Thyroid diseases are prevalent in Malaysia. A community health survey was undertaken to improve community awareness of the risks of common non-communicable diseases such as thyroid disorders and the benefits of screening for prevention and early detection of these conditions. Thyroid stimulating hormone (TSH) is the main biomarker and initial test for thyroid dysfunction. However, no formal reference interval (RI) data for TSH is available in Malaysia and South East Asia. All TSH reference intervals quoted in laboratory reports are from small verification studies laboratories conducted on 20 samples and thereafter adopting the manufacturer's data. We undertook to derive TSH RIs for Malaysia using the gold standard National Academy of Clinical Biochemistry (NACB) guidelines which state reference intervals should be established from healthy volunteers without personal/family history of thyroid dysfunction, no goiter/detectable thyroid autoantibodies and taking no medications.
Health screening was conducted during the 2016 Kembara Mahkota community event in Johor, Malaysia. Over one week, blood samples were taken from willing participants who attended the event after completing a brief health questionnaire. Samples were collected from 9.00 am to 2.00 pm daily and transported directly to the laboratory where they were processed within 4 hours. Samples were tested for a range of biomarkers (lipid, diabetes, cancer, and thyroid) on the Abbott Architect analyser at the Johor Specialist Hospital. Individuals identified as high risk were requested to seek further medical evaluation. Children, pregnant subjects, those with active medical conditions or on any medications were excluded. TSH and anti-thyroid peroxidase antibodies (anti-TPO) results were available from 2833 subjects. All samples with detectable TPO-Ab were excluded. Statistical analyses were performed using MedCalc software v 18.11.6 (Ostend, Belgium).
The distribution of TSH was slightly skewed to the right in the 2124 samples recruited, but log TSH was Gaussian in distribution.The parametric 95% reference interval for TSH in women is 0.47-3.08 and 0.47-2.84 in men. When compared to 5 other reported studies of Architect TSH RIs in the literature, our values for the lower reference limit lies in the middle of the range while the upper reference limit of TSH is amongst the lowest. Reference subjects were 18-80 years old (mean 40.3 ± 12.6). Mann-Whitney test for gender effects achieved statistical significance (p = 0.0388). When the group was divided by age bands in decades, the Kruskal-Wallis test for effects of age on TSH was not significant for both genders.
Community health projects such as ours are required to raise awareness of the risks of common preventable diseases, and to encourage individuals to participate in health checks and disease screening programmes. Besides, a study such as this has firmly established the TSH RIs for the Architect assay.
Thyroid disease, TPO-Ab, TSH, Screening, Reference intervals
RI: Reference Intervals; TSH: Thyroid Stimulating Hormone; TPO-Ab: Thyroperoxidase antibodies
Laboratories put out Reference Intervals (RIs) associated with all the results they generate including that for Thyroid Stimulating Hormone (TSH). Clinicians regard TSH as the most accurate and reliable measure of thyroid dysfunction [1]. Thus, accurate and reliable TSH RIs are vital. RIs have been variously derived even within the same laboratory. At the lowest level manufacturers' package inserts or textbooks are used. Beyond that, laboratories may refer to the published literature or to institutions in the same city using the same instrument [2]. According to guidelines recommended by expert bodies like the Clinical Laboratory Science Institute (CLSI) and the International Federation of Clinical Chemists (IFCC) [3] verification of these transferred external RIs can be performed on a small number of healthy subjects (n = 20). This approach is very popular with many laboratories. However, the CLSI gold standard for laboratories is to derive their own RIs directly from a sufficiently large reference population (> 120). As the direct approach is quite involved and requires effort, simpler indirect methods through data mining of large data sets in the Laboratory Information System is gaining traction [4,5] as exemplified by a recent study involving 136,650 subjects [6].
Despite the large body of literature on RIs [7] controversy regarding the best approach for RIs still remains [8,9]. Besides, the data generated through such RI efforts can be influenced by the inadvertent inclusion of subjects with subclinical disease and the statistical treatment used [2,10,11]. In determining the TSH reference range, the National Academy of Clinical Biochemistry (NACB) recommends that TSH reference intervals should be established from the 95% confidence limits of the log-transformed values of at least 120 healthy volunteers without personal or family history of thyroid dysfunction, no goiter, no detectable thyroid auto-antibodies and taking no medications [12]. In one study, applying these stringent NACB guidelines resulted in a decrease of the TSH Upper Reference Limit (URL) from 4.43 to 3.37 mU/L while the Lower Reference Limit (LRL) remained unchanged [13]. The decrease in URL was mainly attributable to the exclusion of TPOAb-positive individuals. In recent years, the TPOAb assays have improved significantly [14].
A recent comparison of RIs for TSH showed variation between the common analytical platforms in use - Abbott Architect, Beckman DxI, Roche Cobas and Siemens Advia Centaur. In 261 subjects the LRL ranged from 0.51-0.63 while the URL ranged from 3.60-4.31 [15]. Thus, clinicians and patients are faced with the prospect of method related RIs [16] when evaluating their test results despite initiatives to standardize [17] or harmonize TSH assays [18,19]. For the Abbott Architect we identified 12 TSH RI studies in the literature (see Table 1) but only 6 are NACB compliant with direct TSH testing and TPO-Ab screening. Even on the same analytical platform the LRL for the Abbott Architect TSH can vary from 0.17-0.54 mU/L while the URL ranged from 2.82-6.2 mU/L [20-29].
Table 1: Reported reference intervals for the Abbott Architect TSH. View Table 1
As far as we are aware, there has been no formal TSH RI study for non-pregnant adults in the literature for the South-East Asian region. Thus, we undertook this study to determine the TSH RIs in ambulatory adults from a large community health survey using stringent NACB criteria.
The study was approved by the Johor State Department of Health. All participants provided signed informed consent. Health screening was conducted during the 2016 week-long Kembara Mahkota community event in Johor, Malaysia.
Blood samples (10 mL) were collected in vacutainer tubes (SST Becton-Dickinson) between 9.00-14.00 daily from willing participants after completing a brief self-administered health questionnaire with respect to any medical problems (personal or family) and medications. Children (< 18 years), pregnant subjects, and those with active medical conditions or on any medications were excluded.
All samples were transported to the Johor Specialist Hospital laboratory daily and processed within 4 hours. Blood was centrifuged at 3000g for 10 minutes and analysed within 2 hours thereafter. Samples were tested for a range of biomarkers (lipids, diabetes, cancer and thyroid) on the Abbott Architect CI8200 analyser. Individuals with abnormal results were advised to go for further medical consultation.
The Architect TSH and TPO-Ab assays are based on chemiluminescent microparticle immunoassay. The Architect TSH assay, available since 2002 [30], is a third generation immunometric assay with a functional sensitivity of < 0.004 mU/L. This TSH assay, traceable to WHO 2nd IRP 80/558, has a manufacturer provided RI of 0.35-4.94 mU/L. The third-generation Architect TPO-Ab assay has been available since 2007 [31]. The assay has a stated analytical sensitivity of 1 U/mL and any TPO-Ab result above 1 U/mL was considered positive.
Thyroid Stimulating Hormone (TSH) and anti-thyroid peroxidase antibodies (anti-TPO) results were available from 2833 subjects. Results from the Architect were extracted into Microsoft Excel. For our study 2734 subjects had TSH and TPO-Ab results. Subjects with positive TPO-Ab results were excluded from the reference interval study.
The TSH data were analyzed according to CLSI guidelines with outlier screening by the Tukey method. Reference intervals were derived by non-parametric methods and reported as the 2.5 and 97.5 percentile ranges. The Kolmogorov-Smirnov test was used to assess whether the data were Gaussian. Differences in TSH between genders were assessed by the Mann-Whitney U test while the Kruskal-Wallis test was used to examine the influence of age on TSH across the decades. Continuous variables are represented as median values with interquartile range while categorical variables by numbers and percentages. All hypothesis testing was 2-tailed; p values < 0.05 were considered significant. Statistical analyses were performed using MedCalc software v 18.11.6 (Ostend, Belgium).
In total there were 2833 sets of TSH & TPO-Ab results (1498 females and 1335 males). After exclusion of TPO-Ab positive subjects we were left with 2171 participants (1201 female, 970 male). Outlier TSH values (33 female, 24 male) detected by the method of Tukey were excluded leaving a final tally of 2124 persons (1168 F, 956 M) for TSH RI determination. The flow chart for the RI recruitment pathway is shown in Figure 1.
 Figure 1: Recruitment pathway for TSH Reference group.
View Figure 1
Figure 1: Recruitment pathway for TSH Reference group.
View Figure 1
The age of the reference subjects (range, mean, SD) were: 18-80, 40.3, 12.6 for women and 18-86, 42.5, 12.5 for men respectively. The distribution of TSH values in both men and women were non-Gaussian with a right skew (Figure 2a and Figure 2b). However, after log transformation the back-transformed TSH values are indeed normally distributed - Kolmogorov-Smirnoff test (p > 0.1) for both gender.
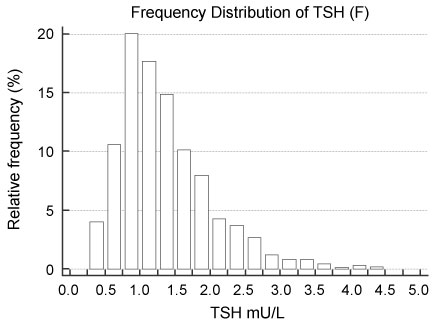 Figure 2A: Distribution of TSH in thyro-healthy women.
View Figure 2A
Figure 2A: Distribution of TSH in thyro-healthy women.
View Figure 2A
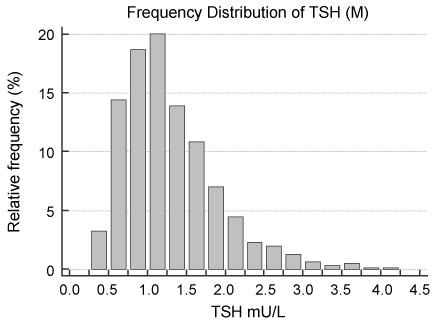 Figure 2B: Distribution of TSH in thyro-healthy men.
View Figure 2B
Figure 2B: Distribution of TSH in thyro-healthy men.
View Figure 2B
The TSH values in women are slightly higher than men and are significant (Mann-Whitney test p = 0.0388; test statistic Z 2.066) as shown in Table 1. The parametric 95% reference interval for TSH in women is 0.47-3.08 and 0.47-2.84 in men.
In our reference population the Kruskal-Wallis test did not show any influence of age on TSH across the age bands although there is a slight uptrend in TSH values towards the older subjects.
This large study was performed to obtain TSH RIs in a multi-ethnic South-East Asian population, an endeavour not previously undertaken. With a large ambulatory cohort of 2124 subjects strict NACB criteria was enforced with respect to undetectable TPO-Ab to exclude those with subclinical thyroid disease as far as possible.
We report three major findings. All previous RI studies on the Architect TSH were not fully NACB compliant as the TPO-Ab cut-offs they employed were the derived TPO-Ab RI in “normal” euthyroid subjects and not undetectable TPO-Ab as specified by NACB. Thus their RIs might have inadvertently included subjects that may have subclinical thyroid disease. From the published literature on the same Architect platform (see Table 1) our derived TSH lower reference limit lies in-between the studies using NACB criteria of non-elevated TPO-Ab while the upper reference limit is among the lowest. This is to be expected as TPO-Ab is a major factor influencing TSH levels [32]. With very pristine reference subjects (undetectable TPO-Ab) we found female TSH values statistically higher than males despite looking quite similar (see Table 2). On the Architect platform our female TSH RI is 0.47-3.1 and 0.47-2.8 mU/L in men respectively. This female preponderance of TSH is in agreement with notable published literature [32]. Just as in some studies [33] but not in others [34] age does not affect TSH levels in our cohort. However, there are too few subjects over 60 (see Table 3) for us to make any definitive statements about age and TSH values. The NHANES III data set had over 3000 subjects older than 60 years [34] in contrast to 155 in our study.
Table 2: Distribution of TSH values in women and men compared. View Table 2
Table 3: Kruskal-Wallis analysis of the influence of age on TSH. View Table 3
Our findings extend and corroborate previous work. It is important to highlight that the strength of this work is the large sample size, truly thyro-healthy subjects, and little diurnal influences as blood collection ends at 2 pm each day. In addition, all the TSH samples were analysed in real-time on the same day as they are received. This is true real-world TSH data and not one finessed through freezing samples for a later analysis by an experience operator in a single session to minimise analytical imprecision.
It is important to appreciate that no strategy can ensure absence of ill-health in the reference population or is able to screen out all subclinical disease. Even in the absence of TPO-Ab and ultrasound abnormalities early thyroid dysfunction is difficult to detect [15]. Some patients with undetectable thyroid antibodies may have the sort of thyroid ultrasound abnormalities seen in autoimmune thyroid disease [35]. Moreover, in thyroid antibody negative subjects 70% of those with thyroid sonographic appearance of autoimmune thyroid disease had normal TSH [36]. Besides the TSH reference interval was unaffected when subjects with thyroid antibodies or ultrasound abnormalities were excluded [37,38].
Our study has some limitations that merit consideration. Iodine insufficiency was not excluded from participants but the iodine intake in Malaysia has been shown to be sufficient in earlier studies [39]. We did not conduct a physical examination for goiter but earlier studies in Malaysia showed that goiter occurs in fewer than 2.3% of the population [39]. Data for participants' personal or family history of thyroid disease was provided by a simple self-administered questionnaire. Serum TgAb levels were not measured but previous studies have shown minor or non-existent association between TgAb and TSH levels independent of TPO Ab [33,40]. Smokers were not excluded from the study and smoking has been reported to have lower TSH levels [41].
A secure and robust TSH reference interval from a south-east Asian population is now available. While reference intervals remain an area of intense research and discussion, it is still a work in progress. In the final analysis it is patients that require treatment and not the numbers [1].