Background: Sierra Leone reported its first case of COVID-19 on March 30, 2020. This report describes the clinical characteristics and the epidemiological features of the first few cases and contacts of COVID-19 in Sierra Leone.
Methods: A prospective case-ascertained study was conducted to identify contacts of laboratory-confirmed COVID-19 cases. Demographic, socio-economic, and clinical presentations were collected. Specimen were collected on day 1, day 7, and days 14-21. Data was analyzed using R, Stata, and Microsoft Excel.
Results: The study included 160 cases and 1,003 contacts (56% males, 66% age 15-49 years). Of the total, 101/160 (63.1%) were primary cases, 59/160 (36.9%) secondary cases. Common symptoms reported included cough (39/160, 24.4%), headache (35/160, 21.9%), and fatigue (29/160, 18.1%). However, 55% (88/160) of the cases were asymptomatic at enrollment, and 83% (49/59) of the secondary cases were exposed to asymptomatic primary cases. The secondary infection rate was 5.8%. Almost 97% of the secondary cases tested positive on day 1and 3.4% on day 7. Pre-existing medical conditions among cases included diabetes and obesity.
Conclusions: We were able to identify clinical and epidemiological characteristics of 160 COVID-19 cases in the early phase of the pandemic in Sierra Leone. The findings of this study helped the Government of Sierra Leone develop and revise its policies and response strategies in order to reduce preventable COVID-19-related deaths. Early investigational protocols such as the first few cases study can be of great utility during outbreaks of relatively unknown diseases.
What is already known on this topic
A cluster of unusual pneumonia cases was reported in Wuhan, China, that was later identified to be caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2, or SARS-CoV-2. However, limited knowledge exists on the epidemiological and clinical characteristics of SARS-CoV-2 and the outbreak dynamics at the time Sierra Leone detected its first case.
What this study adds
This study helped to understand the key clinical characteristics of COVID-19 cases and epidemiological features of COVID-19 diseases in Sierra Leone.
Implications of the study in practice, or policy
The findings of this study provided evidence for targeted and specific prevention and control measures to contain the spread of the disease in Sierra Leone.
On December 31, 2019, a cluster of unusual pneumonia cases was reported in Wuhan, China, that was later identified to be caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2, or SARS-CoV-2 [1-3]. On March 11, 2020, WHO declared the disease caused by SARS-CoV-2, COVID-19, a worldwide pandemic [2,3].
Sierra Leone is a small country of seven million people in West Africa. During January-March 2020, Sierra Leone was at risk of imported COVID-19 cases due to its geographic proximity to Guinea and Liberia, which, at the time, had already recorded 111 and 10COVID-19 cases, respectively [4,5]. The country reported its first case of COVID-19 on March 30, 2020, from a traveler from France [6]. However, with the porous nature of the land borders in Sierra Leone and ongoing outbreaks in neighboring countries, there is a high likelihood of unidentified cases in Sierra Leone before this time, especially since subsequent cases did not have an epidemiological link with the index case [7].
There was limited knowledge on the epidemiological, clinical, and virological characteristics of SARS-CoV-2 and the outbreak dynamics at the time Sierra Leone detected its first case. The First Few X (FFX) cases and contact investigation study for COVID-19 is based on one of the early investigation protocols under WHO’s Unity Studies [8]. We conducted the FFX study in Sierra Leone to understand the key clinical characteristics of COVID-19 in the country, including its clinical severity and proportion of symptomatic cases, and to understand the key epidemiological features of COVID-19 disease transmission, including exposures, secondary attack rate, and case fatality rate to provide targeted and specific prevention and control measures to contain the spread of the disease. A similar study conducted in Nigeria reported higher case fatality rate [9].
Sierra Leone is comprised of 16 districts, of which, Western Area Rural (WAR) and Western Area Urban (WAU), including the capital of Freetown, are the most populous. The study was conducted in these districts due to the population density, early epicenter location, and availability of logistics as we anticipated enrolling several cases in a short time [10]. This case-ascertained study was. A prospective case-ascertained study conducted from June 26 through September 30, 2020 [11], adapted from the WHO FFX study [12], was employed for this investigation.
Laboratory confirmed SARS-CoV-2 positive cases detected through polymerase chain reaction (PCR) testing were identified from the Ministry of Health and Sanitation electronic disease surveillance system. We used the daily COVID-19 results line list shared by the laboratory pillar to enroll those who gave consent to participate in the study. The contacts of cases were identified through case interviews using the investigation definition of a contact during enrollment into the study. A total of637 individuals, majority of them contacts and comprising of non-consenting or critically ill persons who could not be interviewed and had no relatives willing to be interviewed were excluded from the study, and were replaced with subsequent cases and contacts reported [13]. We enrolled a total of 1,163 participants in the study.
A suspected case was defined as, any person presenting with acute respiratory illness (whether mild or severe) who in the 14 days prior to the onset of symptoms had contact with a confirmed or probable case of COVID-19 or history of travel or residence in a place of community transmission, or a patient with severe acute respiratory illness requiring hospitalization and with no other etiology that explains the clinical presentation. A probable case was a suspected case for whom testing for SARS-CoV-2was inconclusive, and a confirmed case was defined as a person with polymerase chain reaction (PCR) laboratory confirmation of SARS-CoV-2 infection, irrespective of clinical signs and symptoms.
A primary case was defined as a confirmed case reported in the national electronic surveillance system and a contact was defined as any individual who had sustained contact with a primary case within a one-meter radius for more than 15 minutes up to2 days before and 14 days after the onset of symptoms. Contacts included household members, visitors, neighbors, colleagues, teachers, classmates, co-workers, and social or health workers. A high-risk contact was defined as having close contact (i.e., being within 1 meter for >15 minutes) with a COVID-19 case or providing patient care to a case without wearing any personal protective equipment. A low-risk contact was defined as being in an enclosed space with a COVID-19 case but without having close contact [14].
A secondary case was defined as a contact of the primary case that became a confirmed case with SARS-CoV-2 positive test result during the study follow-up period.
An electronic questionnaire was developed in Go.Data [15]. Demographics, clinical status, outcome (alive, dead), severity (hospitalized, non-hospitalized), symptoms, self-reported pre-existing medical conditions including diabetes, HIV, cancer, heart disease, chronic lung disease, and obesity, and history of exposure were collected using Go.Data by trained data collectors through a combination of face-to-face or telephone interviews with the case and a household member. During a follow-up visit, 14-21 days later, data were collected on case outcome status, symptoms during the entirety of illness, and clinical course complications. For contacts, demographics, and risk levels (high, medium, and low) were collected at enrollment and symptoms were recorded daily for 21 days for all contacts. Sample collection for contacts occurred on enrollment and between days 14-21 (Figure 1). Contacts that reported symptoms at any time during the follow up period were tested using PCR, if positive classified as secondary cases and their clinical characteristics were collected using Go.Data accordingly. Day 1 in the study is the day of identification of a contact and may not necessarily be day 1 of exposure or start of the incubation period.
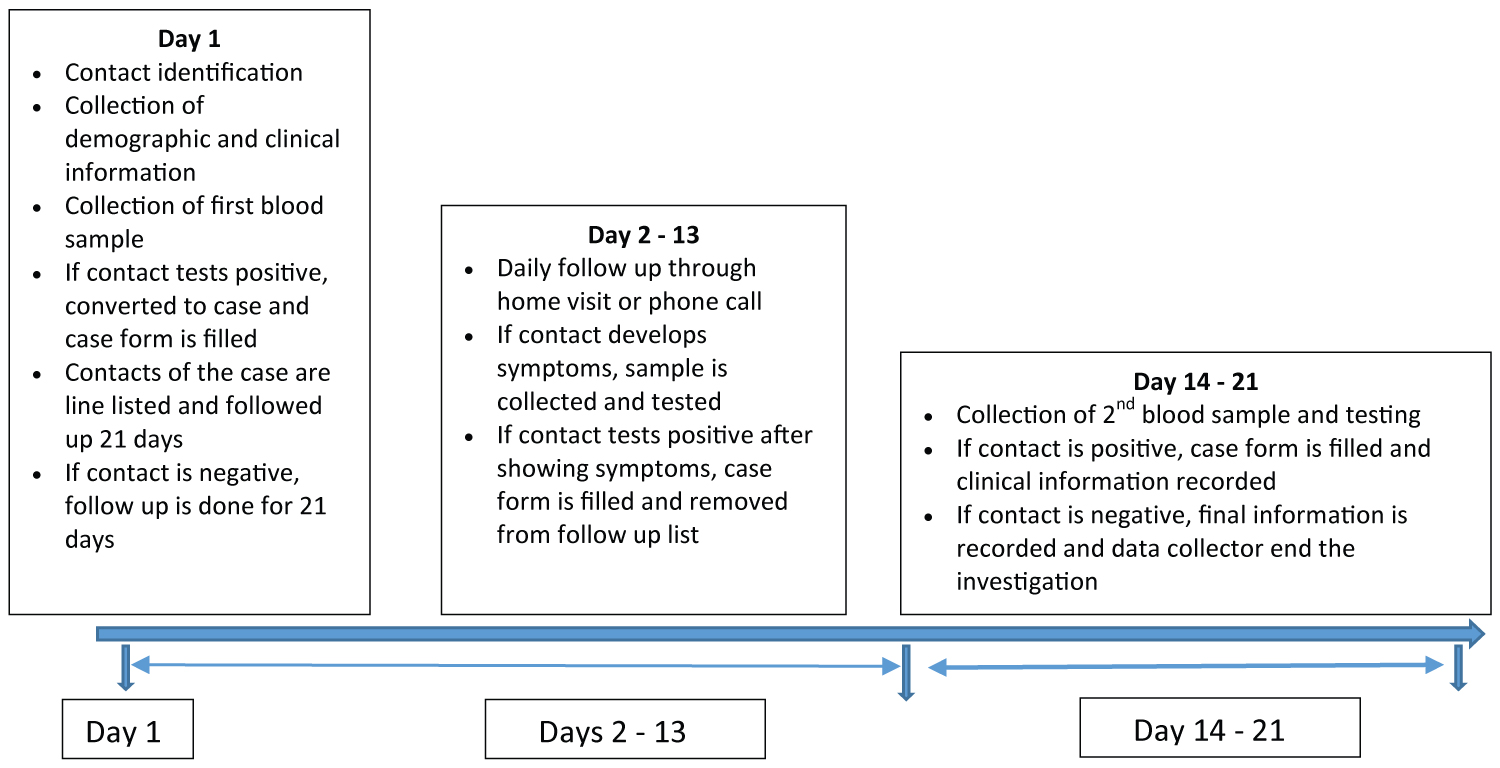 Figure 1: Timeline of data collection during the First Few X number of COVID-19 cases and contacts, Sierra Leone, June 26- September 30, 2020.
View Figure 1
Figure 1: Timeline of data collection during the First Few X number of COVID-19 cases and contacts, Sierra Leone, June 26- September 30, 2020.
View Figure 1
Collected data were downloaded from Go.Data software and cleaned, and computed preliminary analysis using Stata version 15. Further analysis was done using R version 4.0.2 and RStudio version 4.0.2. Frequencies and proportions were computed to determine the distributions of demographic, and clinical characteristics. Using the records on contacts per case, the sum of contacts for each case was computed in R. This data was also used to determine the reproductive rate, chain of transmission, and the mean of the number of contacts who became infected per case.
Administrative approval was given to conduct the study by the Sierra Leone Ministry of Health and Sanitation as part of the COVID-19 response. Written informed consent was obtained from all cases and contacts willing to participate in the study. For children under 18 years, their assent was sought, and informed consent was also obtained from a parent or legal guardian. Each participant was informed about the nature and, potential risk of participating in the study. Each participant was informed that participation in the investigation was voluntary and he or she was free to withdraw, without any justification, from the investigation at any time without any consequences and without affecting professional responsibilities. The purpose of the investigation was explained to all study subjects. All subjects who participated in the study were assigned unique identification numbers to maintain confidentiality. Only the investigation team and individuals within the Ministry of Health had the ability to link the identification numbers with individual study participants. This activity was reviewed by CDC and was conducted consistent with applicable federal law and CDC policy and was deemed not to be research as defined in 45 CFR 46.102(l) and IRB review was not required.
A total of 1,163 participants, consisting of 160 primary and secondary cases and 1,003 contacts were identified from June 26-September 30, 2020.
Among the 160 COVID-19 cases identified, 63% (101/160) were primary cases and 37% (59/160) were secondary cases. The majority of the cases (75%; 120/160) were from Western Area Urban District. More than half (56%, 90/160) of the cases were aged between 18 - 44 years, and 56% (90/160) of the cases were males. Twenty-one percent (34/160) of the cases were students, and 16% were government employees (26/160). Of all the investigated cases, 14% (23/160) were hospitalized and 4% (6/160) died. Of the 23 hospitalized cases, 48% (11/23) of them were aged 45 years and above. Out of the six deaths, five had a reported pre-existing medical condition.
Of the 1,003 contacts, 55% (551/1,003) were aged 18-44 years and 27% (268/1,003) were aged less than eighteen years. The majority of the contacts were male (55%, 553/1,003) (Table 1).
Table 1: Demographic characteristics of the First Few X number of COVID-19 cases and contacts Sierra Leone, June 26-September 30, 2020. View Table 1
Among the 101 primary cases, none reported to have had a travel history from COVID-19 affected country, 11% (11/101) had contact with probable or confirmed COVID-19 case, 13% (13/101) reported an outpatient facility visit and 7% (7/101) reported travel history within the country.
Among the 59 secondary cases, 19% (11/59) reported to have had contact with probable or confirmed COVID-19 case, however, the remaining 81% (48/59) reported having no knowledge on whom they were exposed to. The majority, 95.5% (21/22), of the cases aged less than eighteen years had contact with a suspected or confirmed case.
Forty-five percent (72/160) of the total COVID-19 cases were symptomatic at enrollment. The common signs and symptoms reported were cough, headache, fatigue, and loss of appetite (Figure 2). Of the total cases that were symptomatic at enrollment, 86% (62/72) were primary cases, 63% (45/72) were male, and 56% (40/72) were aged 18-44 years. More than a half, of the COVID-19 cases (55%, 88/160) were asymptomatic at enrollment, of which 51% (45/88) were male, and 57% (50/88) were aged 18-44 years. The majority (83%, 49/59) of the secondary cases were exposed to an asymptomatic primary case and the remaining (17%, 10/59) were exposed to a symptomatic primary case.
 Figure 2: Clinical presentation of the First Few X number of COVID-19 cases, Sierra Leone, June 26-September 30, 2020.
View Figure 2
Figure 2: Clinical presentation of the First Few X number of COVID-19 cases, Sierra Leone, June 26-September 30, 2020.
View Figure 2
Pre-existing conditions were reported by 19% (30/160) of the cases with some having more than one pre-existing condition. Of these, 37% (11/30) were hospitalized, 57% (17/30) were male, and 53% (16/30) were aged 18-44 years. The most reported pre-existing medical conditions were obesity 27% (8/30) and diabetes 23% (7/30). Of the total 30 cases with pre-existing conditions, 27% (8/30) had obesity, 23% (7/30) had diabetics, and 17% (5/30) had heart disease.
There were 6 deaths, 4 of them had pre-existing conditions: 2 had heart disease, 1 had diabetes, 1 had asthma, and 2 were unknown if they had pre-existing conditions.
Of the enrolled contacts, 59 tested positive for SARS-CoV-2, becoming secondary cases, giving a secondary attack rate of 6% (59/1003), and a reproductive number of 0.36. Of the secondary cases, 97% (57/59) cases were confirmed SARS-CoV-2 positive on day 1, 3% (3/59) on day 7of laboratory testing, and zero tested positive between days 14 -21 of laboratory testing.
This study provided clinical and epidemiological characteristics of the first few cases of COVID-19 in Sierra Leone. Most of the cases investigated were asymptomatic; higher proportion of secondary cases were asymptomatic, compared to primary cases, and were most likely identified due to testing regardless of symptoms. Findings from the study were used to inform government policy to manage COVID-19 in Sierra Leone. For instance, this study informed the policy on quarantine and laboratory testing. Testing of contacts was reduced from 14 days after identification to 1 day after identification. Similarly, quarantine duration for asymptomatic contacts was reduced from 14 days to 7 days. Our study confirmed that comprehensive local epidemiological information during the initial stages of an outbreak can be used in the control of emerging diseases [16,17].
Study findings about gender, presence and types of symptoms, clinical course, and medical comorbidities were essential to informing possible public health interventions. In Sierra Leone, more men were affected than women early in the outbreak. This finding may be attributed to the Sierra Leone culture, where men tend to have more person-to-person contact as they work outside of their home than women who usually remain at home during the day [18]. This finding is similar with other studies conducted in other African countries [15,16,19]. More research is needed to understand the factors behind the gender difference in COVID-19 infection rates.
This study indicated that majority of COVID-19 cases were asymptomatic, which is consistent with other studies [16,19]. However, in the initial stages of the pandemic, this information was not available. The finding that most secondary cases were exposed to asymptomatic primary COVID-19 cases was concerning because asymptomatic cases may infect more people if preventive measures are not taken. This information was unknown during the early phase of the response and was used to develop targeted community screening tests in hotspot communities to identify asymptomatic cases, appropriate use of face masks and hand washing, and later, mass vaccinations to reduce community transmission. The finding that 96.6% of the secondary cases tested positive on day 1also provided information that the virus can be detected in the first few days of infection. Therefore, day 1 laboratory testing of contacts exposed to COVID-19 cases was added and proved to be critical for the timely detection of COVID-19 cases in Sierra Leone.
In our study, cough, headache, and fatigue were the most commonly reported symptoms among the COVID-19 cases. Similar findings have also been reported in other countries [20-23], and are used by the disease surveillance program to screen patients presenting with these symptoms as suspected cases and to prioritize them for testing. Few patients in our data presented with atypical symptoms such as diarrhea, vomiting, nausea, and rash. Other studies conducted in other countries also reported similar findings [21,22]. Loss of taste and smell were not symptoms commonly seen in Sierra Leone, contrary to findings in other countries [23-25].
Although not common in our data analysis, patients presenting with atypical COVID-19 symptoms, such as diarrhea, vomiting, and loss of smell were included in the case definition for COVID-19 cases in Sierra Leone. The difference in clinical presentation of the virus may be attributed to the differences in the genotype of the virus, study setting, and sample size, and therefore may change as the pandemic progresses [26-28].
The low hospitalization rate among the COVID-19 cases in our study indicates that the disease was not severe in the initial months of the outbreak in Sierra Leone. One possible reason for the low hospitalization rate could be that most of the cases in the study were young. Studies in other countries have shown that older age, and chronic comorbidities have been found to lead to poor clinical outcomes in COVID-19 patients including hospitalization and death [29,30].
Although the case fatality was low in the current study, 4of the 6deceased COVID-19 cases had pre-existing medical conditions mainly diabetes, heart disease, and obesity which may have contributed to their death. Based on this data as well as global evidence, the Government of Sierra Leone targeted persons with pre-existing medical conditions such as diabetes and hypertension with interventions such as vaccinations, and appropriate treatment, and use of face masks aimed to prevent morbidity and mortality.
Our study has three major limitations. The first is the use of convenience sampling that could have introduced selection or misclassification bias. Moreover, due to case ascertained study design, there is high probability of missing cases and contacts, due to majority of cases being asymptomatic and due to recall bias. Lastly, the analysis does not account for how the dynamics of transmission were altered by preventative measures such as wearing face masks or travel restrictions.
In the early months of the COVID-19 outbreak in Sierra Leone, most cases were male and appear to have been infected by asymptomatic individuals. Symptomatic cases commonly experienced cough, headache, and fatigue, and higher number of individuals with medical comorbidities were hospitalized, compared to individuals with no underlying medical conditions. Information regarding case characteristics early in the outbreak were helpful to determine which individuals were most likely to be infected, and which individuals were likely to suffer from adverse clinical outcomes. The findings of this study helped the Government of Sierra Leone to develop and revise its policies and response strategies including identification of high-risk population groups for screening, case management, and care to reduce preventable COVID-19 related deaths. Response measures such as duration of quarantine, case investigation, and testing protocols were also revised based on the findings of this study. Recommendation for the general population and risk communication materials were developed based on these findings. Before the implementation of COVID-19 vaccine, the findings of this study were essential in determining the public health recommendations such as mass community testing, mandatory use of face masks, and hand washing and to increase and prioritize testing of symptomatic persons with pre-existing conditions. Later, highest priority target populations for COVID-19 vaccination were also identified by these findings, especially when limited vaccines were available in the country. This experience determined that early investigational protocols such as the first few cases study can be of great utility during outbreaks of diseases with limited information and can provide local data for decision making.
We would like to acknowledge the World Health Organization (WHO) for providing the FFX generic protocol and the Go.Data software from which we adapted the protocol and data collection tools for this study. We would like to acknowledge WHO Sierra Leone Country Office staff, specifically Dr. Charles Njuguna, Dr. Wilson Gachari and Mr. Mugagga Malimbo for supporting the implementation of this study. The authors extend sincere thanks and appreciation to the Western Area Urban and Western Area Rural District Health Management Teams, Sierra Leone Ministry of Health and Sanitation and the Sierra Leone Field Epidemiology Training Program for the assistance in ensuring this project becomes a success.
This study was funded from the Centers for Disease Control and Prevention through the African Field Epidemiology Network Cooperative Agreement.
The authors declare no conflict of interest. No copyrighted materials were used in developing this article.
Disclaimer - The findings and conclusions in this report are those of the author(s) and do not necessarily represent the official position of the U.S.Centers for Disease Control and Prevention.