Sepsis is one of major causes of mortality in non-cardiac ICU patients. The screening of the patients for sepsis needs a careful evaluation of criteria and confirmation of infection. SIRS criteria for identification of patients with sepsis is not alone sufficient. Lactate has been shown to be a very important severity marker in sepsis cases and has been found to be more useful for predicting the outcome of sepsis than common severity scores such as the Age and Chronic Health Evaluation (APACHE) II score and the Sepsis related Organ Failure Assessment (SOFA) score. The levels of lactate are very high in sepsis patients and earlier studies have tried to correlate the lactate levels with the onset and mortality associated with sepsis. Though evidence exists that lactate can be used as diagnostic marker in critically ill patients all the studies have been done only in Western countries. The present study aims at correlation between lactate levels and sepsis in Indian patients. Our study found significantly higher mean lactate values among patients of sepsis than control group (3.13 ± 2.86 vs. 2.16 ± 1.86 respectively, p = 0.03). The proportion of subjects with high lactate clearance (> 10% clearance) were higher among survivors as compared to non-survivors, while those with low lactate clearance (< 10% clearance) were higher among non-survivors than survivors within 6 hours, and between 12 to 18 hours of admission. Survival analysis between subjects with high and low lactate clearance within 6 hours and between 12 to 18 hours of admission showed a significantly higher survival in the group with high lactate clearance. Thus, this study is one of the earliest ones which shows that lactate levels and clearance could be used as reliable marker for non-cardiac ICU patients with infection and sepsis onset in Indian scenario.
Sepsis is the leading cause of death in the non-cardiac ICU patients. Mortality in Septic Shock ranges from 40 to 60% despite advances in treatment and provision of intensive care. According to Food and drug administration (FDA) sepsis is the second leading cause of death in USA in non-coronary ICU patients. The morbidity and mortality associated with Sepsis is very high with at least 1 out of 4 cases showing death [1,2]. The presence of infection is a confirmatory feature of Sepsis [3,4]. The accurate confirmation of sepsis is based on systemic inflammatory response syndrome (SIRS) criteria which include features such as tachycardia, hypothermia or fever and leukopenia, leukocytosis, or bandemia [5].
Serum lactate levels are traditionally low in normal individuals. The reference range is around 1 ± 0.5 mmol/L and less than 2 mmol/L in critically ill patients. Lactate elevation in blood is seen only in conditions such as hyperlactatemia and lactic acidosis. The values in such cases varies from 2 mmol/L to 5 mmol/L in hyperlactatemia and above 5 mmol/L in lactic acidosis [6]. Thus, serum lactate levels serve as diagnostic maker in critically ill patients [7]. Lactate is a product of anaerobic metabolism. High levels of lactate in tissue is direct indication of tissue hypoxia. Tissue hypoxia for long time can cause irreversible damage to the tissue resulting in its death. Organ failure is common feature seen in septic shock patients. Organ failure is associated with tissue hypoxia. Hyperlactatemia in fact is associated with long periods of tissue hypoxia and subsequent organ failure [8]. Therapeutic interventions to deal with increased blood lactate levels have been going on for long time. Studies done in patients for correcting lactate levels have shown that the controlling lactate alone does not help to improve the outcome in critically ill patients [9,10]. A combination therapy of improving oxygen supply, decreasing lactate would be far more useful in controlling morbidity and mortality [11-14].
Breakthrough studies done on human lactate levels showed that an increase in lactate levels from 2.1 to 8 mmol/L decreased the survival from 90% to 10% [15]. Studies on hemodynamically stable surgical patients found significantly higher levels of lactate at 12, 24, and 48 hours after surgery in non-survivors. Several other studies also have shown that high levels of lactate despite therapy can cause significant drop in survival. Studies from Nguyen HB, et al. [16] showed that a lactate clearance cutoff of < 10% had the maximum sum of sensitivity plus specificity for predicting in-hospital mortality. The lactate clearance was calculated based on lactate at Emergency Department (ED) presentation at hour 0 minus lactate at hour 6, divided by lactate at ED presentation, then multiplied by 100. The subjects were categorized as being in the low-clearance group which was less than 10% lactate clearance or high-clearance group which is > 10% lactate clearance. After 6 hrs of intervention, a lactate clearance of < 10% had a sensitivity of 44.7%, specificity of 84.4%, and accuracy of 67.6% for predicting in-hospital mortality [16]. A multicenter prospective study by Arnold RC, et al. [17] in 166 subjects with severe sepsis showed, lactate non-clearance was found to be an independent predictor of death (odds ratio, 4.9 [confidence interval, 1.5-15.9]). The study concluded that failing to clear lactate during resuscitation carried a high risk of death [17].
To determine the blood lactate levels in patients with sepsis admitted to a respiratory intensive care unit and to correlate with their hospital outcomes in the Indian scenario.
The definition of sepsis has been subjected to intense analysis and it has been continuously updated with time. Sepsis originally was considered on basis of SIRS scale. However, SIRS based observation and identification of sepsis might not be complete as SIRS could be a host adaptive response and nonspecific at times. So other parameters that are non-SIRS based have been also taken into consideration for defining of sepsis. Nonspecific SIRS criteria such as pyrexia and neutrophilia are helpful in diagnosis of infection. Sepsis is a complex phenomenon which involves destruction of own tissue by immune cells. This often is seen in the form of organ failure and dysfunction. The various cellular defects underlying each of the organ failures also need to consider while addressing the sepsis. So, based on these criteria newer definitions of sepsis have been developed. Sepsis is defined as life-threatening organ dysfunction caused by a dysregulated host response to infection. Sepsis is a syndrome which is caused by activity of both infective pathogen and the host immune system. Organ dysfunction is usually assessed on basis of sepsis related organ failure assessment (SOFA) score. A higher SOFA score is associated with mortality. The SOFA score is a six-organ dysfunction/failure score, which is graded from 0 (Normal) to 4 (the most abnormal) providing a maximum of 24 points. qSOFA criteria for assessment of sepsis include three major considerations which are Respiratory rate ≥ 22/min, Altered mentation and Systolic blood pressure ≤ 100 mmHg [18-20].
Systemic inflammatory response syndrome or SIRS is nonspecific and can be caused by ischemia, inflammation, trauma, infection or any other insult to the body. SIRS is not always related to infection. SIRS is defined and identified based on atleast two of the four parameters identified by American College of Chest Physicians (ACCP) and Society of Critical Care Medicine's (SCCM) respectively. The four parameters include: Heart rate > 90 beats per minute (tachycardia), Body temperature < 36 ℃ (96.8 ℉) or > 38 ℃ (100.4 ℉) (hypothermia or fever), Respiratory rate > 20 breaths per minute or, on blood gas, a PaCO2 less than 32 mmHg (4.3 kPa) (tachypnea or hypocapnia due to hyperventilation) and White blood cell count < 4000 cells/mm³ or > 12000 cells/mm³ (< 4 × 109 or > 12 × 109 cells/L), or greater than 10% band forms (immature white blood cells). If any of the two are met, then it is defining as SIRS. SIRS do not lead to sepsis. Sepsis is systemic response to infection. Patients with sepsis have SIRS along with infection which complicates the whole condition. Patients with severe sepsis have SIRS, infection and additional organ dysfunction symptoms with hypoperfusion and hypertension usually seen. The distinction between SIRS and sepsis is important though both are usually used in conjunction with one another [5].
Diagnostic features for sepsis and severe sepsis and septic shock are indicated below.
1. Fever (core temperature, > 38.3 ℃).
2. Hypothermia (core temperature, < 36 ℃).
3. Elevated heart rate (> 90 beats per min or > 2 SD above the upper limit of the normal range for age).
4. Tachypnea.
5. Altered mental status.
6. Substantial edema or positive fluid balance (> 20 ml/kg of body weight over a 24-hr period).
7. Hyperglycemia (plasma glucose, > 120 mg/dl [6.7 mmol/liter]) in the absence of diabetes.
1. Leukocytosis (white-cell count, > 12,000/mm3).
2. Leukopenia (white-cell count, < 4000/mm3).
3. Normal white-cell count with > 10% immature forms.
4. Elevated plasma C-reactive protein (> 2 SD above the upper limit of the normal range).
5. Elevated plasma procalcitonin (> 2 SD above the upper limit of the normal range).
6. Hemodynamic variables.
7. Arterial hypotension (systolic pressure, < 90 mmHg; mean arterial pressure, < 70 mmHg; or decrease in systolic pressure of > 40 mmHg in adults or to > 2 SD below the lower limit of the normal range for age).
8. Elevated mixed venous oxygen saturation (> 70%)‡.
9. Elevated cardiac index (> 3.5 liters/min/square meter of body-surface area)§.
1. Arterial hypoxemia (ratio of the partial pressure of arterial oxygen to the fraction of inspired oxygen, < 300).
2. Acute oliguria (urine output, < 0.5 ml/kg/hr or 45 ml/hr for at least 2 hr).
3. Increase in creatinine level of > 0.5 mg/dl (> 44 μmol/liter).
4. Coagulation abnormalities (international normalized ratio, > 1.5; or activated partial-thromboplastin time, > 60 sec).
5. Paralytic ileus (absence of bowel sounds).
6. Thrombocytopenia (platelet count, < 100,000/mm3).
7. Hyperbilirubinemia (plasma total bilirubin, > 4 mg/dl [68 μmol/liter]).
8. Tissue-perfusion variables.
9. Hyperlactatemia (lactate, > 1 mmol/liter).
10. Decreased capillary refill or mottling.
Lactate represents a useful and clinically obtainable surrogate marker of tissue hypoxia and disease severity, independent of blood pressure [23]. Vital signs are rendered poor. Due to decreased tissue perfusion there is surge in catecholamines and neural alteration in regulation of maintaining arterial pressure the resuscitation end points, prognosticators of outcome are altered [24]. Use of a single measurement of venous lactic acid, the results of which can be made available soon after admission to the emergency department, provides the clinician with a better risk assessment, possibly enabling a clearer direction to diagnosis and therapy, than a patient's vital signs [25,26].
The surviving sepsis campaign (SSC) guidelines include multiple parameters with lactate levels as one of the main indicators. This primary bundle of parameters is useful for predicting the severity of sepsis and mortality outcome. However, SSC guidelines have included lactate clearance as a significant indicator for improvement over mortality. The extent of clearance in the first 6 hrs is a good predictor of survival rate in patients. Thus, the addition of the lactate clearance parameter to the primary bundle showed lactate clearance as independent predictor of the outcome [27]. Various other studies that have shown lactate as marker in sepsis have been summarized (Table 1) [28,29].
Table 1: Studies showing lactate as marker in sepsis. View Table 1
The interpretation of single lactate measurements has several limitations. First, blood lactate concentrations reflect the interaction between the production and elimination of lactate. For example, a sepsis patient with hepatic dysfunction may have a higher lactate compared with the patient without liver disease but may have a similar degree of stress. Second, an increased lactate concentration may indicate mechanisms other than cellular hypoxia, such as up-regulation in epinephrine-stimulated Na/K-adenosine triphosphatase activity in skeletal muscle and inhibition of pyruvate metabolism or an increase in its production. Therefore, serial lactate measurements are more important as an outcome prognosticator than a single lactate measurement [21,30,31]. Some of the studies done to measure the serial serum lactate levels have been summarized in the Table 2.
Table 2: Studies done to measure the serial serum lactate levels. View Table 2
Lactate is produced by most tissues in the body with muscle producing the highest amount. The serum lactate is usually cleared very rapidly. The main organ for clearance being liver followed by kidney. Any lactate level above 4 mmol/L is considered high and between 2-2.5 mmol/L is considered to be elevated. Elevated lactate is multi factorial, highly patient specific and disease specific phenomenon. Lactate elevation is usually due to decreased clearance or increased production of lactate or combination of both. Several other factors such as hypoperfusion, mitochondrial dysfunction, liver dysfunction and hypermetabolic state can also elevate levels of lactate in blood [32,33]. Some of the factors responsible for elevated lactate have been discussed briefly below.
Is often associated with enhanced lactate levels and lactate infact serves as useful biomarker in such cases. In a study of 1278 patients admitted with infection it was found that lactate levels could correctly stratify patients according to mortality. Lactate levels of 0-2.4, 2.5-3.9 and ≥ 4 mmol/L were associated with mortalities of 4.9%, 9.0% and 28.4% respectively [34].
Patients with myocardial dysfunction resulting in shock after cardiac surgery had elevated lactate levels. Investigators found that the elevation was primarily due to increased tissue lactate production and not due to decreased clearance of lactate. In patients with cardiogenic shock requiring extracorporeal membrane oxygenation, lactate has been found to be a useful parameter for predicting mortality.
It is another factor that could elevate lactate. Lactate levels were significantly elevated in both traumatic and non-traumatic hemorrhagic shock patients as compared to controls. The role of lactate in the post-cardiac arrest population has also been explored. The ischemia that occurs due to lack of blood flow during arrest in conjunction with the inflammation resulting from ischemia-reperfusion injury, is the most likely cause of the initial rise in lactate. Post-arrest patients with an initial lactate < 5 mmol/L had a mortality of 39% whereas mortality rose to 92% with an initial lactate > 10 mmol/L. The ability to clear lactate in the post-arrest period served as useful marker of increased survival. This was confirmed in two different studies of post-arrest patients.
Traumatic injury patients often show Hypoperfusion which is related to blood loss. Like sepsis and cardiac arrest, initial lactate levels are found to be significantly higher in non-survivors compared to survivors of trauma. One study reports a calculated sensitivity of 84% and specificity of 86% for death in patients with torso trauma at a lactate level > 4 mmol/L [34].
It can result in a profound elevation of lactate. However, the variation in lactate depends upon the nature of the seizure. Elevated lactate levels in seizures are transient with rapid clearance seen in short time. However, if levels do not come down in few hours post seizure then one needs to investigate for any other health conditions associated with increased lactate.
It increases lactate levels mainly due to anaerobic metabolism. It was found that lactate levels were elevated in 95% of collapsed marathon runners, with levels ranging from 1.1 to 11.2 mmol/L. Elevated lactate due to excessive muscle activity has also been associated with the use of restraints. A delirious or intoxicated patient may struggle against restraints and produce lactate due to muscle activity and tissue hypoxia.
It is also one of the factors for increasing the lactate levels. Researchers have found that elevated lactic acid levels to be 96% sensitive and 38% specific for mesenteric ischemia. Furthermore, elevated lactate in mesenteric ischemia has been correlated with increased mortality.
Lactate has been found to be a strong predictor of outcome. Studies have shown that the initial lactate level was a useful parameter to separate survivors from non-survivors. Smoke inhalation victims are at particular risk of elevated lactate due to potential inhalation of cyanide or carbon monoxide.
Elevated lactate may occur in diabetic ketoacidosis (DKA). The outcomes of this lactate increase are not associated with significant mortality like in other diseases. Studies done with cohorts of DKA and lactate levels were not conclusive.
It serves as a co-factor for multiple cellular enzymes including pyruvate dehydrogenase and α-ketoglutarate dehydrogenase. These enzymes are components essential to the tricarboxylic acid cycle and aerobic carbohydrate metabolism. In the absence of thiamine, anaerobic metabolism predominates and lactate production increases. Risk factors for thiamine deficiency include factors such as alcoholism, chronic liver disease, chronic wasting diseases, hyperemesis gravidarum, anorexia nervosa, and gastric bypass surgery.
The liver is the organ primarily responsible for lactate clearance. In cases of liver dysfunction lactate clearance may be impaired. Additionally, studies have shown that the acutely injured liver may itself act as a source of lactate [32].
Most of the drugs used to treat diabetes or another syndrome have been implicated to increase lactate levels. However, no direct correlation has been found and results are not conclusive.
Elevated levels of lactate can be caused by in born errors of metabolism. The genetic disorders associated with gluconeogenesis, pyruvate dehydrogenase, respiratory chain pathway and tricarboxylic acid cycle can result in increased lactate production in the body [35].
The study was carried out in Respiratory Intensive Care Unit (ICU) of National (LRS) Institute of TB and Respiratory Diseases, New Delhi, which provides a 24-hour care to the critically ill patients of respiratory diseases transferred from the wards/operation theatre or admitted through emergency. A cohort of minimum 60 subjects with sepsis were taken as cases and 60 subjects without sepsis served as controls. All of them were consecutively enrolled in the study irrespective of age, sex, nature of complaints or presence of co-morbidity on the same day of their admission in ICU.
Annexure I contained detailed clinical history, examination and investigations for all enrolled subjects. Investigations comprised of hemoglobin, TLC, DLC, Platelet count, peripheral smear for MP, Random Blood Sugar, LFT, S electrolyte, Culture of blood, urine, sputum or other body fluid, chest X ray, ultra sound or CT scan of chest or abdomen or another organ as needed. Arterial blood gas (ABG) measurement was done by Stat Profile Nova pHOx plus L ABG machine in all subjects on admission to the intensive care unit & every 3 to 12 hourly for the first 3 days. Blood lactate was measured by enzymatic methodology in the ABG machine at the above specified intervals.
Following criteria were used for the inclusion of subjects as cases (with Sepsis) or controls.
Subjects with more than one manifestation which includes
1. Body temperature greater than 38 ℃ or less than 36 ℃.
2. Heart rate greater than 90 beats per min.
3. Hyperventilation as indicated by respiratory rate > 20/min or PaCO2 less than 32 mmHg.
4. An alteration in the WBC count such count greater than 12000/cmm, a count less than 4000/cmm or presence of more than 10% immature neutrophils 'band'.
5. Evidence of infection.
Subject having no evidence of infection.
Subjects refusing to consent or participate or receiving biguanides, isoniazid, nucleoside analogue reverse transcriptase inhibitors, fructose, acetaminophen, salicylates, parenteral nutrition or papaverine therapy were excluded from study.
Based on criteria of Nguyen HB, et al. [16], lactate clearance was defined as lactate at presentation in ICU (hour 0) minus subsequent lactate, multiplied by 100. The enrolled subjects were categorized as being in the low-clearance group (< 10% lactate clearance) or high-clearance group (> 10% clearance) within 6 hours, as well as, between 12 to 18 hours of admission in ICU.
Presence of individual organ system dysfunction was assessed from the worst recorded value for each organ system at admission in ICU:
a. Cardiovascular = initial SBP less than 90 mmHg;
b. Pulmonary = PaO2/FIO2 < 300;
c. Renal = serum creatinine > 2.0 mg/dL;
d. Hepatic = serum bilirubin > 2.0 mg/dL;
e. Hematologic = platelets < 100,000/cmm;
The parameters based as per the Vincent, et al. [22] modification of Sequential Organ Failure Assessment (SOFA) score [29]. Multi Organ Dysfunction Syndrome (MODS) was defined as the presence of 2 or more organ dysfunctions.
Annexure II: Informed consent was taken from all the enrolled subjects. If the subject was not in the state of giving consent, informed consent was taken from his/her attendant. Full confidentiality was assured. The hospital outcome was recorded as recovery/discharge or death.
Statistical calculations were performed with SPSS statistical software (version 12.0). The mean values were compared in different groups by t-test. The proportions in different groups were compared by chi-squares tests. A p-value less than 0.05 was considered as statistically significant. Kaplan-Meier survival analysis was done between high and low lactate clearance groups.
Mean pH was found to be significantly lower in the subjects of sepsis than those of the control group. No significant differences were noted on comparison of the mean values of PO2, PCO2, standard bicarbonate, HCO3-actual, and base deficit between the sepsis and control groups. Mean values of admission lactate, serial lactate measured at 0 hrs, 6 hrs, 12 hrs and 18 hrs are higher in sepsis group than in control (Table 3 and Figure 1).
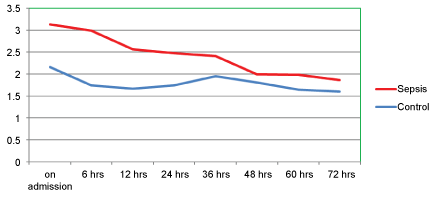 Figure 1: Association between serial blood lactate values and group.
View Figure 1
Figure 1: Association between serial blood lactate values and group.
View Figure 1
Table 3: Mean values of ABG Parameters vs. Group. View Table 3
The number of subjects with high lactate clearance (> 10% lactate clearance from admission value) were lower in sepsis as compared to control group, while those with low lactate clearance (< 10% lactate clearance from admission value) were higher in sepsis than control group. The difference between the groups was statistically significant for lactate clearance within 6 hours (p = 0.035) and insignificant for lactate clearance between 12 to 18 hours (p = 0.138) (Figure 2).
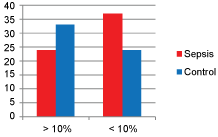 Figure 2A: Lactate clearance within 6 hours.
View Figure 2A
Figure 2A: Lactate clearance within 6 hours.
View Figure 2A
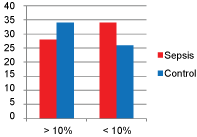 Figure 2B: Lactate clearance between 12 hours and 18 hrs vs. Group 6 hrs, 12 hrs and 18 hours vs. Group.
View Figure 2B
Figure 2B: Lactate clearance between 12 hours and 18 hrs vs. Group 6 hrs, 12 hrs and 18 hours vs. Group.
View Figure 2B
Mean length of ICU stay was insignificantly higher among the subjects of sepsis group than those of control group (Table 4).
Table 4: Length of Stay vs. Group. View Table 4
Majority of the subjects in both sepsis and control groups were discharged after recovery (36 and 59 respectively). The number of deaths were higher in sepsis than control group (26 vs. 1, p = 0.00) (Table 5).
Table 5: Hospital Out-come vs. Group. View Table 5
Majority of survivors and non-survivors were males. The mean age was survivors was statistically higher than non-survivors. A higher proportion of non-survivors had tachycardia, tachypnoea, hyperthermia and leukocytosis as compared to survivors for the respective SIRS parameter. A higher proportion of non-survivors had an organ dysfunction of cardiovascular, pulmonary, hepatic, renal and haematological systems as compared to survivors for respective organ system.
All non-survivors belonged to the medical (respiratory) diagnostic category with majority (44%) having chronic obstructive airways disease (COAD) with acute exacerbation followed by pneumonia (26%). On other hand, majority (60%) of survivors had a surgical (post thoracic) diagnosis and pneumonia constituted the commonest medical diagnosis. The difference was statistically significant between the groups (p = 0.000) (Table 6, Figure 3 and Figure 4).
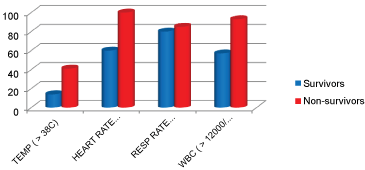 Figure 3: SIRS criteria vs. Out-come.
View Figure 3
Figure 3: SIRS criteria vs. Out-come.
View Figure 3
 Figure 4: Organ dysfunction vs. Out-come.
View Figure 4
Figure 4: Organ dysfunction vs. Out-come.
View Figure 4
Table 6: Characteristics of Survivors vs. Non-survivors. View Table 6
Mean values of Initial (Lactate 1), as well as, Serial lactate at all time intervals (Lactate 2 to 8) were observed to be significantly higher among non-survivors than survivors (p = 0.000). The association between mean blood lactate values and survival is shown in Figure 5.
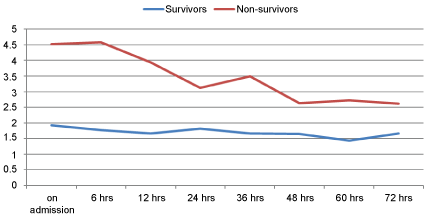 Figure 5: Association between mean blood lactate values and survival.
View Figure 5
Figure 5: Association between mean blood lactate values and survival.
View Figure 5
The proportion of subjects with high lactate clearance (> 10% clearance) was found to be greater among survivors as compared to non-survivors, while the proportion of subjects with low lactate clearance (< 10% clearance) was noted to be higher among non-survivors than survivors both within 6 hours Figure 6a, as well as, between 12 to 18 hours Figure 6b of admission. However, the differences between the groups was significant only for the latter (p = 0.182 and p = 0.039 respectively).
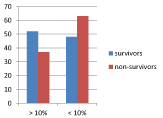 Figure 6A: Lactate clearance within 6 hours.
View Figure 6A
Figure 6A: Lactate clearance within 6 hours.
View Figure 6A
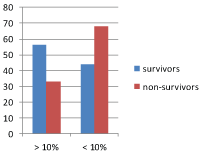 Figure 6B: Lactate clearance between 12 vs. Survival to 18 hours vs. Survival.
View Figure 6B
Figure 6B: Lactate clearance between 12 vs. Survival to 18 hours vs. Survival.
View Figure 6B
Kaplan-Meier survival analysis showed a survival benefit for the subjects in the higher lactate clearance quartiles. Figure 7a and Figure 7b depicts survival curves over the duration of hospital stay for high lactate clearance (lactate decrease by > 10%) and low lactate non-clearance (lactate decrease by < 10%) groups. The curves diverged insignificantly by log-rank test [P values of 0.0864 and 0.0527 following admission within 6 hours Figure 7a and between 12 to 18 hours Figure 7b respectively].
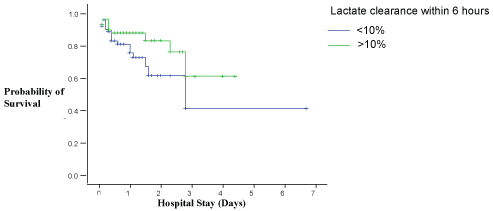 Figure 7A: Kaplan-Meier survival analysis between subjects with high and low lactate clearance within 6 hours of admission.
View Figure 7A
Figure 7A: Kaplan-Meier survival analysis between subjects with high and low lactate clearance within 6 hours of admission.
View Figure 7A
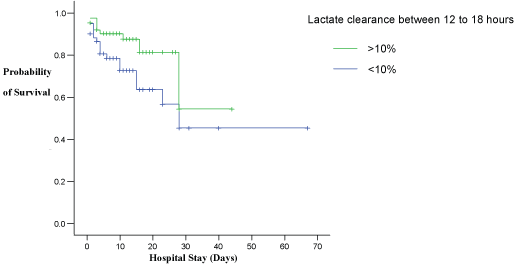 Figure 7B: Kaplan-Meier survival analysis between subjects with high and low lactate clearance between 12 to 18 hours of admission.
View Figure 7B
Figure 7B: Kaplan-Meier survival analysis between subjects with high and low lactate clearance between 12 to 18 hours of admission.
View Figure 7B
Majority of subjects among both sepsis and control groups (64.5% and 53.3% respectively) were aged between 21 to 50 yrs as seen from Table 7 and Figure 8. Mean age of subjects in both groups was not significantly different (38.85 ± 17.12 yrs versus 38.96 ± 17.48 yrs in sepsis and control groups respectively, p-0.972 as shown in Table 7 and Figure 8.
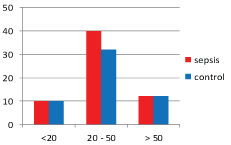 Figure 8: Shows age-distribution among sepsis and control groups.
View Figure 8
Figure 8: Shows age-distribution among sepsis and control groups.
View Figure 8
Table 7: Age distribution vs. Group. View Table 7
Majority of subjects in both sepsis and control groups were males (71% and 80% respectively) as seen from Table 8 and Figure 9.
 Figure 9: Shows gender distribution among sepsis and control groups.
View Figure 9
Figure 9: Shows gender distribution among sepsis and control groups.
View Figure 9
Table 8: Gender distribution vs. Group. View Table 8
The number of subjects with hyperthermia, tachycardia, tachypnoea and leucocytosis were higher in Sepsis groups than in controls (Table 9 and Figure 10).
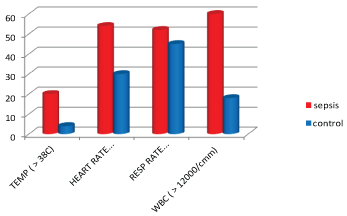 Figure 10: Shows SIRS Criteria among sepsis and control groups.
View Figure 10
Figure 10: Shows SIRS Criteria among sepsis and control groups.
View Figure 10
Table 9: SIRS criteria vs. Group. View Table 9
Organ dysfunction variables such as PO2/FiO2 (< 300), Hypotension, bilirubin, serum creatinine, platelet count and MODS were significantly higher in sepsis group than in controls (Table 10 and Figure 11).
Table 10: Organ dysfunction vs. Group. View Table 10
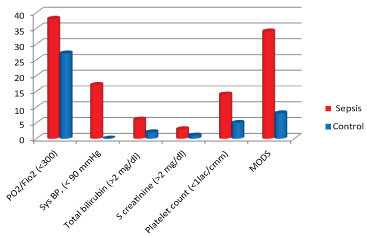 Figure 11: Organ Dysfunction vs. Group.
View Figure 11
Figure 11: Organ Dysfunction vs. Group.
View Figure 11
The routine investigations for haemoglobin, hyponatremia, hypokalemia and Hyponatremia showed it to be lower in sepsis patients however it was statistically insignificant (Table 11).
Table 11: Other Routine investigations vs. Study group. View Table 11
Majority of subjects in the sepsis group had a medical disease as compared to those in the control group (41 vs. 24 respectively, p = 0.00). Pneumonia was observed in 56% of the subjects in the sepsis group and was the commonest medical diagnosis in the group against none in the control group (Table 12, Table 13 and Figure 12).
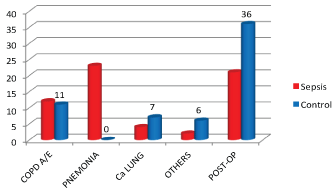 Figure 12: Diagnostic category vs. Group.
View Figure 12
Figure 12: Diagnostic category vs. Group.
View Figure 12
Table 12: Diagnostic Category vs. Group. View Table 12
Table 13: Arterial blood gas values of survivors vs. Non-survivors. View Table 13
Mean pH and PO2 were found to be significantly lower among non-survivors than survivors. No significant differences were noted on comparison of the mean values of PCO2, Standard bicarbonate, HCO3-actual, and base deficit between the non-survivors and survivors.
Most of the subjects in control and sepsis groups were middle aged males. Many of the disease parameters were encountered in sepsis conditions. Hyperthermia, tachycardia and leukocytosis were significant features of the sepsis group. Multi organ dysfunction syndrome and pneumonia were significantly seen in the sepsis group. Our study found significantly higher mean lactate values among patients of sepsis than control group (3.13 ± 2.86 vs. 2.16 ± 1.86 respectively, p = 0.03). The average lactate levels for the first 3 days of admission were higher in sepsis group. Lactate levels were monitored were measured within 6, 12 and 18 hrs of joining in the hospital. The subjects in the sepsis group exhibited low lactate clearance within the first 6 hours. There were higher number of deaths in ICU in sepsis group with the prolonged stay in hospital. The medical diagnosis among non-survivors was COPD with acute exacerbation followed by pneumonia.
In short, the study highlights the importance of a serial measurement of serum lactate in subjects admitted to an ICU with sepsis. Significantly higher lactate levels were found in subjects with sepsis than those without infection and in non-survivors than survivors. Higher lactate clearance was seen more commonly among survivors than non-survivors. Hence, serum lactate measurement should form a part of the standard management protocol of the critically ill patients admitted with suspected sepsis.
This study has been done with a small population size. The study needs to be supported by larger population sizes and should be split and analysed community wise. Based on such studies conclusions can be drawn about alterations in lactate levels across communities and in larger group sizes. Central Venous Pressure Line insertions were carried out in only those subjects, in whom requirement was felt clinically and not as mandated for every case of sepsis under Early Goal Directed Therapy Guidelines. The use of early fluid resuscitation if guided by the CVP insertion in every subject, the serial lactate values might have shown different results than seen in present study.