Hypertension is associated with impaired left ventricular (LV) diastolic function. However, the impact of stress on LV filling of treated hypertensive individuals is unclear. Fourteen hypertensive patients (mean age 55 years) and 14 age-sex matched normotensive controls underwent Doppler echocardiographic studies at rest and one minute following supine ergometer bicycle. The evaluations were done before and under treatment with diltiazem (180 mg/day) during 24 weeks. Resting blood pressure control was achieved under treatment. However, the pressure-rate product was unchanged during exercise. Left ventricular mass index decrease from 153 ± 7 to 129 ± 8 g/m2 (p < 0.01). There was a significant increase in the resting peak velocity of early filling (E) (57 ± 3 VS. 48 ± 4 cm/s, p < 0.01) and E/A ratio (1.1 ± 0.1 vs. 0.9 ± 0.1, p < 0.01) while peak velocity of late filling (A) remained unchanged (56 ± 4 vs. 57 ± 3 cm/s). The deceleration time was Shortened (p < 0.05). With exercise, percentage of change in E (%E) and E/A (E/A%) were lower in patient than control group (21 ± 6 vs. 37 ± 8 and 5 ± 5 vs. 21 ± 12 respectively; p < 0.05 for both). The treatment didn't improve these differences. These findings suggest that LV filling in response to stress remain blunted in treated hypertensive patients.
Hypertension, Exercise, Diastolic function, Diltiazem
Diastolic dysfunction is a sensitive marker of congestive heart failure [1,2]. Most studies of diastolic function were performed under resting conditions and showed a deleterious effect of elevated blood pressure (BP) on filling indices [3-5]. This abnormal resting left ventricular (LV) filling is reversed with antihypertensive treatment that effectively lowers BP and reduces LV hypertrophy (LVH) [3,6].
We and others reported exercise-induced normal physiologic variation of LV filling, defined by an enhancement in both early (E) and late (A) filling with the ratio of filling waves (E/A) remaining constant [7,8] in normotensive as well as hypertensive individuals. However, others reported that early filling and E/A ratio were still decreased 15 minutes after stopping exercise in hypertensive patients [9]. Another study [10] on hypertensive patients with normal cardiac function at rest reported a decline in pump function due to abnormal LV filling during exercise. Thus, we hypothesized that hypertensive patients who had abnormal filling pattern at rest would show a lesser contribution of early filling phase to the exercise demand. We further hypothesized that antihypertensive treatment would normalize this response. To test these hypotheses, we followed up a group of hypertensive patients with pulsed Doppler examinations for a period of 24 weeks.
During one-year period, a total of 14 never treated hypertensive patient with LVH determined by echocardiography were selected from the annual screening program of the hypertension outpatient clinic at the Tarami Hospital (Nagasaki, Japan). There were 11 men and 3 women ranging from 41 to 66 years (mean: 58 years). Blood pressure screening was done on 3 consecutive visits at 2 week-intervals. At each visit BP readings were done after 15 minutes rest in sitting position using a manual sphygmomanometer. Subjects were eligible if BP readings were greater than 140 mmHg for systolic and 90-110 mmHg for diastolic BP on at least two occasions during the screening period. Exclusion criteria included obesity, mitral valve stenosis and/or regurgitation, previous antihypertensive treatment and positive family of myocardial infarction. Each had a complete clinical evaluation to exclude coexisting cardiovascular disease. This evaluation included a routine laboratory screening (i.e., complete blood count, serum electrolytes, and kidney function), chest x-ray, 12 lead electrocardiogram and echocardiography. A subset of 14 age-sex matched healthy volunteers was randomly selected for comparison. The protocol was approved by our institution's ethical committee and informed consent was obtained from all participants.
Complete two-dimensional directed M-mode echocardiographic examination was performed with an Aloka SSD-ultrasound system with 3.5 or 2.5 MHz transducers. Measurements were performed according to the recommendations of the American Society of Echocardiography [11]. The average of five cardiac cycles was used for data analysis. Left ventricular mass (LVM) was calculated using an anatomically validated formula [12]. Echocardiographic LVH was defined as an interventricular septal and posterior wall thickness greater than 12 mm at end-diastole and/or left ventricular mass index greater than 130 mg/m2. End-Systolic stress and midwall fractional shortening were calculated as previously recommended by the Simone, et al. [13].
Mitral flow recordings were obtained in the apical two-chamber view with the sample volume placed at the tips of the mitral valve leaflets using a 2.5 MHz transducer. Traces of five consecutive cardiac cycles having the highest velocity in early diastole were recorded at rest and following exercise on a supine ergometer. Post-exercise filling waves obtained within one minute after stopping the exercise as previously described [7]. Peak of early filling, late filling, the ratio of early to late filling, and the deceleration time of early filling (Dec.T) were examined by three experienced echocardiographers blinded to the subject status. The stress response was defined as the percentage change from rest to one minute following exercise in E (E% change); A (A% change) and E/A ratio (E/A % change). After the initial Doppler echocardiographic exploration, patients were treated with 180 mg slow-release diltiazem once a day during 24 weeks. Subsequent visit was performed every 2 weeks and the Doppler echocardiographic studies were done after 12 and 24 weeks. The explorations were performed at the same time in the morning. At each visit tablet counting assessed compliance. Sonographer was blinded to the clinical status of the study participant. The Doppler indices interobserver and intraobserver variability in our laboratory have been previously reported [8,14].
A moderate exercise protocol was performed on a supine ergometer driven at constant speed of 50 cycles/min. Starting at 25 W the workload was stepwise increased by 25 W every 3 minutes until the attainment of a maximum workload of 75 W designated for comparative purpose. During the test, BP, heart rate and electrocardiography were continuously monitored with an automatic device (Nippon Colin STBP-6807, Tokyo, Japan) and recorded once every minute.
Data are expressed as mean ± standard error. Where appropriate, Student t test and analysis of variance with repeated measures were used to assess statistical significance set at a probability of less than 0.05. Individuals served as their own controls in the follow-up period.
All individuals had normal body mass index at baseline and in the follow-up period. The higher resting BP in hypertensive group was normalized during the follow up period as shown in Table 1. In this group peak exercise BP and heart rate were similar before and during treatment. No patient was forced to discontinue the exercise because of cardiac symptoms. A predicted submaximal heart rate of 70% to 75% (116 to 124 beat/min) was reached by all participants within 6 to 9 min. No ST-T depression was observed. The exercise-induced changes in Doppler signals were recorded at a mean heart rate of 80 ± 3 beats/min. The ratio of systolic BP at 1 minute to its peak exercise was similar in both groups (control: 0.8 ± 0.1; 0.8 ± 0.2 patients).
Table 1: Echocardiographic measurements in control and patient groups. View Table 1
Table 2 displays echocardiographic data. Interventricular septum and LVMI were increased in hypertensive patients. No difference was observed in wall stress and systolic function between the two groups. In treated patients, a decrease of LVMI was observed by 12 weeks. Further reduction was observed at 24 weeks. Left ventricular regression accounted for a significant reduction in wall thickness (p < 0.01). No significant change was observed in LV inner dimension during the treatment period. Finally, midwall systolic function was similar in the two groups and remained unchanged during the follow-up period.
Table 2: Blood pressure, heart rate and double product in controls and hypertensive patients at different stages. View Table 2
Resting peak of early filling velocity E and E/A ratio were lower in hypertensive patient than control group. Changes of resting LV filling indices in hypertensive group throughout the treatment period are shown in Figure 1. E and E/A normalized by 12 weeks and remain stable at the end of the study. Deceleration time of early filling progressively shorten during the follow-up period. With exercise, percentage of change in E %E and E/A (E/A%) were lower in patient than control group (21 ± 6 vs. 37 ± 8 and 5 ± 5 vs. 21 ± 12 respectively; p < 0.05 for both). The treatment didn't improve these differences.
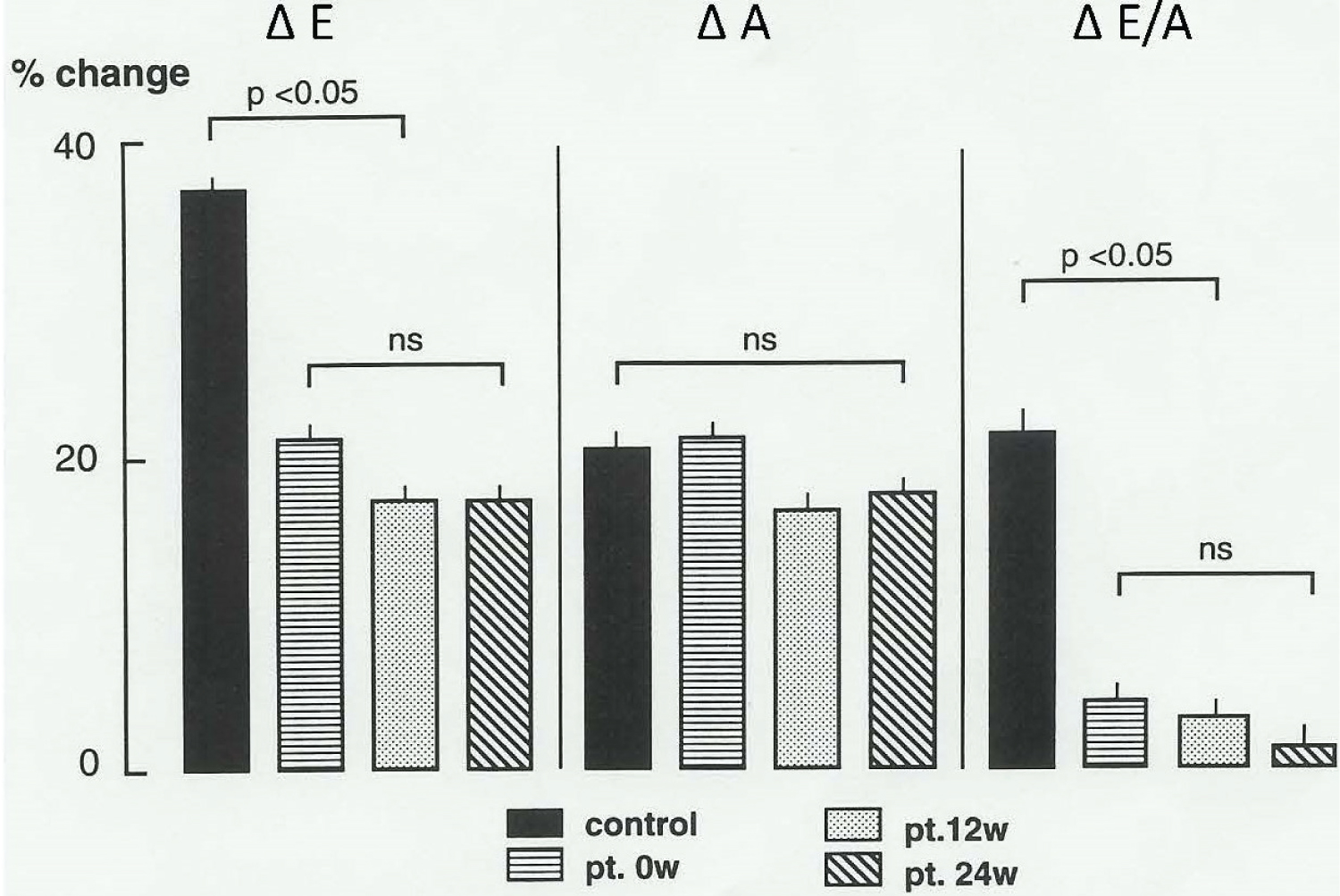 Figure 1: Left ventricular filling: Follow up of 12-weeks and 24-weeks.
Figure 1: Left ventricular filling: Follow up of 12-weeks and 24-weeks.
ΔE: Early left ventricular filling (percent change); ΔA: Late left ventricular filling (percent change); ΔE/ A: Ratio of filling waves (percent change); ns: Not significant; Control: Normotensive patients
View Figure 1
The major finding of this study was that the LV filling response to exercise in hypertensive patients remained blunted despite the antihypertensive therapy. This contrasts with improvement of resting LV filling under anti-hypertensive treatment suggesting that stress plays an important role on the pathogenesis of congestive heart disease in hypertension.
Conflicting data have been reported in regard to the effect of exercise on LV filling profile. The variability of E/A ratio in response to exercise has been shown to be dependant on population characteristics and testing modalities. Compared to control, patients in the present study exhibit a lesser magnitude of increase of early filling and E/A ratio suggesting inadequate adaptation to exercise demand. It is possible that the diltiazem effect (i.e., diminution of cytolosic calcium) may have been overridden by the neurohumoral changes associated with exercise rendering stress LV filling again abnormal. It is also possible that persistent microangiopathy, interstitial fibrosis, decreased coronary flow reserve contributed to the impairment of ventricular relaxation in our patients [15-18]. Even so, impairment of LV early filling is complex and might be due to altered load condition, active and passive LV relaxation properties. Data in the present study can not specifically identify which of these factors or their combination account for our findings.
Our results on resting LV function are consistent with some [3,19] but not all previous studies [20,21]. Improvement of diastolic function was shown to be associated with reduction BP and LVM. Studies without LV mass reduction also show no improvement of LV filling characteristics.
Whether the observed LV filing improvement in our study accounted solely for LVH regression remains unclear. Our hypertensive subjects showed an impaired resting LV filling when compared to normotensive counterparts. A progressive increase of resting E/A ratio was observed during the treatment period owing to an increase in early filling and reflecting a possible improvement of diastolic function. Given that patients had both chamber and midwall systolic function well preserved throughout the study period and that there was no sign of disease progress, it is unlikely that the progressive increase of E/A ratio reflected pseudonormalisation [22,23].
Abnormality of diastolic function was consistently shown to parallel BP increase. The potential weight of the contribution of BP is attested by the following: after 12 weeks, if BP is normal (i.e., at rest), diastolic function is normal; if BP is abnormal (i.e., during exercise), diastolic function is abnormal. As hypertrophy is present under both conditions, this is evocative of the predominant influence BP on ongoing ventricular filling process.
The findings are provocative, however, there were number of limitations to our study. The invasive determination of pressure-volume is the gold standard of diastolic function. However, the noninvasive assessment of diastolic function by pulsed Doppler has been well validated against invasive standards with an excellent correlation [24]. Doppler recording during exercise provides the clearest information about the change of filling pressure. But it is often difficult to obtain Doppler signals due to increased respiratory rate; thus, it is reasonable to also consider obtaining the filling signals immediately after stopping the exercise. Recently, tissue Doppler, color mapping and strain have been recognized as more sensitive modality of diastolic function assessment. We choose only to use pulsed Doppler given that the E/A ratio is yet the only diastolic function parameter that has been proven to predict cardiovascular disease [25,26].
Observer bias is always a concern in open study. In our study care was taken to ensure that measure of diastolic function was observer-blind. While no group difference was found for an indirect measure of myocardial ischemia (i.e., the ratio of systolic BP at 1 minute to its peak exercise) direct measure of myocardial perfusion would help define the role of myocardial hypoxia on the undergoing filling process.
Because of the sub-maximal exercise protocol in the present study the potential impact upon exercise tolerance could not be determined. Previous studies have demonstrated the importance of diastolic dysfunction on exercise performance [10,27,28].
In summary, our findings suggest that exercise may unveil diastolic dysfunction in controlled hypertension. Further prospective large studies are needed to establish the potential benefit of assessing exercise related LV filling profile as marker of hypertensive disease progression.