKD. It is possible that this COS could be repeated with a larger group or international participants which may provide more confidence in the COS. However, the COS was derived from a list of outcome concepts that had been used in a global range of studies, providing further basis for confidence in the COS.
Strengths of this study are the rigorous systematic searches of the literature undertaken in accordance the Cochrane Handbook for Systematic Reviews of Interventions [25] and PRISMA guidance [24] to derive the list of outcome concepts to use in the consensus phases. The study followed COMET methodology [19], and derived outcome concepts from a representative group of experts that included - in accordance with COMET initiative recommendations - expert patients to identify the critically important outcome concepts that must be used in a final COS. In this study grey literature searches were not undertaken, and only papers published in the English language were used, meaning there is a possibility of relevant information not having been retrieved.
Two concepts were identified on the day of the consensus workshop (referral/utilisation of health services, and knowledge to plan future exercises) and both concepts were included in the final COS. It is acknowledged that a possibility of bias towards these concepts by the consensus group took place, or they were the subject of more discussion than other concepts, which led to their inclusion. However, requesting consensus group participants to derive concepts on the day of the meeting is in accordance with COMET methodology [19].
This COS is helpful in two ways: It creates an opportunity for future researchers investigating EPA interventions for MSKD to understand how they could standardise their data collection in ways that are meaningful to relevant stakeholder groups. The fundamental lack of standardisation has been identified as one of the key reasons for not being able to fully understand the effectiveness of EPA interventions for pMSKD. Secondly, EPA interventions are widely used to support pMSKD [8,12] and the COS will allow clinical services to start to consider and standardise their outcomes for patients. Researchers and clinicians must still choose valid and reliable measures that reflect the COS to evaluate the effectiveness of their EPA intervention. This provides some flexibility to either choose measures for interventions that support any pMSKD or for interventions supporting specific MSKD such as for LBP or shoulder problems. The standardisation and benchmarking that this COS provides creates the opportunity to compare similar interventions both nationally and internationally and in doing so enable understanding of where variation exists between EPA interventions for pMSKD.
The benefits of EPA interventions to enable pMSKD to self-manage their condition are well known, EPA interventions are widely used to facilitate this transition. This study utilised PRISMA [68], Cochrane [25] and COMET [19] guidelines, patients and key stakeholders to agree nine outcome concepts that should be used in evaluations of the effectiveness of EPA interventions for pMSKD. The study had a comparatively low number in the consensus group, though there was representation from a range of stakeholder groups and patients. The study has produced a COS which could be used in research investigating EPA interventions. In addition, the COS supports clinical services to evaluate the effectiveness of their EPA interventions and develop interventions in ways that optimise these important outcome concepts for relevant stakeholders.
Ethics approval was obtained from the NHS Health Research Authority, East Midland- Leicester South Research ethics committee. IRAS project ID: 225119: REC Reference 17/EM/0342.
Ethics permission was also obtained from Sheffield Hallam University Ref: 2017-8/HWB-HSC-02.
Informed consent was obtained from all subjects.
The authors declare that they have no competing interests.
We would like to thank the consensus group participants who gave up their time to be involved. This is especially to the patient representatives. We would also like to thank Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust Research team for their support and guidance.
It is with great sadness to say that one of our study group and listed author Deb Harrop passed away before submission of this manuscript to BMC Musculoskeletal Disorders. Deb's contribution was essential in terms of literature and synthesis but also in supporting literature synthesis skills in the research group.
AT undertook literature synthesis, recruitment, consensus group organisation and write up. RM undertook consensus group design, literature extraction, data gathering and participated in write up. TP undertook literature synthesis and data extraction. DH designed search strategy and provided systematic search expertise. MK undertook participant recruitment and workshop design. SM Provided consensus group expertise, and design, and contributed to write up. All authors read and approved the manuscript.
The study was funded by a joint grant from Doncaster and Bassetlaw Teaching Hospitals NHS Foundation Trust and The Yorkshire and Humber Collaboration for Leadership in Applied Healthcare research and care.