Background: Growing tumor syndrome (GTS) occurs rarely in women with immature ovarian teratoma. Unlike many ovarian malignancies, it can be successfully treated with chemotherapy and fertility-sparing surgery. Early diagnosis is crucial to avoid unnecessarily aggressive treatment.
Case: 31-year-old nulliparous female with grade 2 immature teratoma presented following presumed treatment failure. Tumor markers remained consistently normal. Biopsies demonstrated gliomatosis peritonei (GP). Despite chemotherapy, tumor growth persisted. She then underwent fertility-sparing cytoreductive surgery. Final pathology showed no evidence of malignancy and presence of only mature teratoma cells.
Conclusion: Early recognition of GTS in young women is crucial to avoid excessive use of chemotherapy, radiation, and nonfertility-sparing surgery. Oncologists should consider GTS in patients with a diagnosis of immature teratoma, histologic findings of GP, and normal tumor markers.
- In growing tumor syndrome (GTS), ovarian malignancies react uniquely in response to chemotherapy, rapidly increasing in benign tumor size with normal serum tumor markers [1].
- Gliomatosis peritonei (GP) is thought to be an indicator of development of GTS, with one study finding nearly 40% of patients with GP with development of GTS [2].
- The mainstay of treatment of GTS in the setting of immature teratoma is fertility-sparing surgical resection (if desired), and avoidance of prolonged chemotherapy and radiation.
In this report, we describe a rare case of a patient with ovarian immature teratoma who developed gliomatosis peritonei (GP) and growing tumor syndrome (GTS) and was successfully treated with adjuvant chemotherapy and fertility-sparing surgical resection. While this phenomenon is well known in men after testicular germ cell tumors, GTS is rare in women. Just over 100 cases of GTS have been described after an ovarian malignancy, with an even smaller subset after ovarian immature teratomas, though recent literature indicates that this syndrome may be more common than previously thought, present in up to 20% of reported cases at some centers [2,3]. While GTS in the setting of immature teratoma has been reported in women from ages 5-52, the mean age at diagnosis is 22 [4].
Unlike many ovarian malignancies, immature teratoma with growing tumor syndrome can be successfully treated with chemotherapy and fertility sparing surgery, as was the case for our patient. It is important that providers know how to recognize this pathology so that consideration can be given to a fertility sparing approach, and that increasing tumor size is not necessarily regarded as treatment failure.
The patient was a 30 year-old nulliparous female who In June of 2020presented to her local emergency department with abdominal bloating, pain, and an increasing abdominal girth. CT scan showed a large pelvic mass consistent with an adnexal mass. Her pre-operative tumor markers showed a slightly elevated Ca-125 at 50 U/mL with normal AFP and HCG. She subsequently underwent an exploratory laparotomy, via Pfannenstiel incision, right salpingo-oophorectomy and omental biopsy. A diaphragmatic nodule was noted at time of surgery but could not be accessed for biopsy given the Pfannenstiel incision. Final pathology showed a 20 cm, grade 2, immature teratoma. Her omental biopsy was positive formature teratoma, and pelvic washings were negative for malignancy. Further post-operative imaging of the chest, abdomen and pelvis revealed 2.7 cm metastatic lesions involving the pleural side of the right hemi-diaphragm with a mixed density lesion in the pelvis likely consistent with residual disease. She then underwent a PET-CT confirming pleural implants on the diaphragm, hepatic capsular and subdiaphragmatic disease and residual pelvic disease. She was hence diagnosed with Stage IV, grade 2, immature teratoma of the ovary. She underwent 3 cycles of BEP and was planned for a fourth cycle. The fourth cycle was held due to development of thrombocytopenia and anemia. Post-treatment imaging in January of 2021 did not show a significant response, and she was switched to systemic Taxotere and Carboplatin. She received two cycles but had some delay between cycles secondary to cytopenias. She then presented to our Gynecologic Oncology clinic for evaluation in March 2021.
Our work-up consisted of repeat tumor marker studies which showed normal HCG, AFP, CA-125, and HE-4 levels. Pathology from prior specimens was reviewed at Mayo Clinic and was consistent with outside findings, except for the omental biopsy. The omental biopsy was found to have peritoneal nodular gliomatosis but no evidence of teratoma. Findings were otherwise in agreement. Repeat CT imaging of the chest, abdomen and pelvis were compared to outside studies with the following results:
1. Moderate amount of loculated extraluminal markedly increased from 08/2020.
2. Cystic and solid lesions in bilateral adnexa than larger from 01/2021 and 08/2020.
3. Greater omental nodularity/metastases increased from 01/2021, new or markedly increased from 08/2020.
4. 4 Chest CT showing stable appearing necrotic lymphadenopathy right cardiac phrenic angle region.
Her case was discussed at multi-speciality tumor board and the decision was made to proceed with cytoreductive surgery of the chest, abdomen and pelvis. The patient was adamant that she wanted fertility preservation if at all possible. She hence underwent a robotic assisted mediastinal excision, thymectomy, and removal of all right pleural disease. This was followed the same day by exploratory laparotomy, omentectomy, completion right oophorectomy, appendectomy, partial recto-sigmoid resection with anastomosis with protective diverting ileostomy, and generalized tumor cytoreduction. Large tumor masses were encountered within the peritoneal cavity (Figure 1). Fortunately, the uterus and left adnexa were normal and were preserved (Figure 2). Final surgical pathology showed mature teratoma, extensive benign peritoneal nodular gliomatosis, and no evidence of malignancy. The patient’s post-operative recover included development of a pelvic abscess, managed with percutaneous IR guided drain placement and antibiotics. Two months following surgery she underwent additional ileostomy takedown.
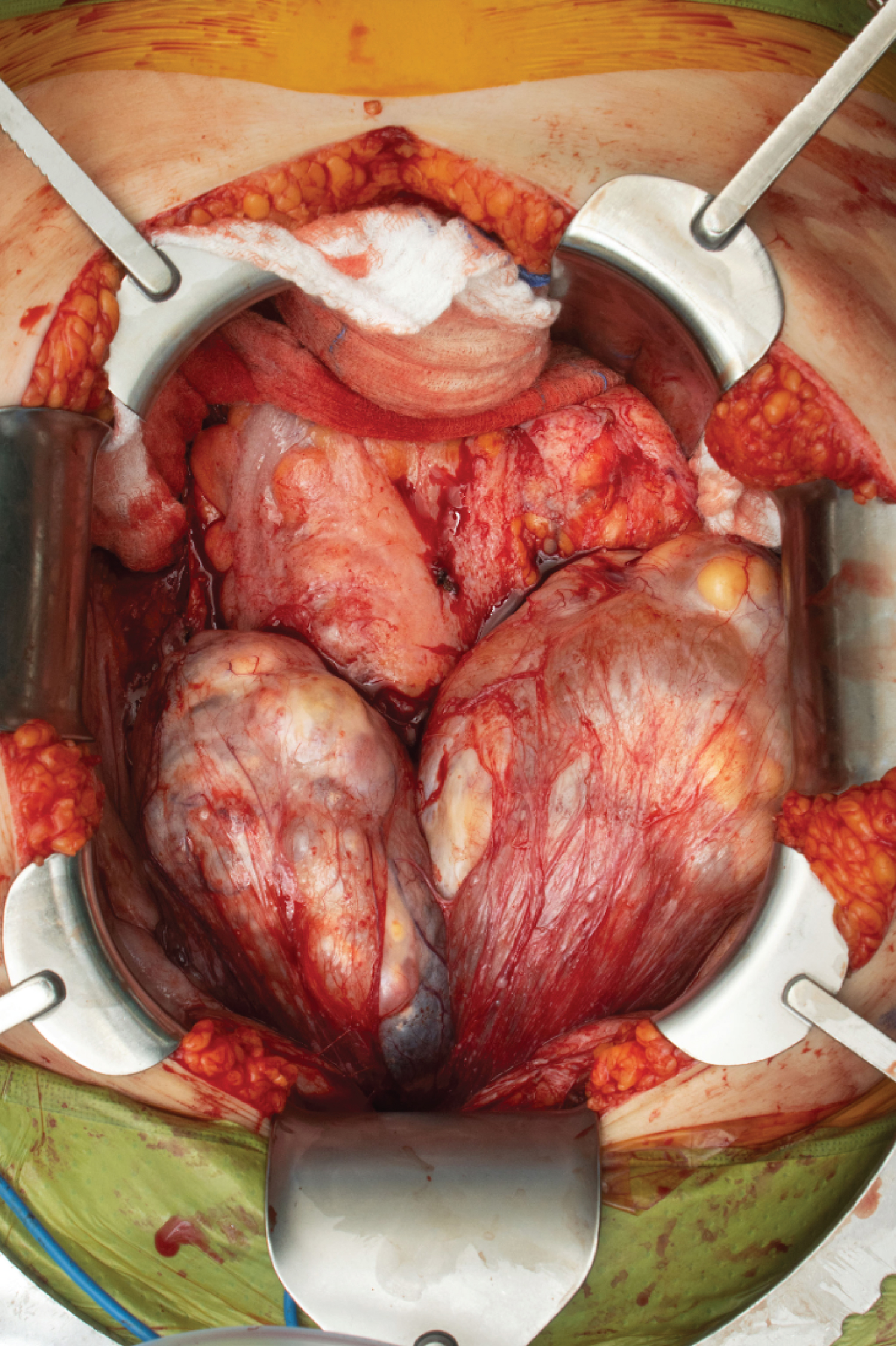 Figure 1: View of large intraperitoneal masses encountered in the peritoneal cavity.
View Figure 1
Figure 1: View of large intraperitoneal masses encountered in the peritoneal cavity.
View Figure 1
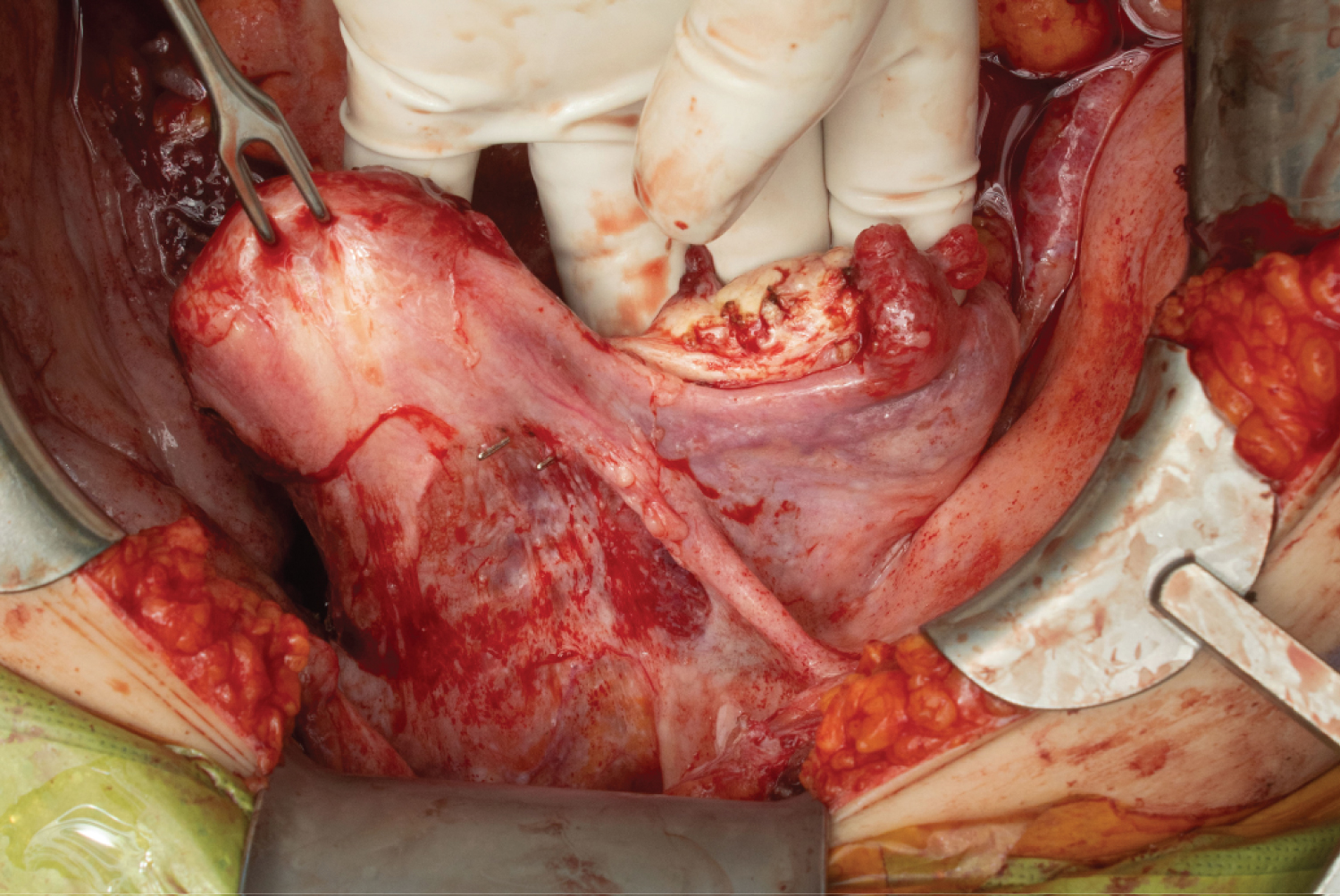 Figure 2: Uterus and left adnexa appeared grossly within normal.
View Figure 2
Figure 2: Uterus and left adnexa appeared grossly within normal.
View Figure 2
Immature teratomas of the ovary accounts for around 1% of ovarian tumors, affecting a wide range of women from children to adults [1]. While treatment varies between pediatric and adult patients, adult cases are typically managed with both chemotherapy and surgical resection [4]. A rare subset of patients treated for immature teratomas develop a "growing teratoma syndrome" (GTS), with a unique response to chemotherapy, resulting in rapidly increasing tumor size but with normalization of serum tumor marker levels [1]. GTS varies in presentation, with some cases occurring early in the course of chemotherapy and others presenting years later [4,5]. The pathogenesis of GTS is still poorly understood, but proposed mechanisms include: (1) Chemotherapy induces the malignant differentiation of immature teratoma into a mature component; (2) Chemotherapy destroys only the immature malignant cells and spares the mature benign teratomatous elements, allowing them to proliferate; or (3) A combination of these two explanations [6,7]. Some murine models additionally suggest that GTS may involve an inherent and spontaneous differentiation of malignant cells into benign tissues [8]. Gliomatosis peritonei is a unique phenomenon that can occur with any immature teratoma, but seems to be a significant indicator of subsequent development of GTS, with one study finding nearly 40% of patients with GP developing GTS [1]. It is defined histologically as the presence of mature glial tissue implants on the peritoneum. Interestingly, GP can arise in adjuvant chemotherapy-naïve patients, unlike GTS [9]. Its pathogenesis is poorly defined, with early theories suggesting it is a form of peritoneal spread, but more recent studies indicating that GP is genetically unrelated to the original tumor, making this theory improbable [9]. Despite its mature, benign nature, some studies have found GP to be associated with more frequent recurrence of immature teratoma, though there was no effect on overall survival [10].
Treatment for immature teratomas depends on the grade and stage at diagnosis [1]. Initial resection followed by adjuvant chemotherapy is used routinely in adults, but as noted in our patient, can be complicated by development of GP and GTS. Early recognition of GTS avoids unnecessary treatment, as GTS it is refractory to both chemotherapy and radiation therapy [11]. The mainstay of treatment for GTS is surgical resection, but if this is not an option, alternative medical management using alpha-interferon has shown promise in stabilization of growth in GTS [11]. In most cases, fertility-preserving surgery is an option, with several reported cases of pregnancy after treatment of GTS [12-14]. In GP, while biopsy of identifiable lesions is important to rule out misdiagnosis, complete resection with surgical management is rarely feasible with many small implants scattered throughout the peritoneum. Instead, GP tends to go into quiescence after resection of the primary tumor in most cases [9].
Rate of recurrence in GTS varies with grade and stage, but complete resection reduces the rate to 0-12.7% [4]. For early detection of initial GTS and recurrence, routine tumor markers and ultrasound examinations are highly recommended, with three-month follow ups during the first year, six-month intervals during the second year, and yearly surveillance through the fifth year following total resection. CT imaging is also recommended, twice in the first year and annually after that [4]. As previously stated, the presence of GP is associated with higher rates of recurrence of immature teratoma, but does not affect overall survival [10]. The prognosis of patients with completely resected GTS is generally excellent, with a five year survival rate of 89-90% [2]. Subsequent malignant transformation of GTS appears to be low at approximately 3% [4].
This case demonstrates a classic presentation of GTS in a young woman with normal tumor markers and diagnosis of immature teratoma. The resolution of malignant elements of her disease with progressive growth of mature teratoma cells following chemotherapy fits the proposed models of pathogenesis of GTS. Early recognition of this pathology avoided the need for unnecessarily aggressive chemotherapy and non-fertility sparing surgery. Gynecologic Oncologists must consider the diagnosis of GTS in patients with normal tumor markers, a diagnosis of immature teratoma, lack of response or tumor enlargement despite chemotherapy and the histologic finding of gliomatosis peritonei.