Fistuloclysis and chyme reinfusion is a complex method of feeding that has not been widely used at our tertiary hospital. This is a case study of a 61-year-old male admitted with post-operative complications to our facility for consideration of surgical repair. This case study aims to describe the challenges faced by the multidisciplinary team when implementing fistuloclysis and chyme reinfusion while attempting to achieve nutritional goals and achieve positive patient outcomes. We also describe some of the barriers, reflections and learnings from our experience of these complex methods of nutrition support.
Fistuloclysis, Chyme reinfusion, Nutrition support
Enterocutaneous fistula is an abnormal connection that forms between the gastrointestinal tract and the skin or the atmosphere [1] with up to 75-80% of fistulas occurring as a post-operative complication [2]. When this complication occurs, chyme, a combination of digestive secretions that facilitate enzymatic breakdown of food that facilitates intestinal absorption, is lost through the fistula. Enterocutaneous fistula output can be high, resulting in dehydration, renal impairment, electrolyte disturbances, malnutrition and intestinal failure [1-3]. With high fistula losses, parenteral nutrition remains the main form of nutrition support to ameliorate fluid and nutrient losses. However, when there's viable small bowel distal to the fistula with enough absorptive capacity, chyme reinfusion may present an appropriate alternative or adjunct to parenteral nutrition [1].
Chyme reinfusion was initially described and developed by Dr Etienne Levy in the 1970s, allowing endocrine and exocrine secretions for nutrient absorption within the utilized gastrointestinal tract [4]. The advantages of chyme reinfusion include nutrition improvement, weight gain, improvement of liver profiles and volume of fistula output [5]. Although there are several reports of chyme reinfusion through double enterostomies [3,5], the authors are not aware of any case reports describing the use fistuloclysis and chyme reinfusion in a person with multiple fistulae.
A 61-year-old male presented to his local hospital with complicated diverticulitis, with a colonic perforation at the splenic flexure deemed to require emergency surgery. This was managed with an operative washout, extended right hemicolectomy and primary ileocolic anastomosis on the 14 February 2015. The patient was discharged on day nine postoperatively but was then readmitted six days later with a wound dehiscence and abdominal sepsis, due to anastomotic breakdown. He was returned to theatre for open washout, further resection of ileum and colon, with the formation of an ileostomy, and a colonic mucous fistula. An open abdomen was maintained to facilitate ongoing abdominal washouts. He was admitted to the intensive care unit and commenced on parenteral nutrition (providing 8200 kilojoules (kJ) and 90 grams (g) protein per day once rate was achieved). He was returned to theatre eight days later for a repeat laparotomy where three perforations in the proximal jejunum were identified. Approximately 20 days after the initial surgery, multiple enterocutaneous fistulae developed as a postoperative complication. We then resumed care when he was transferred to our tertiary metropolitan hospital under the specialty Colorectal Surgery team for ongoing management of his complicated fistulae and intestinal failure (Figure 1).
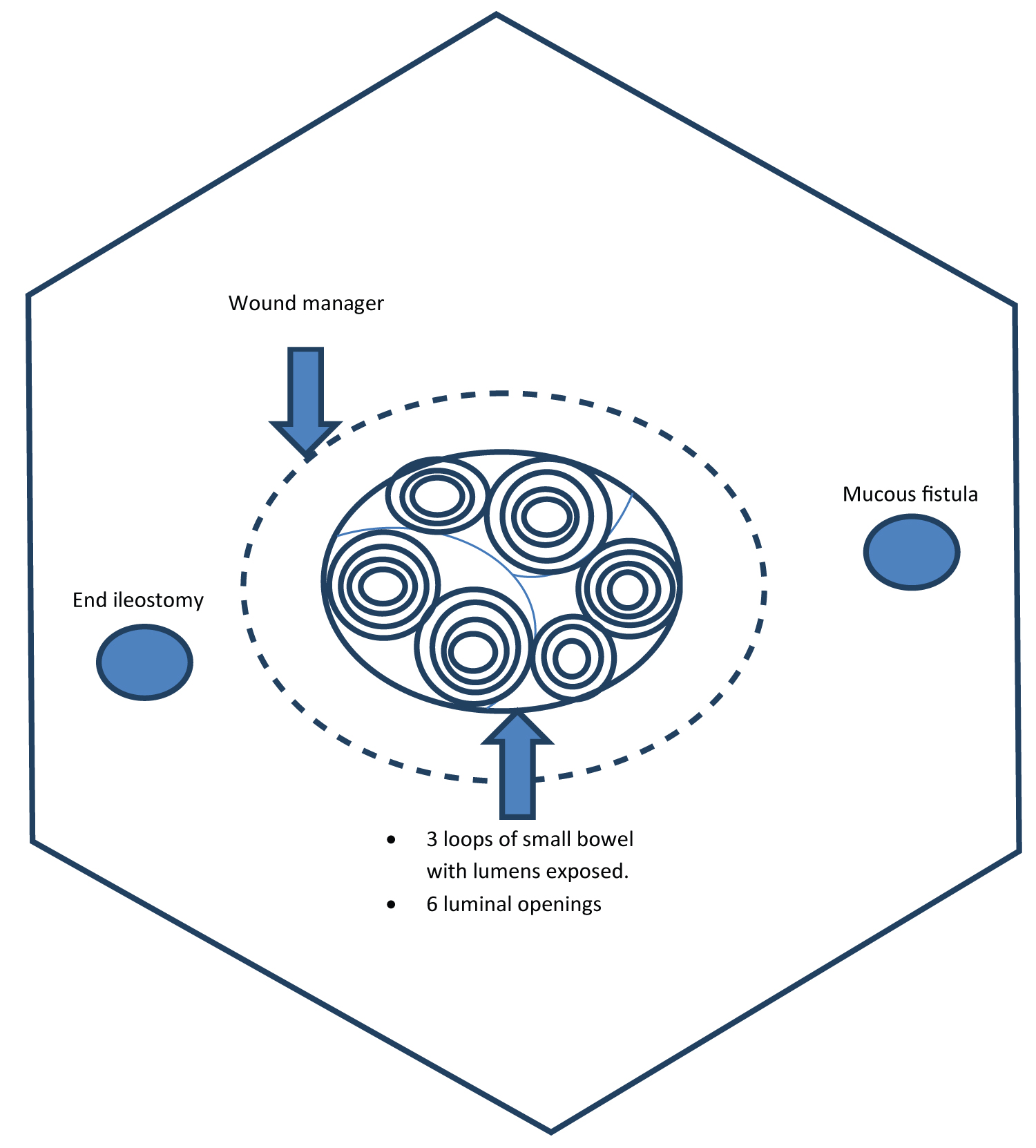 Figure 1: Anatomy of open abdomen.
View Figure 1
Figure 1: Anatomy of open abdomen.
View Figure 1
On admission to our tertiary center on the 30th March 2015, he had a tracheostomy in situ and continuing parenteral nutrition with approximately 200 milliliters (mL) daily fistula output from the open abdomen. On admission, he was commenced on the hospital's standard parenteral nutrition formulation providing 9500 kJ and 90 g protein per day and his tracheostomy was removed the next day. Speech pathology confirmed a safe swallow and a trial commenced of semi-elemental enteral trophic feeding at 10 mL per hour to help maintain gut integrity. To improve personal comfort, the nasogastric tube was removed several days later. It was discussed he could consume 300 mL of an electrolyte drink daily with a cup of tea subsequently added, increasing total oral intake to 500 mL of fluid. Unfortunately, introduction of oral intake increased fistula output from 200 to 800-1000 ml per day.
He was initially provided energy at 100-125 kJ per kilogram (kg) of body weight (7900-9800 kJ). A weight loss of approximately 17 kg from his first surgery between February and April 2015 had been observed. In response to weight changes and taking into consideration his subsequent surgical complications, his estimated energy requirements were increased to 125-145 kJ per kg of body weight (10000-11600 kJ) to accommodate higher levels for repletion. Consequently, parenteral nutrition volume was increased to provide a total of 11400 kJ and 105 g protein (attaining estimated protein requirements of 95-120 g daily, 1.2-1.5 g protein/kg). Parenteral Nutrition was infused overnight allowing mobilization free from attachments during the day.
Given the high output losses and inability for enteral feeding, fistuloclysis was initiated. Fistuloclysis needed to consider the additional complexity of multiple small bowel fistulae with multiple afferent and efferent loops. Six separate enterocutaneous fistulae involving small bowel, presumed jejunal and proximal based on available operative records. Please refer to figure one to see a pictorial representation of the individual's abdomen. To minimize loss of enteral feed and maximize nutrient provision and absorption, the most distal fistula needed to be identified, allowing access and utilization of the residual small bowel between the distal fistula and the end ileostomy. This offered a mechanism to allow maximum absorption of nutrients and utilization of functioning small intestine.
Achieving fistuloclysis with multiple fistulas was a challenging undertaking. The anatomy of the fistulas had to be defined, particularly the distal most fistula opening. The proximal most fistula opening was evident by the highest output. The distal most fistula was identified via cannulation of each opening with a Foley catheter, and confirmation of intestinal passage of contrast into the ileostomy appliance. Feeding could then be planned to use the proximal afferent fistula, and the distal efferent fistula to the intact distal small bowel.
Obtaining effective enteral delivery was equally challenging. One option involved insertion of a Foley's catheter into the fistula limb and anchoring into the desired position by inflating the catheter's balloon, with a modified exit via the wound management system. This was not pursued given concern regarding possible perforation of the bowel by an inflated balloon.
The decision was made instead to feed a smaller diameter tube into the fistula limb, wrapping a stitch around the tube and clamping it into place. This was largely effective but was dislodged at times. An indwelling catheter was selected with feeds commencing slowly at 20 mL per hour of a 1 kilocalorie (kcal) per mL fiber free feed over 24 hours, providing 2000 kJ and 20 g protein. A formal feeding tube was not used as it had caused significant discomfort when attempting to insert and progress through the fistula. During this time, parenteral nutrition regimen remained unchanged. The next day output was evident in the stoma bag, indicating feed progression through the remnant bowel and exiting via the ileostomy, effectively reducing the total losses via reintroduction of chyme.
With stable insertion of a new feeding tube, the individual was able to tolerate fistuloclysis whereupon feeds were increased to 40 mL per hour of the 1 kcal per mL feed (provided 4000 kJ and 40 g protein) with a reduction in his parenteral nutrition a few days later to 125 mL per hour over 16 hours (provided 9500 kJ and 90 g protein). Total nutrition (enteral plus parenteral nutrition) was 13500 kJ and 130 g protein. Tube dislodgement continued to be an issue while the individual remained on fistuloclysis resulting in intermittent feeding only. These interruptions meant no enteral feeds were received, often over several days with only insignificant nutrition from 500 mL of allowed oral intake before a tube could be reinserted and feeding could be recommenced.
To provide the same amount of nutrition in a decreased volume, the enteral feed was changed to a 2 kcal/mL feed at 20 mL per hour which was subsequently increased to 40 mL per hour and was tolerated well by the individual. At this rate, fistuloclysis provided 8000 kJ and 70 g protein. To optimize the utilization of endogenous digestive enzymes the multidisciplinary team planned to commence chyme reinfusion and considered two different approaches evident in the literature presented by Parrish and Quatrara (2010) [6]. After careful consideration, dietetics recommended straining the wound manager output, drawing up and pushing through 120 mL of chyme every four hours whilst the feed was running. Once the individual was tolerating fistuloclysis at goal rate (50 mL per hour over 24 hours, providing 10000 kJ and 90 g protein), parenteral nutrition was ceased.
After many months of no oral intake, the individual was very pleased to commence a high protein high energy diet (for quality of life). Minor nausea and vomiting were experienced after over-eating his first meal. This oral intake affected the consistency of the wound manager output thickening the chyme and making it difficult to drain from the wound manager and strain before chyme reinfusion. Ultimately a system was arranged utilizing a unidirectional bladder irrigation pump and tubing connected to the feeding catheter, removing the manual step of straining the contents thus making it easier to perform chyme infusion during fistuloclysis. Dietetics discussed trialing a liquid diet to improve the consistency of the output however the individual declined due to the reduced palatability and the marked reduction in variety of the liquid diet.
A complex fistula repair was performed in September 2015 (approximately six months after admission) with repair and closure of all fistulas and the ileostomy. After a period of post-surgical recovery and approximately two weeks on parenteral nutrition, the individual transitioned to normal oral diet, and was then transferred in the beginning of November 2015 to his local hospital where he was discharged home two days later. There were no further intestinal or wound related complications.
Despite active nutrition support, weight declined during the clinical journey. The weight decline is even more significant when weight prior to admission for the first surgery is considered. During the period of parenteral nutrition administration, theoretical nutritional requirements were being achieved. Please refer to figure two which outlines weight and energy intake over the individual's clinical journey (Figure 2).
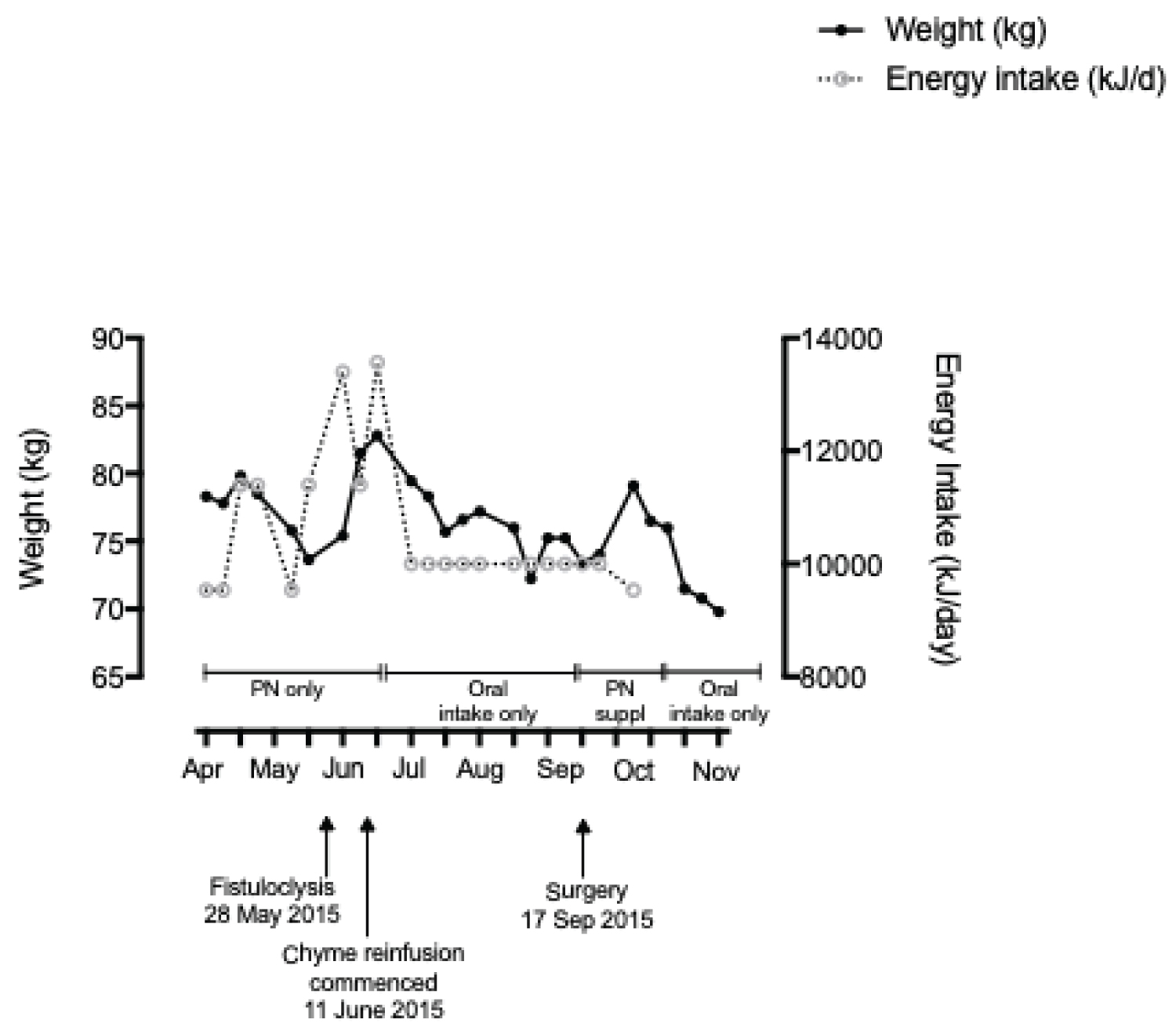 Figure 2: Weight and energy intake over time.
View Figure 2
Figure 2: Weight and energy intake over time.
View Figure 2
During fistuloclysis, confirmation of the volume absorbed compared with the volume delivered is more difficult to ascertain. The volume absorbed does not necessarily equate to the volume delivered as one would expect a degree of malabsorption due to the entire gastrointestinal tract not being utilized.
There are several reasons for the observed weight loss. Measured weight may have been compounded by fluid status. The individual may have also been in an increased catabolic state due to multiple wounds and many surgeries thus increasing his nutritional requirements beyond those estimated. Chyme reinfusion delivered as a bolus does not physiologically replicate the natural excretion of enzymes in response to food ingestion potentially impacting nutrient absorption. Additionally, it cannot be certain that the same scales were utilized at each weight measurement and there will always be fluctuations between scales. However, fluctuations do not explain the general downward trend observed.
Other reasons for weight loss and less than ideal energy intake was reduced hours of enteral feed delivery occurred when the feeding tube in the distal fistula would dislodge. Initially, there were extended periods of time before the feeding tube could be re-inserted and therefore the accumulated time lost in feeding could be an important contributor to the individuals weight loss particularly after parenteral nutrition was ceased.
When displaying the weight graphically, the enormity of his weight loss was much more apparent. During admission, the hospital transitioned from paper to electronic medical records. The current functionality of electronic medical records includes the ability to graph tabulated values such as weight, allowing clinicians to see trends more easily over time, bringing great value to patient's treatment and care.
There were some challenges relating to implementing chyme reinfusion in tandem with fistuloclysis. One was the time required to collect the output from the wound manager, strain the contents and administer for chyme infusion by nursing staff already within busy workloads. To simplify administration, a device named Enteromate 2 had been developed and patented, specifically for the purpose of chyme reinfusion, however this device is currently available only within France [3]. An improvised version utilizing a bladder irrigation pump was constructed. In addition to the problems with the feeding tubes, significant pain was reported around his wound management system as it was constantly leaking further irritating already excoriated skin, a recognized wound management issue of enterocutanous fistulas. Whilst fistuloclysis and chyme reinfusion achieved a positive clinical outcome, this method likely impacted negatively on the patient's quality of life.
Keeping a person-centered approach to care for individuals who are experiencing long admissions secondary to complex clinical conditions is of paramount importance. The extended admission period away from home and family as well as the pain he experienced from an open abdomen and skin excoriation for an extended period is challenging and it was observed that the individual exhibited a low mood at times. Some negative staff attitudes towards chyme reinfusion were observed where it was referred to as "fecal feeding" during discussions with other members of the team and around the individual. Different terminology may be more appropriate in the ward setting, and strictly speaking, the intestinal contents are chyme, and not fecal. Consideration of the most appropriate and technical term of chyme reinfusion should be paramount when speaking with the individual and other professionals and ensuring the individual has appropriate education and understanding particularly for complex and rare methods of feeding such as fistuloclysis and chyme reinfusion. Given many stressors, a person-centered approach is additionally important including the terminology employed during care provision and being sensitive with the language used while discussing these complex cases with all health care professionals.
While there was a period of approximately one month to determine the logistics of this complex feeding method, fistuloclysis with chyme reinfusion was an effective method of feeding with a successful transition from parenteral nutrition to total enteral nutrition. Our experience suggests that this method could be utilized in regional hospitals thus allowing patients to be nearer to their friends and family. There is also the potential for patients to be discharged home while awaiting corrective surgery although this was not explored in this case.
This individual experienced weight loss despite being provided the higher end of his estimated requirements (both with parenteral and enteral nutrition) indicating that he needed to be provided over and above his theoretical requirements to achieve weight maintenance. This is even more significant when the individual was receiving fistuloclysis and chyme reinfusion as there would be an aspect of malabsorption. More visual representations of weight change assists with more timely identification of weight loss trends during admission and trigger an increase in the level of nutrition support provided. Actively adopting person centered care will ensure the language used when describing chyme infusion will not have the potential to negatively influence opinions of a legitimate feeding method.
We thank the individual himself for his willingness to share his story and to the nursing staff, stomal therapy staff, allied health staff and other medical officers including Dr Helen Benham for their care of the individual.
The authors have no conflicts of interest to declare.
The authors have no funding sources to declare.
All authors contributed to the conception and design of the manuscript, the acquisition, analysis and interpretation of the data for the manuscript, drafted the manuscript, critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript as outlined in the ICMJE Criteria for Authorship.