Granular cell tumors (GCTs), also known as Abrikossoff tumors, are rare skin and soft tissue tumors originating from the Schwann cells of peripheral nerves [1]. While predominantly seen on the head and neck, they can occur anywhere on the body and arise on the vulva in 5-16% of cases [2]. GCTs have also been described in the ovaries, uterus, cervix, vagina, episiotomy scar, and pubis [3,4]. GCTs can present at any age and sex but they are most prevalent in females ages 30-50 [2]. The tumors are typically solitary, subcutaneous, painless, and mobile. The overlying skin may be normal or may be ulcerated, thickened, or hyperpigmented, increasing concern for malignancy [5,6]. GCTs present as multifocal in 25% of cases [7]. GCTs are difficult to distinguish clinically from other similar masses and require histology and immunohistochemistry to confirm [5].
Here, we discuss a classic case of a vulvar GCT in a 51-year-old female. In this case report, we describe the clinical presentation of rare vulvar GCTs, highlight the pathology findings, review treatment, and discuss possible risk factors for predisposition and progression to malignancy.
A 51-year-old G3P2012 female presented to the gynecology clinic after referral from her primary care provider for a lump in the vulva. The lump was first noticed two years prior, at which time it was pea-sized per patient report and has slowly grown since. The patient reports the lump is not painful or pruritic but has become more bothersome due to its periclitoral location. There was no drainage, bleeding, ulceration, or inflammation at the site of the lump and no changes in vaginal bleeding or discharge. Past medical history is remarkable for psoriatic arthritis, for which she takes methotrexate and etanercept, as well as hypercholesterolemia, polycystic ovarian syndrome, rosacea, and a recently removed axillary follicular cyst. Additionally, she has a 7.5 pack-year smoking history and quit 20 years prior to presentation.
On exam, a 2 cm firm, smooth, nodular, mobile skin lesion was palpated in the clitoral hood. The mass was not affixed to the clitoris, did not drain, and was not indurated. Physical exam was otherwise unremarkable with no palpable lymph nodes. The initial differential diagnosis included an epidermoid cyst or lipoma. There were no concerning findings suggesting malignancy. The patient was counseled on her options for surgical excision versus observation and chose the former.
Surgical excision was uncomplicated. The mass grossly appeared to be an epidermoid cyst. The specimen was sent to pathology for further evaluation, at which time morphological features and immunohistochemistry profile were compatible with granular cell tumor (Figure 1). The tumor was not transected but had less than 0.1 cm margins in several areas. Cells had increased mitotic activity of 1/10 HPF with an atypical form. Focal cytologic atypia with vesicular nuclei and prominent nucleoli were also present. No necrosis was seen. The cytoplasmic granular material was diastase-resistant with PAS-D staining (Figure 1C and Figure 1D). Immunohistochemistry stained positive for S-100 (Figure 1E and Figure 1F), Sox-10, Inhibin, Calretinin, and CD56 and negative for Keratin AE1-AE3, HMB45, Mart-1, Desmin, Myogenin, and Myoglobin. CD68 was faintly positive with a very light pink stain (Figure 1G and Figure 1H). This antibody profile is similar to that of Schwann cells.
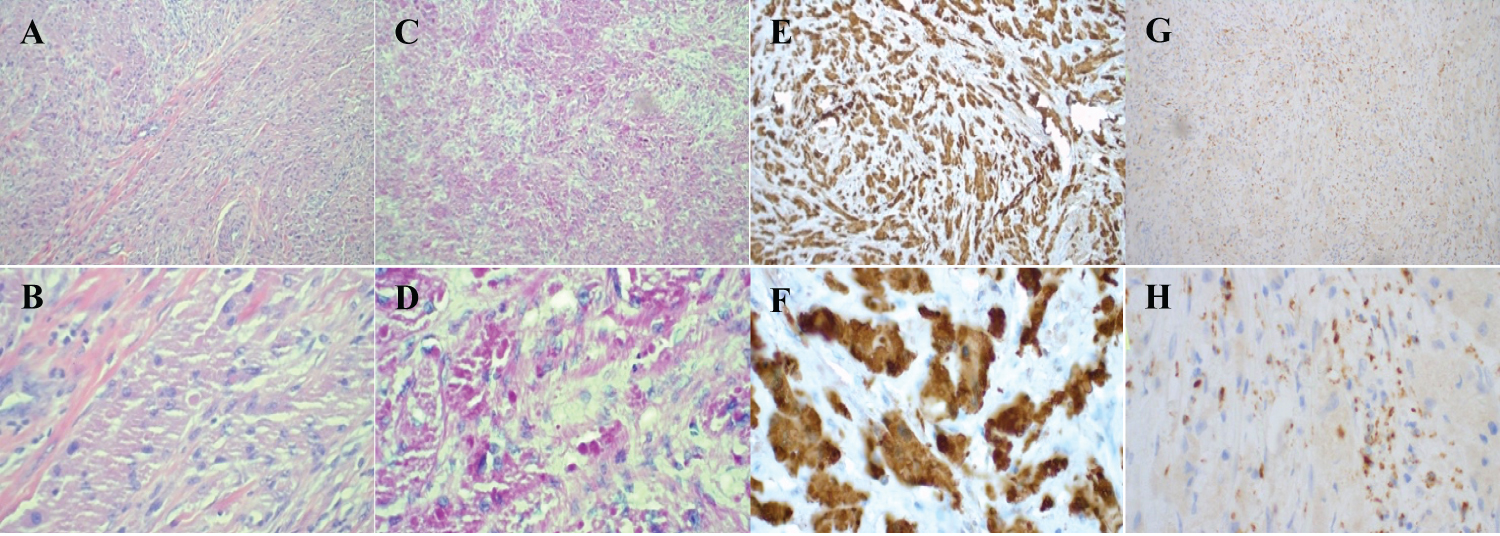 Figure 1: A) H&E stain of vulvar tissue at 10x; B) 50x magnification; C) PAS stain of vulvar tissue at 10x; D) 50x; E) Immunohistochemistry S-100 antibody stain of vulvar tissue at 10x; F) 50x; G) Immunohistochemistry CD68 antibody stain of vulvar tissue at 10x; H) and 50x.
View Figure 1
Figure 1: A) H&E stain of vulvar tissue at 10x; B) 50x magnification; C) PAS stain of vulvar tissue at 10x; D) 50x; E) Immunohistochemistry S-100 antibody stain of vulvar tissue at 10x; F) 50x; G) Immunohistochemistry CD68 antibody stain of vulvar tissue at 10x; H) and 50x.
View Figure 1
This case was presented at tumor board given its rarity, the lack of standardized follow-up, and narrow margins. The slow growth, size < 4 cm, lack of ulceration, necrosis, or vascular invasion, and the patient's younger age supported a benign tumor. However, the increased mitotic activity and vesicular nuclei classified this tumor as atypical, falling short of malignant. Given the atypical classification, it was agreed that the patient should be seen every six months by gynecologic oncology for a minimum of five years. The patient was counseled on the pathology results and agreed with the plan for surveillance given the potential risk of recurrence.
Granular cell tumors (GCTs) are uncommon, Schwann-cell derived tumors typically found in the head and neck, most commonly on the tongue, but have been reported nearly everywhere in the body. Our case is a classic presentation of a very rare vulvar mass, givenour patient's age, gender, and history of a slowly growing painless mobile mass. However, this presentation is also classic for other, more common vulvar masses, such as epidermoid cysts, the working diagnosis pre-excision in our case, dermatofibromas, Bartholin's duct cysts, and lipomas. Due to their rarity and indistinct character, GCTs are frequently left off the differential.
Diagnosis is made via pathologic assessment. Histopathology shows sheets or ribbons of polygonal cells with eosinophilic cytoplasm abundant in lysosomes and PAS-positive diastase-resistant granules known as pustulo-ovoid bodies of Milan [8]. Nuclei are uniform, small, and centrally located [5]. This histology is not specific to GCTs; therefore, immunohistochemistry is required to make the diagnosis. Given the neuronal origin, GCTs stain positive for S-100 protein, neuron-specific enolase, laminin, vimentin, and CD-68 [5,9]. The GCT in our case stained positive for S-100, Sox-10, Inhibin, Calretinin, and CD56 and very lightly for CD68.
Treatment of choice is wide local excision. Margins in GCTs are often poorly demarcated, making excisions with clear margins difficult. There is debate over re-excision in benign cases for positive or slim margins. Recurrence rates are higher in benign cases with positive margins at 20% compared to 2-8% for benign cases with negative margins, however, there have not been any randomized controlled trials comparing re-excision to observation [10]. A case series of 17 cases in 13 patients found that 8/17 excisions had positive margins and 2/8 required re-excision for recurrences at 8 and 15-years post-excision, while none of the 9/17 with negative margins had recurrence [11]. Given the risks of surgery and long intervals between recurrence, it is acceptable to forego re-excision in benign cases regardless of margins and follow the patient closely for recurrence.
While only malignant in 2% of cases, malignant GCTs have a poor prognosis [12]. The tumors are very aggressive with rapid regional and metastatic spread. Malignant GCTs respond poorly to radiation and chemotherapy, so once a GCT has spread to the point that complete excision is not possible there are no effective treatment modalities [13]. The five-year survival rate for malignant GCTs is 60% with a ten-year survival rate of 30%. Recurrence is high at 50% within five years [14]. Characteristics suggesting malignancy include rapid growth, size greater than 4 cm, necrosis, vascular invasion, local recurrence, advanced age, and overlying skin changes such as ulceration, thickening, or hyperpigmentation [10]. A set of six histologic criteria for GCTs were developed by Fanburg-Smith including: necrosis, spindling, vesicular nuclei with large nucleoli, increased mitotic activity, high nuclear-to-cytoplasm ratio, and pleomorphisms [14]. Benign GCTs meet none of these criteria, atypical GCTs meet one-to-two, and malignant GCTs meet three or more. Our case was classified as atypical, falling short of malignant, with only two criteria met: increased mitotic rate and vesicular nuclei with prominent nucleoli.
There is a paucity of research on any possible risk factors for predisposition to GCTs, vulvar or otherwise. Our patient has a history of psoriatic arthritis and takes methotrexate and etanercept, two immunomodulating medications. To date, there has been no described association between GCT and psoriatic arthritis or any other autoimmune diseases. While rare, low-dose methotrexate is associated with increased rates of malignancy, mainly lymphomas and skin cancers [15,16]. Etanercept is associated with increased rates of squamous cell cancer, although there is debate about the causal relationship due to the frequent multi-modal treatment of psoriatic arthritis making controlling for confounding factors difficult [17]. No relationship has been described between either methotrexate or etanercept and GCTs, but given the possible relationships with other tumors and cancers it is worth exploring further. Additionally, our patient had a history of tobacco use, albeit relatively modest and remote. Smoking has not been studied as a risk factor for vulvar GCTs. Given the association between smoking and vulvar squamous cell carcinoma as well as other gynecological cancers, further research should explore any possible association between smoking and vulvar GCTs.
Granular cell tumor of the vulva is a rare diagnosis for a vulvar mass. Diagnosis is made by histopathology showing polygonal cells with PAS-positive granules and immunohistochemistry positive for S-100. While typically benign, GCTs frequently recur, and malignant GCTs have a poor prognosis once metastasized. Treatment of choice is excision with close follow-up due to a high recurrence rate. GCTs can be multifocal and arise in many locations in the body, requiring surveillance to extend beyond the vulva. Further research on risk factors for predisposition to GCTs would be helpful in identifying at-risk patients and counseling on treatment options for unidentified vulvar masses.
There are no grants, funds, or other financial or material support for this research. There are no conflicts of interest to disclose for any of the authors. No assistance from medical writing experts was received for this manuscript.