Mucormycosis is an opportunistic mycosis common in poorly-controlled insulin dependent diabetic patients particularly with ketoacidosis. It often begins as a nasal and paranasal sinus infection with inhalation of spores into the paranasal sinuses of a susceptible host. In this case report we review the clinical symptoms, relevant imaging, and management of a diabetic patient with mucormycosis threatening his better seeing eye. This case report offers the clinician a review of important clinical and diagnostic findings that can help direct the need for orbital exenteration. In addition, various reconstruction options are considered to help guide informed management.
Mucormycosisis an opportunistic mycosis caused ubiquitous heat-tolerant saprobes [1]. Mucormycosis is common in poorly controlled insulin dependent diabetic patients particularlyin the setting of ketoacidosis. Ketoacidosis inhibits the binding of iron to transferrin allowing serum iron levels to rise which is favorable for fungal proliferation [2]. Other conditions that increase the risk of mucormycosis include any immunosuppression including advanced HIVinfection, leukemias, lymphoma, systemic treatment with corticosteroids, and severe burns [3]. The most common mode of spread of these fungi is through inhalation of airborne fungal spores [3]. The most common site for infection is the nose and paranasal sinuses. Upon entering the tissues, spores infiltrate blood vessels, inducing an inflammatory reaction, which leads to thromboses and ischemic necrosis. Direct culture of nasal discharge, tissue, or sputum will reveal broad hyphae with uneven thickness, irregular branching and septations [4].
The most advanced and life-threatening clinical form of opportunistic mycosis in patients with Diabetes Mellitus (DM) is rhinocerebral mucormycosis, which involves rapid progression with invasion of the sinuses, eyes, cranial bones, and brain [5]. Patients initially develop edema of the involved facial region, a bloody nasal exudates and orbital cellulitis [6]. Cerebral and neural invasion often begins in the nasal turbinates and paranasal sinuses and spreads along infected vessels to the retroorbital tissues and cavernous sinus. Clinical findings may include proptosis, ophthalmoplegia, and edema of the lids and retina and then hemorrhagic infarction when spread to brain parenchyma [7].
All procedures described in this manuscript were carried out in accordance with national law and the Helsinki Declaration from 1964 (in its present revised form). This case report was approved by the local ethics committee and the patient consented to the use of their medical data and identifiable photographs to be published as part of this report. This case report was completed in full compliance with the Health insurance Portability and Accountability Act of 1996 (HIPAA).
A 63-year-old gentleman presented to the Emergency Department in ketoacidosis with an ulcerating, necrotic defect of the left hard palate with left facial ecchymosis and left scleral chemosis with extraocular muscles and vision intact (Figure 1A and Figure 2A). Computed Tomography (CT) imaging of the maxillofacial bones revealed extensive paranasal sinus mucosal thickenings most advanced in the distribution of the left anterior osteomeatal unit, erosion of left maxillary sinus wall and air-fluid level in the left sphenoid and frontal sinus. Axial images of the maxillofacial bones and coronal reconstructions demonstrate attenuation of the deep soft tissues of the left face with osteolysis involving portions of the maxilla near the midline and towards the left, medial, inferior and lateral bony walls of the left orbit, the left maxillary sinus, portions of the left ethmoid and left sphenoid sinuses, and the left greater and lesser sphenoid wings. Prominence of the left inferior rectus muscle secondary to inflammation with mild left proptosis was evident.
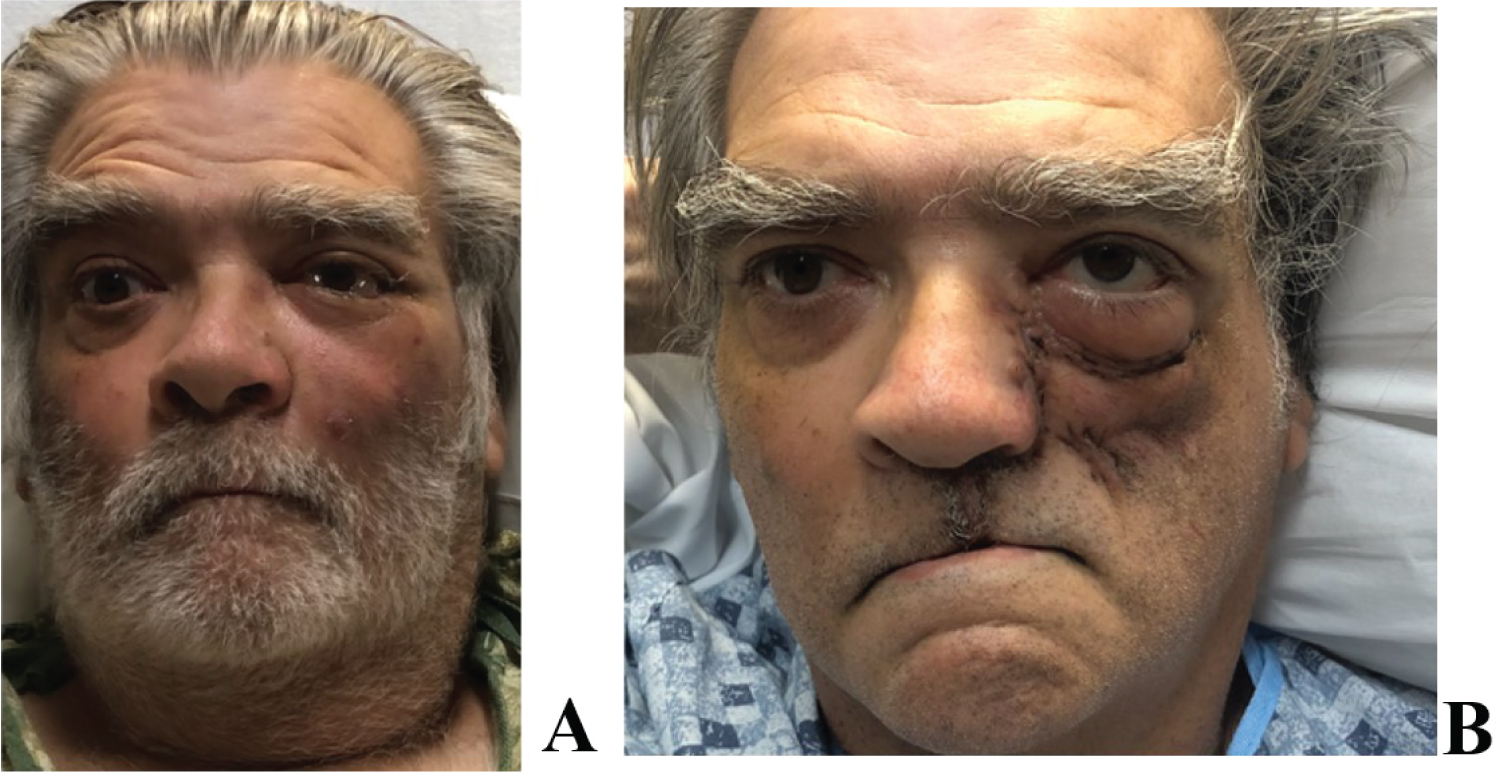 Figure 1: A) Preoperative picture of patient with left cheek skin ecchymosis and left scleral chemosis; B) Postoperative week 2.
View Figure 1
Figure 1: A) Preoperative picture of patient with left cheek skin ecchymosis and left scleral chemosis; B) Postoperative week 2.
View Figure 1
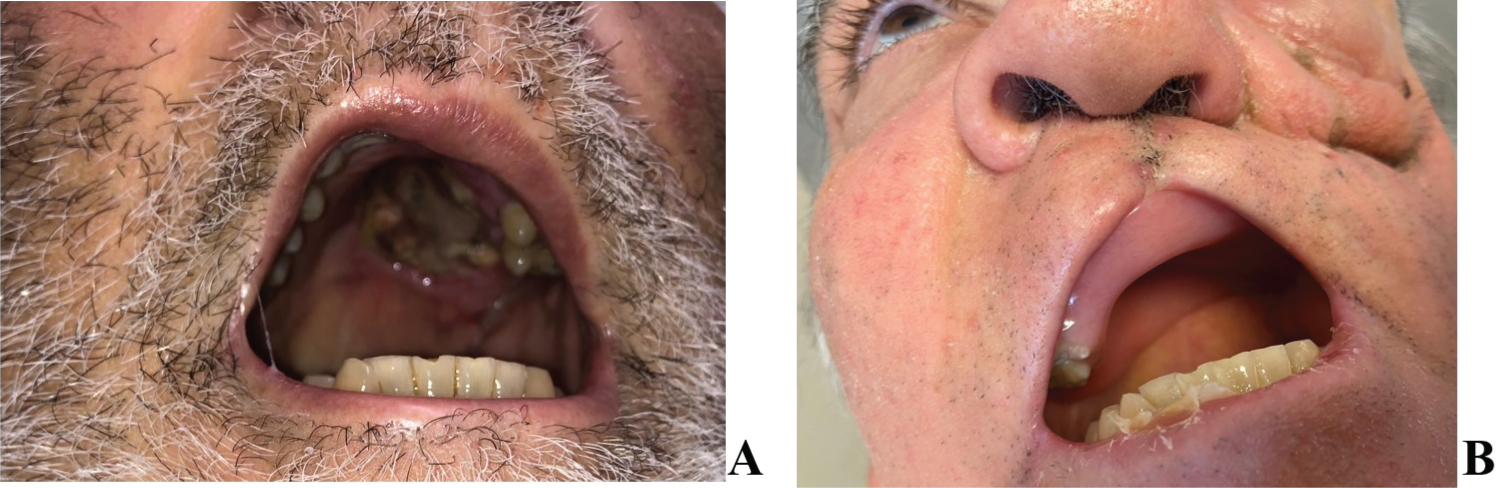 Figure 2: A) Preoperative palatal necrotic lesions; B) Postoperative palatal defect.
View Figure 2
Figure 2: A) Preoperative palatal necrotic lesions; B) Postoperative palatal defect.
View Figure 2
After biopsy confirmed mucormycosis, the patient underwent a left infrastructure maxillectomy with orbital sparing and two subsequent debridements. Extensive resection of the left zygomatic arch, left hard and soft palate and the surrounding soft tissues was performed (Figure 2). During the procedure, severe involvement and necrosis of the left side lateral nasal wall, floor, and septum were visualized. Surgical resection resulted in a large hard palate defect with an oral-nasal-maxillary continuity. Postoperative CT imaging demonstrates opacification of the left ethmoid air cells, left sphenoid and left frontal sinuses indicative of residual paranasal sinus focal infection. Post-operative MRI revealed residual enhancement of the inferior rectus muscle as well as subtle asymmetric enhancement along the optic nerve sheath on the left compared to the right, but no definite evidence of dural enhancement to suggest intracranial extension. CT maxillofacial revealed some concern for inferior orbital wall thinning without orbital fat herniation.
The patient had a gastrostomy tube placed and was discharged with subsequent outpatient treatment including three months of Amphotericin Band improved diabetic management. Post-surgically hewas fitted with a palate obturator which was well tolerated and allowed normal speech and nutrition by mouth.
The most common clinical presentation of mucormycosis is nasal and paranasal sinus infection which begins with inhalation of spores into the paranasal sinuses of a susceptible host [8]. An early biopsy is paramount as nasal and paranasal symptoms can be confused with bacterial rhinosinusitis before progressing to blood-stained rhinorrhea, intranasal mucosal necrosis and hard palate necrosis [4].
The spread of the fungus via the nose into the orbit can occur via the ethmoid air cells and adjacent lamina papyracea or through defects in the orbital floor which allows access to the inferior orbital fissure, superior orbital fissure, and the orbital apex [4].
Symptoms vary and can include worsening vision and diplopia. More advanced features include chemosis, proptosis, ophthalmoplegia, optic atrophy, and blindness. Further intracranial spread can begin with a headache before advancing to altered mental status and death [9]. Once the disease reaches the brain or cavernous sinus the prognosis is almost invariably fatal.
The anatomical extent of (rhino-oculo-cerebral mucormycosis) ROCM has been classified into stage I (limited to the nasal mucosa), stage II (paranasal sinus extension), stage III (orbital extension), and stage IV (extension to central nervous system) [10]. Contrast enhanced MRI is the preferred imaging to help guide debridement of devascularized tissue. Patients with Stage III disease may undergo orbital exenteration to avert cerebral extension [11]. Globe involvement is suggested by thickening and enhancement of any of the three coating layers of the globe on MRI [9]. Optic nerve involvement can be secondary to arterial or venous occlusion, nerve infarction, or direct infiltration of the optic nerve. Infarction presents on MRI as high-signal intensity of the nerve on diffusion-weighted imaging [12]. Direct invasion of the optic nerve can be visualized as increased caliber with signal intensity changes within the nerve. Isolated involvement of the optic nerve suggests spread of infection through branches of the ophthalmic artery [12].
Hanover, et al. recommend in their management algorithm of ROCM, orbital exenteration is recommended for ROCM patients with severe vision changes including ophthalmoplegia and/or blindness [10]. However, even if the orbit can be spared, patients with advanced disease are often still left with a major mid-face deformity following maxillectomy which impairs normal speech and swallow [13]. Reconstructive options include prosthetic obturators, regional flaps, and free tissue transfer. A prosthetic palatal obturator is a simple and inexpensive post-surgical restorative option. The advantages of the palatal obturator include: Acceptable cosmesis, cost-efficiency, relative ease of manufacture, facilitation of disease surveillance, deferred surgery in elderly patients or those with poor health. The disadvantages include: Prosthesis shift and need for iterative adjustments, need for daily cleaning, less mastication resistance relative to osseo-integrated implants as may be possible with a free flap [14]. Free tissue transfer may be a better option when available particularly in very extensive defects when sufficient bony landmarks are not present to anchor a prosthesis.
Free flaps can resolve many of the issues associated with prosthetic obturators including nasal leakage, need for removal and cleaning, and iterative prosthetic refinement. The options for microvascular bone free tissue transfer include scapula and fibula [15]. Autologous tissue transfers help restore and support facial contours while also allowing for near normal restoration of speech and swallow. The disadvantages of free tissue transfer include need for prolonged surgery and highly technical surgical expertise [14].
In this case the patient had sufficient remnant native dentition and access to a low-cost prosthetic expert that was available to fit the patient for an obturator. The patient was able to regain normal speech and swallow with the obturator. There was, however, some visible facial deformity from the loss of volume which was not restored.
Treatment involving a combination of surgical debridement and antifungal therapy should be undertaken as soon as the diagnosis of rhino-orbital-cerebral mucormycosis is suspected to limit progression of disease and resulting facial disfiguration as well as possible death. Sparing the orbit particularly in the case of disease near the better or only-seeing eye is critical to post-recovery quality of life. In the setting of mild to moderate orbital findings, sparing the orbit should be heavily favored and an understanding of acceptable clinical and MRI findings should be well understood by the surgeon and clinical team caring for the patient. In this case report we have highlighted these important MRI findings in the case of a patient that had mild to moderate orbital findings but ultimately was successfully treated without compromising his vision. Moreover, a review of reconstructive options is helpful both to counsel the patient and to maintain a broad outlook to offer the patient a tailored surgical and medical management scheme.
None.
J. Adams and A. Bender-Heine declare that they have no competing interests.