The purpose of this study was to assess the formation of biogenic amines (BA) during chilled storage (60 days) in cooked sausages made without added nitrite. Various combinations of ingredients celery, sodium lactate, carmine, orange dietary fibre and vitamins C and E - were used as substitutes for nitrite. BA levels were low irrespective of formulation, the largest variations between products were found in the case of cadaverine. The lowest (P < 0.05) cadaverine content was registered in samples with 0.1% added vitamin C, which also registered the lowest levels of enterobacteria and residual nitrite. The process offers health and safety benefits for consumers, and this strategy could thus be a good alternative to reduce and/or eliminate added nitrite in sausages and maintain product quality and safety (including BA formation) without this posing any risk to healthy consumers.
Biogenic Amine, Cooked Sausage, Nitrite, Quality, Safety, Risk, Health
Sodium and potassium nitrates and sodium and potassium nitrites are commonly used in meat curing because they stabilize red meat color, inhibit some spoilage and food poisoning anaerobic microorganisms (mainly Clostridium botulinum), delay oxidative rancidity and contribute to flavor development. Nitrates and nitrites are considered to be essential food preservatives [1-6], but their use as additives has generally aroused controversy because of their potential effects on human health. Nitrates and nitrites are profoundly implicated in physiological systems needed for survival. However, nitrites can react with secondary amines to form nitrosamines, which are recognized as having carcinogenic effects [7,8].
In recent decades the meat industry has modified its technologies so as to reduce the use of additives, including nitrates and nitrites, during meat production to achieve safer and healthier products [3]. Various ingredients have been used to replace added nitrites and ensure that their functions are performed in the end product while maintaining quality [2,9-15]. To that end our group [15] recently examined the effect of total replacement of added nitrite on the quality characteristics (technological, sensory and microbiological properties) of hot dog sausages during chilled storage. Different combinations of celery, carmine, sodium lactate, orange dietary fiber and vitamins C and E were used as nitrite replacers. Sensory evaluation showed that all samples presented similar scores for acceptability, with similar color values irrespective of formulation. It was also observed that the inclusion of vitamin C in the formulation limited microbial growth and improved the antioxidant stability of the meat system when compared with other reformulated samples. Additionally, the reformulated samples were microbiologically safer than samples containing added nitrite. This study demonstrated that the tested strategies for reformulation of hot dog sausage without added nitrite based on combinations of complementary ingredients, including vitamin C, can be used to produce healthier and safer products while overcoming the limitations associated with the absence of nitrite in meat products such as hot dog sausages.
However, that study [15] did not address an important additional safety-related aspect, namely the formation of biogenic amines (BAs) in new products reformulated using a global strategy for replacement of added chemical nitrites. In fact various biogenic amines are themselves toxic (chiefly tyramine and histamine) and many of them (mainly secondary types, putrescine and cadaverine) can generate nitrosamines in the presence of nitrites [8] as noted above.
BAs are formed mainly by decarboxylation of free amino acids (FAAs) through the action of decarboxylase enzymes, their formation is influenced by a number of factors such as: FAA content and availability, microorganisms capable of producing decarboxylases, the nature of the medium; processing, and storage conditions. All these factors are interdependent and act together in various combinations that affect microbial growth and hence determine the presence and the activity of substrate (FAA) and enzyme [16]. Studies on the formation of BAs in nnitrite-reformulation of meat products are very important since reformulation produces some changes in the matrix with potential consequences for BA formation.
The concentration of amines is much lower in cooked products than in others such as raw cured products, as cooking eliminates most of the microbial load [16-18]. However, it is important to note that the cooking will not eliminate any biogenic amines that have already formed, as these are heat-resistant. The biogenic amines present in both non-meat ingredients (such as garlic or green vegetables) and raw meat therefore enter the pool of final products [16]. Moreover, these reformulation ingredients will influence amine formation during storage; this will also affect microbial growth, one of the main factors in biogenic amine formation. Then again, long-term chilled storage of such products, generally under vacuum, also affects microbial growth, mainly of lactic acid bacteria [15], and hence will affect the final concentration of biogenic amines. It is therefore essential to understand how biogenic amine formation is affected by the reformulation process in order to assess their potential presence in these meat products.
As far as the authors know little information has been published on the question of how a global strategy for producing a cooked meat product (sausage) without added nitrite affects biogenic amine formation. Therefore, following up on previous reports [15], the aim of this study was to evaluate how a cooked sausage reformulation process using a combination of ingredients (celery, sodium lactate, orange dry fiber and, carmine, vitamin C and vitamin E) as a global strategy for replacing added chemical nitrites in these products affects the formation of BAs during processing and chilling storage. This approach will surely help to provide safer products for consumers, which is especially important when reformulating processes entail changes in the presence of compounds with health implications.
The cooked (hot dog) sausages used in this study were as reported by Ruiz-Capillas, et al. [15]. Briefly, fresh beef (flank steak and shank meat) (with 19.98 ± 0.43 and 13.49 ± 1.46 protein and fat respectively) was obtained from a local meat market, ground with a 0.4 cm plate (Mainca, Granollers, Spain), frozen and stored at -20 °C until used within 2 weeks. Five different sausages were formulated with 50 % meat and a combination of ingredients as described in the table 1. A control sample (Control) was prepared traditionally with added chemical nitrite (120 mg/kg). The four different reformulated samples (C5E5, C0E10, C10E0, C10E10) were prepared without nitrite and with various combinations of celery (1%), sodium lactate (3%), carmine (0.05%) and orange dietary fibre (1%) and different combinations of vitamins C and E depending on the sample, sample C5E5 with 0.05% vitamin C and 0.05% vitamin E, sample C0E10 with 0.0 % vitamin C and 0.1% vitamin E, sample C10E0 with 0.1% vitamin C and 0% vitamin E, and sample C10E10 with 0.1% vitamin C and 0.1% vitamin E. The following ingredients were also added to all samples: 3.8% starch, 0.5% flavouring, 1.5% garlic powder, 1.9% egg white, 17% sunflower oil, 1.2% milk powder, 1.4% sodium chloride, 0.46% sodium tripolyphosphate.
Table 1: Formulation (%) of different cooked sausage. View Table 1
Raw meat (previously thawed at 2 ± 1 °C for 18 h) was comminuted and homogenized and combined with the rest of the ingredients (Table 1) in a chilled cutter (2 °C) (Stephan Model UM5 Universal, Stephan u. Sohne, Hameln, Germany). Sausages were stuffed into 20 mm diameter Nojax cellulose casings (Viscase S.A., Bagnold Cedex, France) and heat processed in an Eller smokehouse (model Unimatic 1000, Micro 40, Eller, Merano, Italy) until the core of the product reached 72 °C. After cooling, the sausages were vacuum packed (Cryovac1 BB3050, Madrid, Spain) and stored at 2 °C (± 1 °C) for analysis.
For each sample, 10 g (in replicate) was taken and placed in a sterile plastic bag (Sterilin, Stone, Staffordshire, UK) with 90 ml of peptone water (0.1%). After 1 min in a stomacher blender (Colworth 400, Seward, London, UK), appropriate decimal dilutions were pour-plated on the following media: Plate Count Agar (PCA) (Merck, Germany) for total viable count (30 °C for 72 h) and De Man, Rogosa, Sharpe Agar (MRS) (Merck, Germany) for lactic acid bacteria (LAB) (30 °C for 3-5 days) [15]. The results were expressed as logarithms of colony forming units per gram (Log cfu/g).
Tyramine, phenyl ethylamine, histamine, putrescine, cadaverine, agmatine, spermidine and spermine were determined (at the beginning and at the end of storage) in an extract prepared by blending 25 g of each sample with 50 ml of 7.5% trichloroacetic acid in an Ultra turrax homogenizer (IKA-Werke, Janke, & Kunkel, Staufen, Germany) (20,000 rpm, 3 min) and centrifuged at 5000 g for 15 min at 4 °C in a desktop centrifuge (Sorvall RTB6000B, DuPont, USA). The supernatants were filtered through a 0.45 μm Millipore filter, and 10 μl of this filtrate was injected into an HPLC model 1022 (Perkin Elmer) with a Pickering PCX 3100 post-column system (Pickering Laboratories, Mountain View, Ca, USA) following the methodology of Triki, et al. [17]. The results are averages of at least 2 determinations from two extractions per sample.
Two-way analyses of variance (ANOVA) were carried out to evaluate the statistical significance (P < 0.05) of the formulation and storage time, using the general linear model (GLM) procedure of SPSS Statistics (v.19, IBM SPSS Inc., Chicago, IL). Formulation and storage time were assigned as fixed effects and replicate as random effect. Least squares differences were used for comparison of mean values between treatments and Tukey's HSD test to identify significant differences (P < 0.05) between formulations and storage time.
A previous paper [15] reported an evaluation (technological, sensory and microbiological properties), during chilled storage, of different characteristics of hot dog sausages as analysed in this study. These sausages presented the typical characteristics of this kind of meat products. The proximate composition in terms of protein (around 13%), fat (from 23.50% to 24.12%) and moisture (from 55.43% to 57.68%) contents was similar in all the formulated products (Table 1) [15]. The main differences among them were the result of addition of some ingredients (celery, sodium lactate, orange dietary fibre, carmine, vitamin C and vitamin E), as a global strategy for replacing added chemical nitrites in these products.
The following section discusses aspects relating to the formation of biogenic amines and some considerations on shelf-life and safety characteristics. Some aspects of product characteristics (mainly residual nitrite and microbiological behaviour) need to be considered in order to understand the formation of BAs in this type of meat matrices. This is essential to gain a clearer understanding of safety in relation to the formulation of these products.
Biogenic amines were affected by storage time and reformulation of the cooked sausage (Figure 1, Figure 2 and Figure3). Tryptamine and agmatine were not detected. Histamine levels were also very low (< 1 mg/kg over storage), although lower in the reformulated samples without added nitrite (< 0.30 mg/kg) than in the control sample (0.85 mg/kg). Phenyl ethylamine was also detected at levels < 1.5 mg/kg, with the lowest (P < 0.05) in sample C10E0 (Figure 1). These results are comparable to reports by other authors in similar cooked products [18,19]. Note that products of this kind contain lower BA concentrations than fermented products [16,20,21]. This is because as non-heated products, the latter undergo fermentation and ripening, which promote microbial growth and hence biogenic amine production. In cooked products the microbial load is very much reduced, severely limiting the production of biogenic amines.
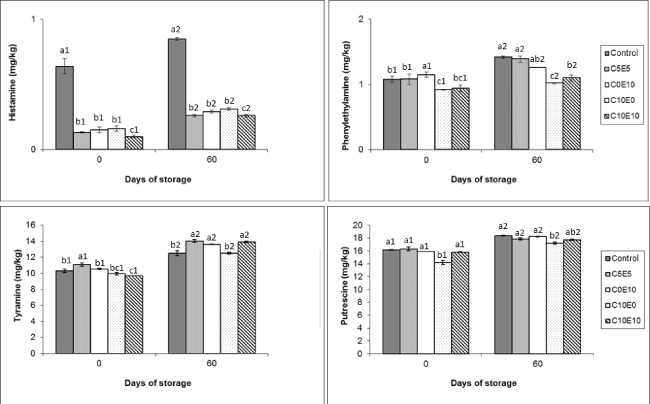 Figure 1: Biogenic amines (hismine, phenyilethylamine, tyramine and putrescine) levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 1
Figure 1: Biogenic amines (hismine, phenyilethylamine, tyramine and putrescine) levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 1
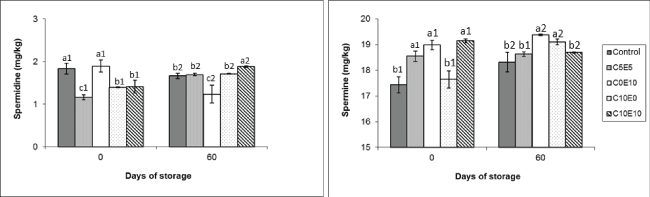 Figure 2: Biogenic amines (spermide and spermine) levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 2
Figure 2: Biogenic amines (spermide and spermine) levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 2
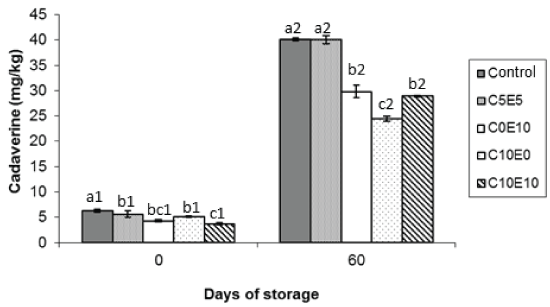 Figure 3: Cadaverine levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 3
Figure 3: Cadaverine levels (mg/kg) of different cooked sausage samples chilling storage. Data were expressed as mean ± SD. Different letters (a,b,c) in the same days of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 3
BA formation is closely associated with microbiological counts. In this regard initial microorganism levels were very low in all samples, with and without nitrite replacer, in terms of both total viable counts (TVC) (between 2.38 and 2.92 Log cfu/g) and lactic acid bacteria (LAB) (Figure 4) and Enterobacteriaceae, which were lower than 1 Log cfu/g [15]. This helps to explain the initial levels of tyramine and putrescine, around 9-11 mg/kg and 14-16 mg/kg respectively, with no great differences between the reformulated and control samples except in putrescine of sample C10E0 (Figure 1). These amines registered higher levels than observed by other authors in cooked products [18,19]. This could be due to the presence of added ingredients such as garlic, which may contribute to the total amine pool. On the other hand, it is also the case that the type of meat used (beef) presents a different amine profile from pork sausage [18,22]. This can be seen more clearly in the spermidine and spermine profile (Figure 2), showing the characteristic physiological biogenic amines, whose presence depends on the type of meat used in the reformulation. The profile of these physiological amines found in this experiment was similar to that reported by other authors in sausages made with beef [22].
 Figure 4: Total viable count (TVC) and Lactic acid bacteria (Log cfu/g) of different cooked sausages samples during chilling storage (Adapted from [19]). Different letters (a,b,c) in the same day of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 4
Figure 4: Total viable count (TVC) and Lactic acid bacteria (Log cfu/g) of different cooked sausages samples during chilling storage (Adapted from [19]). Different letters (a,b,c) in the same day of storage and different numbers (1,2) in the same type of samples indicate significant differences (P < 0.05).
View Figure 4
Increasing biogenic amine contents during chilled storage have been widely reported, again in association with microbial growth [16]. In the samples reformulated with nitrite replacers, the evolution of the microbiota in the course of storage was very similar in terms of TVC and LAB (Figure 4), considering that LAB were the predominant flora in the TVCs [15]. In these same conditions the latter authors reported that microorganism levels remained low, with no significant changes, up to day 20 of storage. Thereafter levels increased significantly in all lots up to day 40 and up to day 60 of storage (Figure 4). The control (C) registered TVC and LAB levels in excess of 6 Log cfu/g at day 40 of storage, while levels in the lots reformulated with the combination of nitrite replacers were significantly lower (Figure 4), indicating a considerable increase in their shelf life [15] all this despite the fact that the decrease of residual nitrite over the storage period was greater in the control than in the lots reformulated without added nitrite. In this control lot, the level of residual nitrite remained higher (37.06 mg/kg) than in the lots reformulated without added chemical nitrite (13-25 mg/kg) through to the end of storage [15]. In the given conditions it was not possible to establish a clear link between TVC and LAB growth and the production of biogenic amines, since tyramine and prutrescine levels did not differ significantly in the lots reformulated without added nitrite or in the control at the end of storage (Figure 1). This could have been because the formation of biogenic amines lags behind microbial growth to some extent, as reported in other studies [22]. Although tyramine is mainly produced by lactic acid bacteria, in this study no clear relation was observed between amine production and growth of lactic acid bacteria (Figure 4). This may have been due also to the growth of certain strains with greater amino acid decarboxylase capacity. Within the same species, the presence, activity and specificity of decarboxylases are strain-specific, as reported in other studies [22,23].
The most significant changes in the concentration of biogenic amines at the end of storage were observed in cadaverine (Figure 3). While the initial levels of this amine ranged between 3.7 and 6.0 mg/kg, higher (P < 0.05) in the control sample, by the end of storage the levels of cadaverine in the control sample had reached around 40 mg/kg, similar (P > 0.05) to C5E5 sample, without difference (P > 0.05) between the two samples. Significant increases in cadaverine levels have also been reported in meat batters during chilling storage [24], although the final amine levels were lower than registered in the present study, possibly because of the difference in the product reformulation. Also, cadaverine production has been linked to growth of enterobacteria in beef [25,26]. This could help explain the findings in the present study, in that the control and C5E5 lots registered the highest enterobacteria levels at the end of storage and the highest levels of residual nitrite [15]. The other lots reformulated without nitrite (C0E10, C10E0 and C10E10) contained lower levels (P < 0.05) of cadaverine, especially sample C10E0 where levels (24.50 mg/kg) were lowest (P < 0.05) at the end of storage (Figure 4). Note that the same sample additionally registered low enterobacteria levels < 1 Log cfu/g and the lowest concentration of residual nitrite [15]. This behaviour may have been due to the antimicrobial effect of vitamin C, especially when it is used alone, not combined with vitamin E. Vitamin C has been reported to exert an antimicrobial effect on two of the ingredients used in the nitrite replacer combination, sodium lactate and celery [27], but the difference between these lots lies mainly in the levels of vitamins C and E used (Table 1). Moreover, the antimicrobial effect of vitamin C is greater the larger the quantity used (Figure 2). And this could further help to explain why the tyramine levels registered in C10E0 were also the lowest (P < 0.05) of all the reformulated samples, but were similar (P > 0.05) to the levels in the control sample at the end of storage (Figure 1).
The current legal limits on histamine contents in products of this kind stand at 50 mg/kg in the USA (FDA 2011) [28] and 100 mg/kg in the EU (EFSA 2011) [20]. It can therefore be assumed that the very low histamine levels considered in this study pose no risk to healthy consumers (Figure 1). Then again, although amines like cadaverine and putrescine are known to enhance the toxic effect of histamine and tyramine, the levels of these were also low when compared for instance to fermented products [16,20].
Cooked sausages reformulated with a combination of nitrite-replacing ingredients registered lower biogenic amine levels than sausages containing the normal amount of added nitrite. The most effective combination of nitrite replacers in terms of reduced microbial load, amine formation and residual nitrite was the one including vitamin C. Its effect was proportional to the levels of vitamin C used.
These results suggest that the use of added nitrite-free substitutes in cooked sausages will not pose a risk to healthy consumers. Biogenic amine levels remained below 20 mg/kg except for cadaverine, which registered around 40 mg/kg. From the standpoint of food safety, then, this combination of ingredients seems to be suitable as a substitute for chemical nitrites.
This research was supported by projects, AGL 2011-29644-C02-01 of the Plan Nacional de Investigación Científica, Desarrolloe Innovación Tecnológica (I+D+I) Ministerio de Ciencia y Tecnología and Intramural projects CSIC: 201470E073 and Comunidad de Madrid project MEDGAN: S2013/ABI-2913.